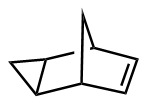
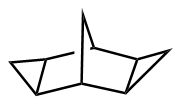
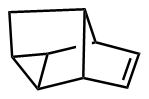
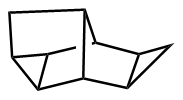
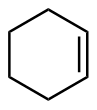
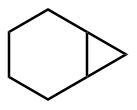
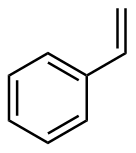
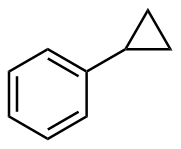
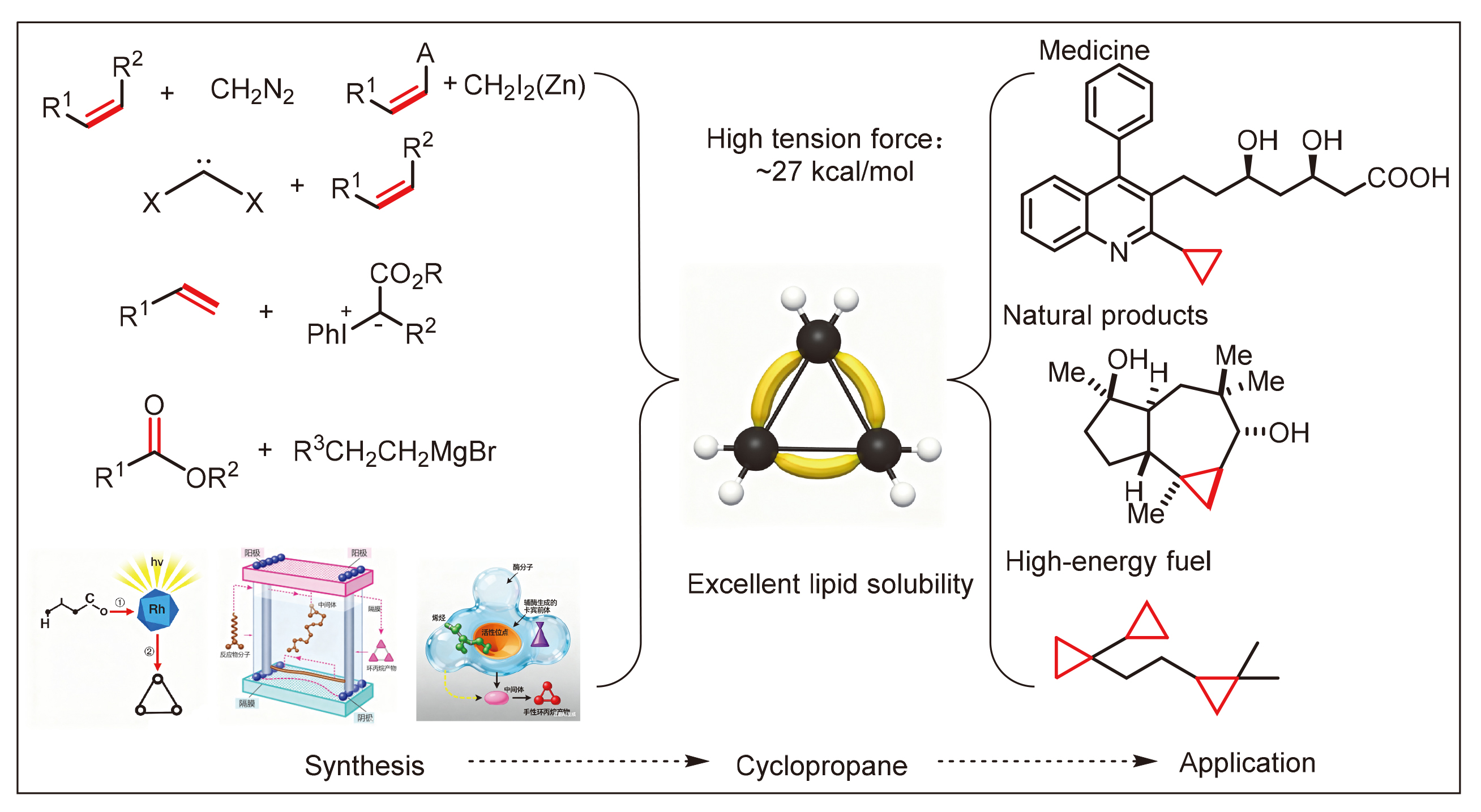
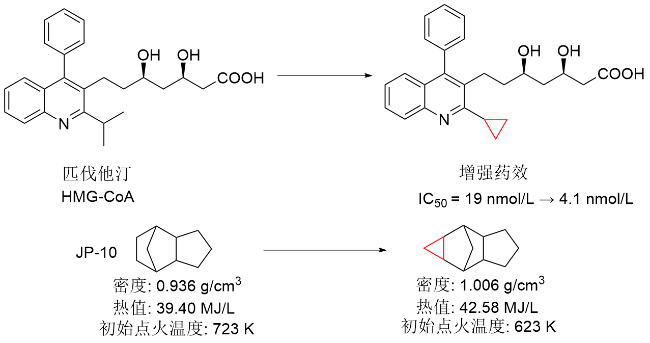
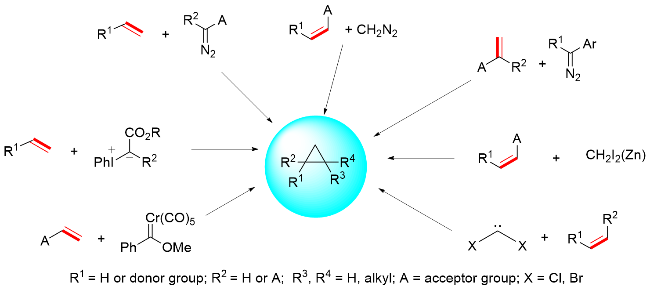
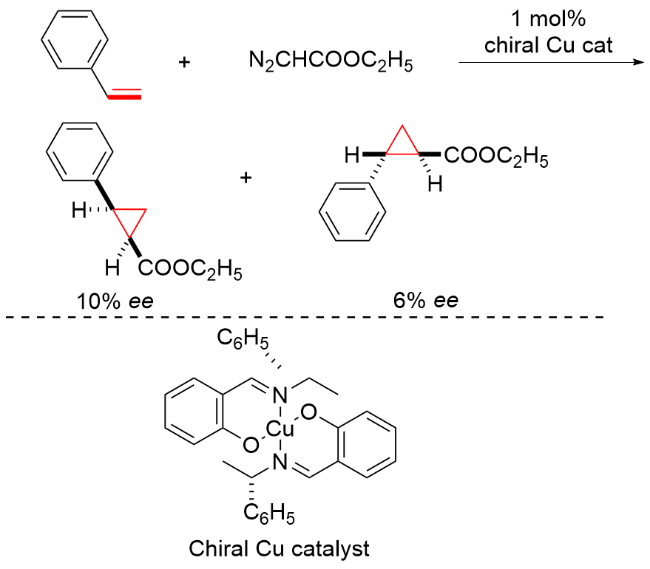
