

Chinese Journal of Organic Chemistry >
Recent Progress in Copper-Catalyzed Remote Yne-Allylic Substitution Reaction
† These authors contributed equally to this work.
Received date: 2025-07-15
Revised date: 2025-09-25
Online published: 2025-11-05
Supported by
National Natural Science Foundation of China(22301095)
Opening Funding of Project of National Key Laboratory of Green Pesticide in 2025(GPLKF202509)
Copper-catalyzed remote yne-allylic substitution system is an efficient approach for constructing functional molecules containing unsaturated bonds, and a variety of new catalytic systems and reaction modes have been successfully developed. Yne-allylic ester substrates with remote leaving groups are converted into copper vinyl-allenylidene intermediates under the cooperative catalysis of copper/ligands. The nucleophilic addition of nucleophiles to the γ-position or ε-position of the intermediates can efficiently construct important skeletons such as structurally diverse 1,3-enynes, 1,4-enynes, allenes, spirocyclic compounds, and C—C/C—N/N—N axially chiral compounds. From the perspective of classification by nucleophile types, the latest research progress of copper-catalyzed remote yne-allylic substitution reactions involving various nucleophiles such as carbon, nitrogen, and sulfur is systematically summarized. It focuses on the discussion of reaction mechanisms and prospects for the future development direction of this field.
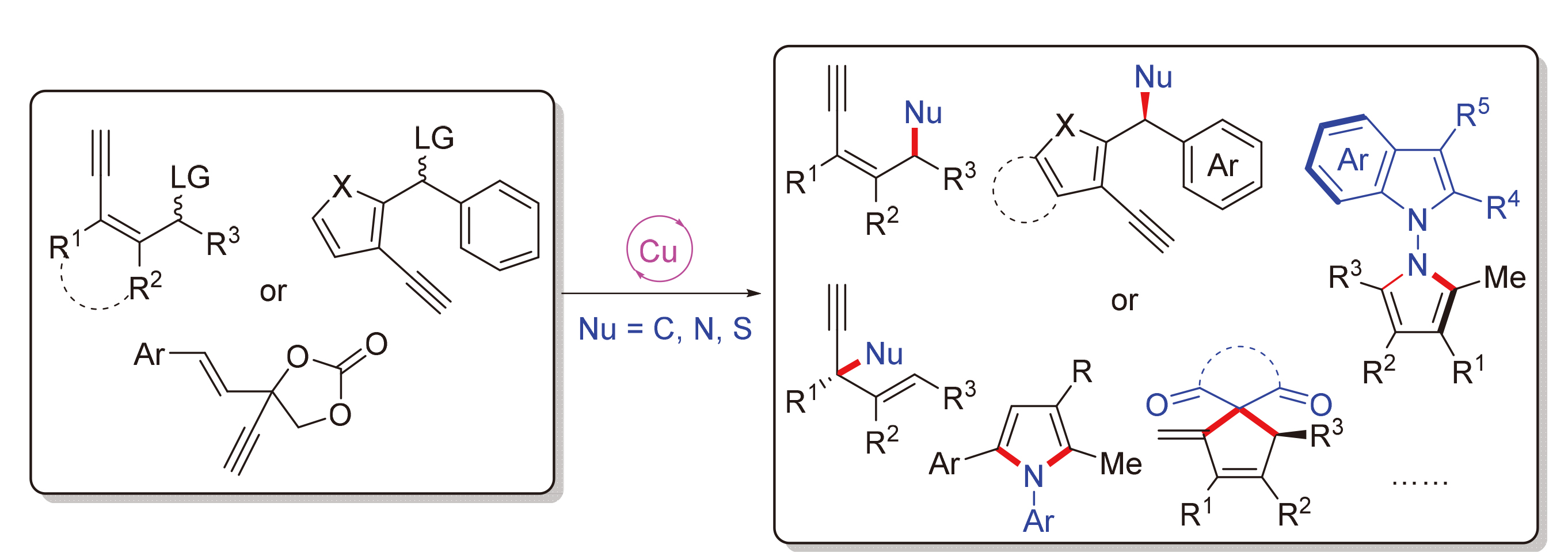
Mengdie Li , Zihan Wang , Taoyan Lin . Recent Progress in Copper-Catalyzed Remote Yne-Allylic Substitution Reaction[J]. Chinese Journal of Organic Chemistry, 2026 , 46(2) : 356 -378 . DOI: 10.6023/cjoc202507019
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
(a)
(b)
|
| [6] |
(a)
(b)
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
/
| 〈 |
|
〉 |