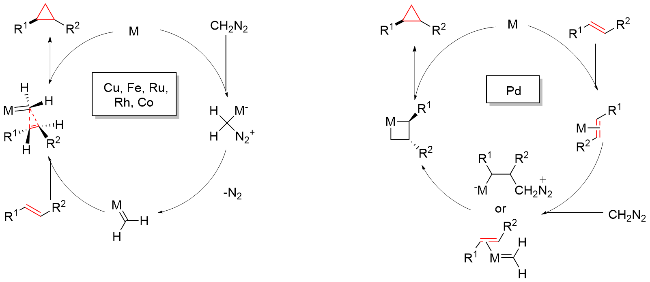
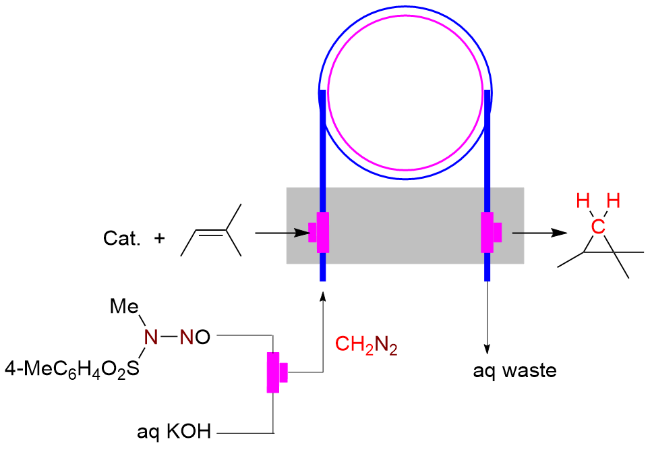
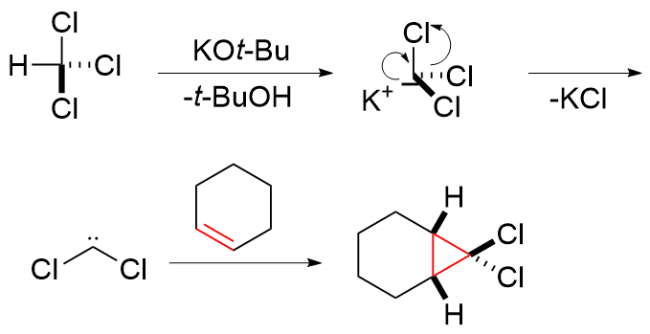
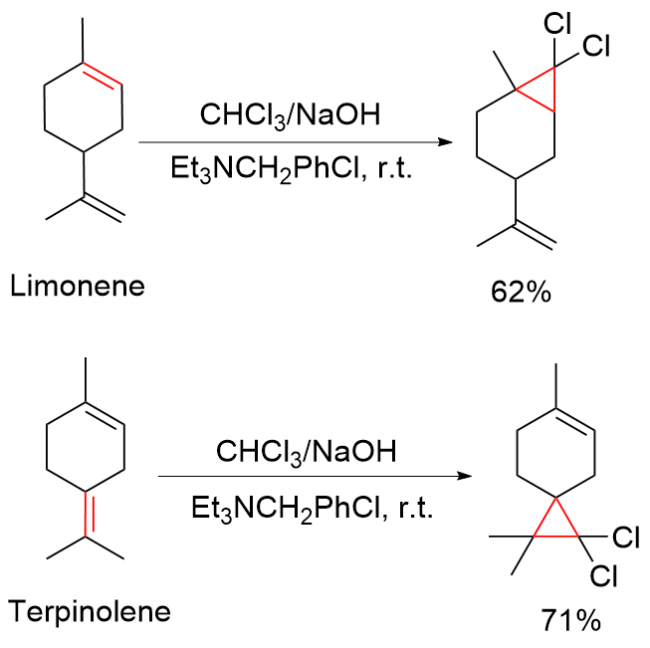
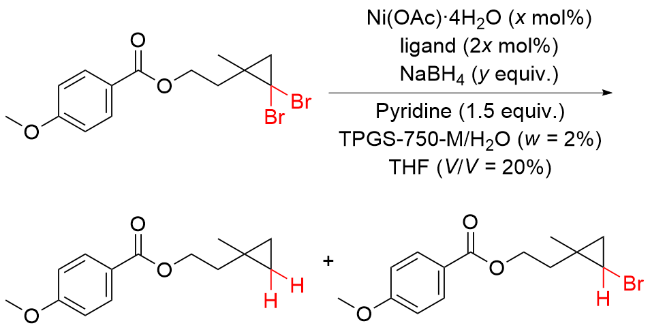
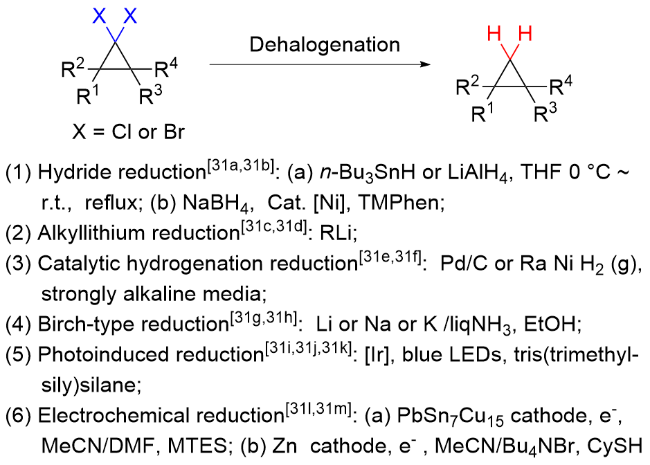
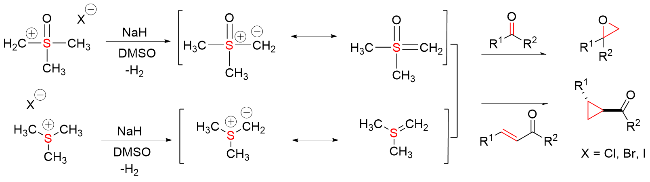
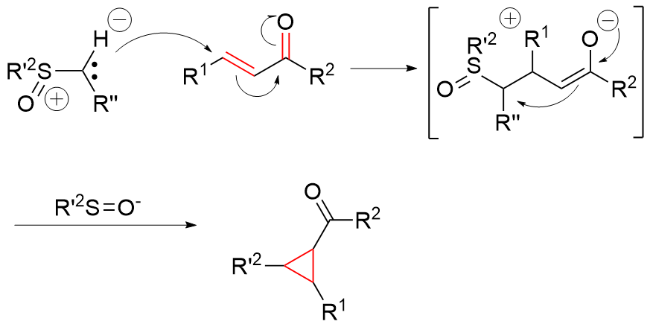
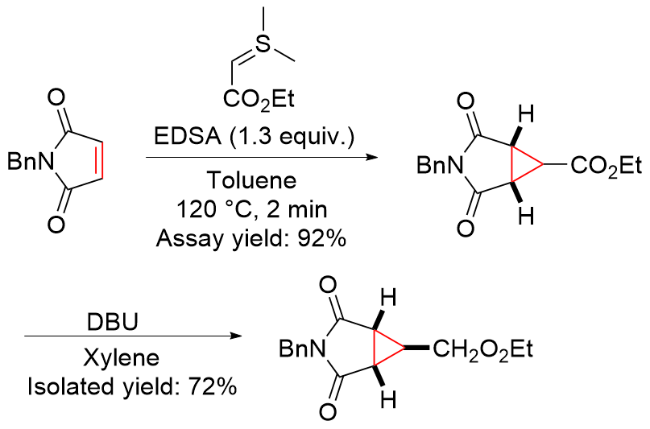
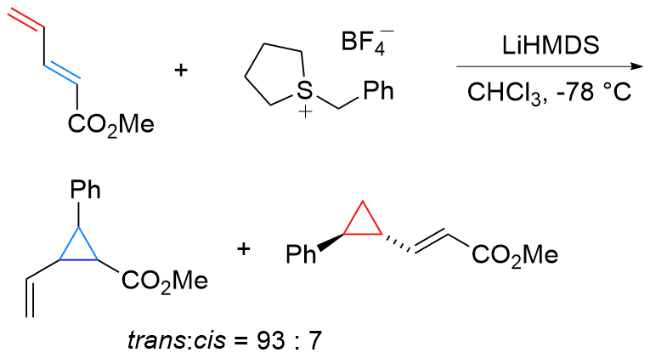
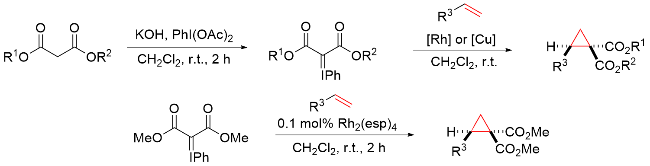
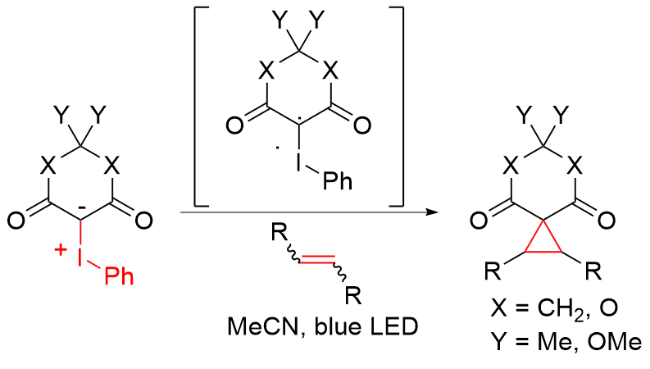
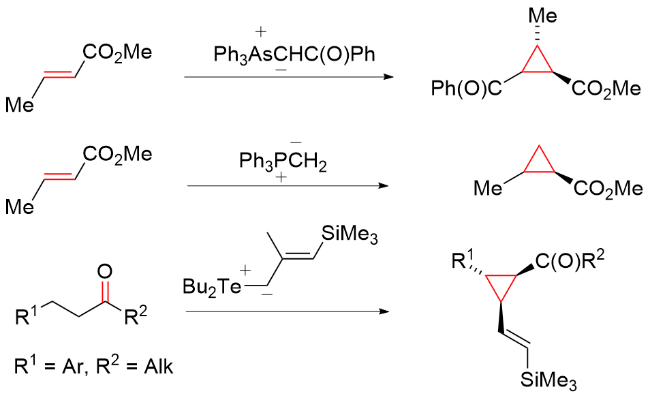
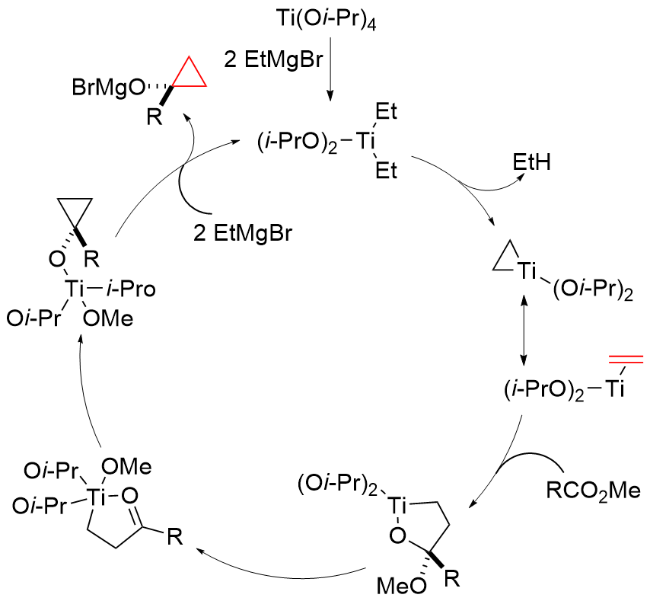
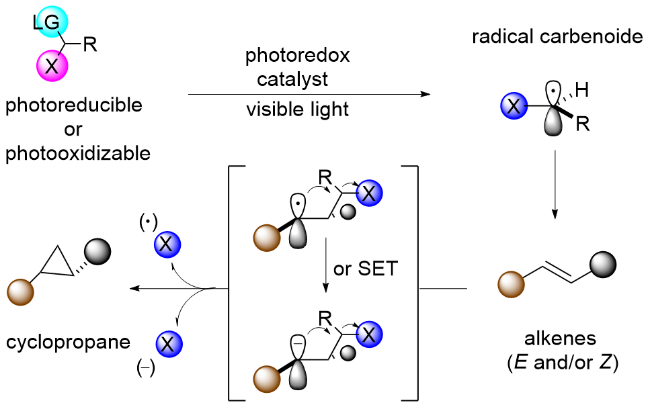
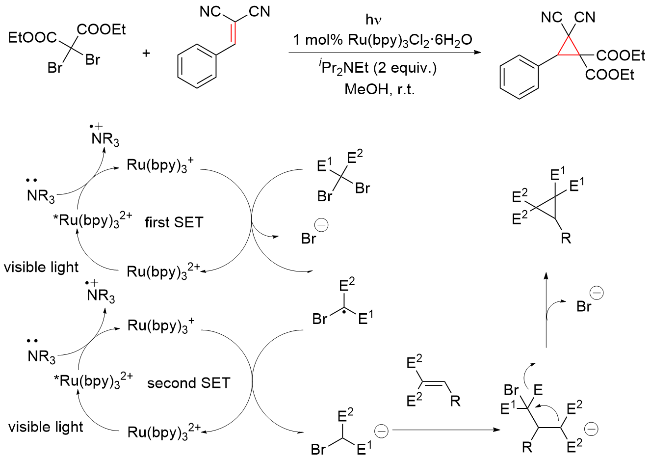
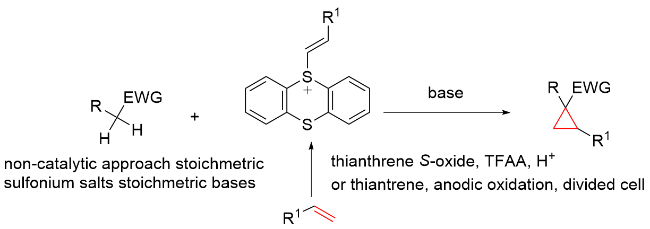
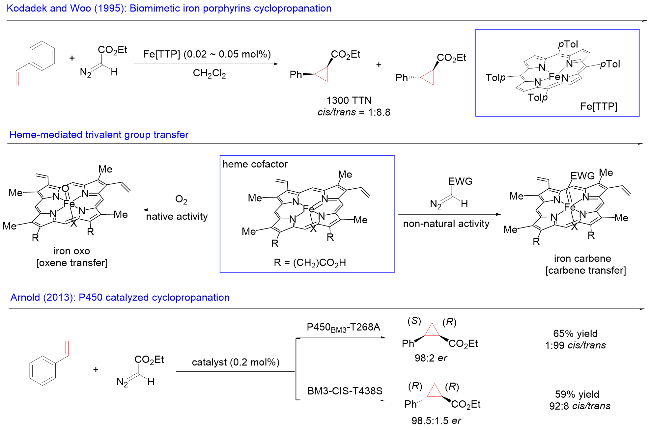
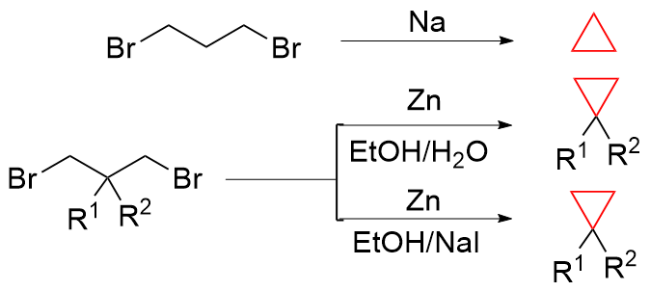
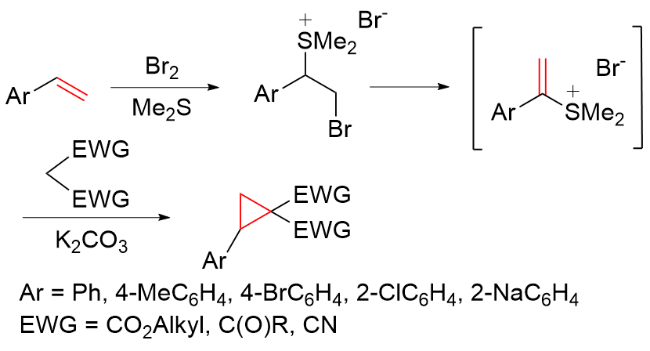
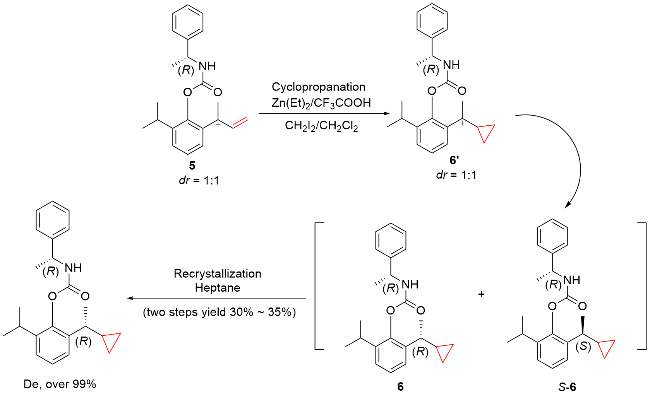
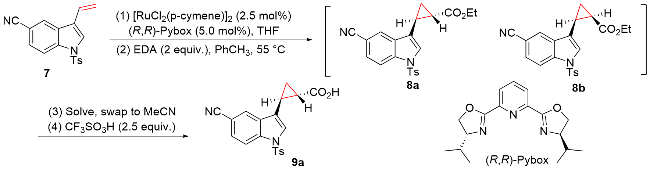
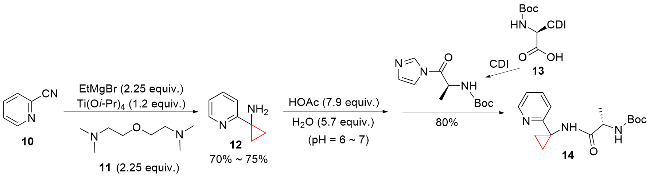
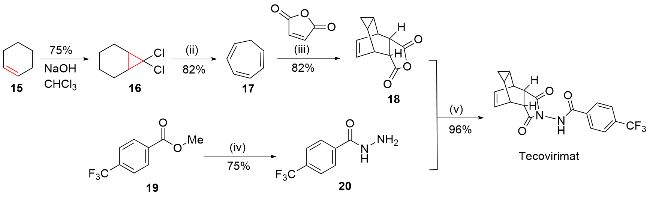
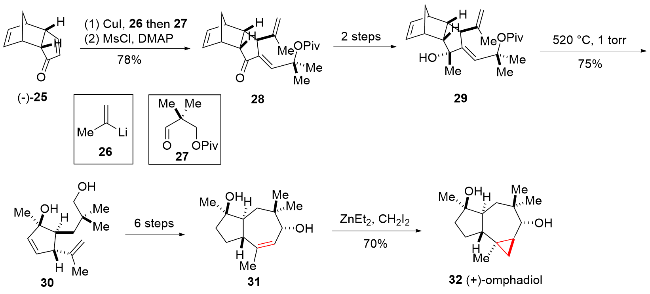
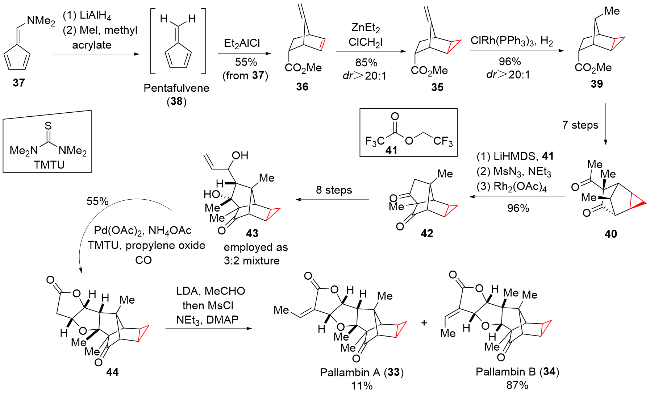
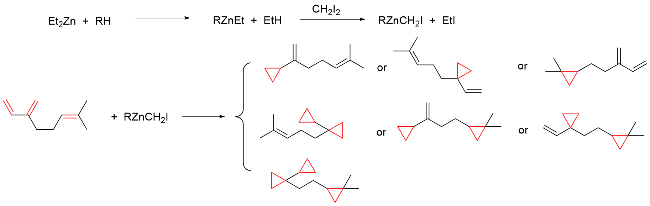
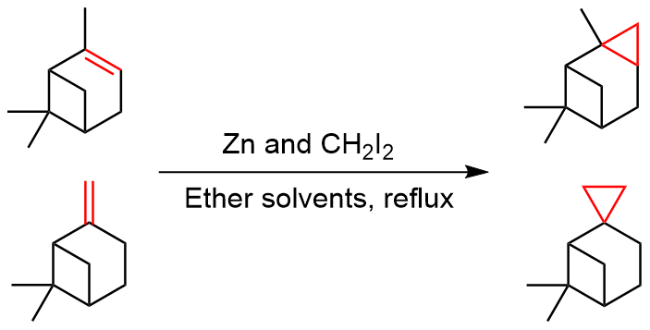
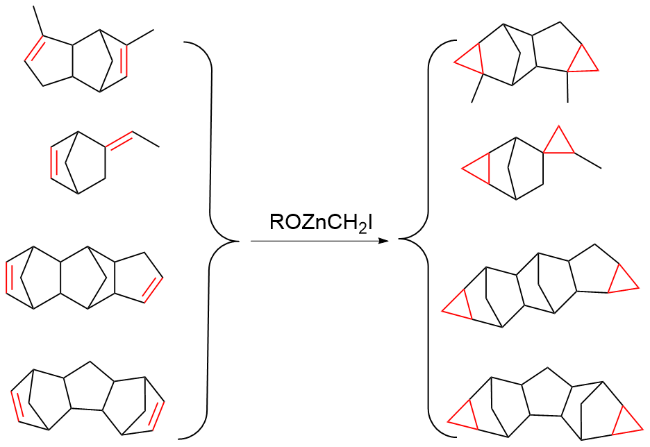
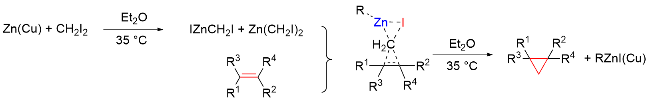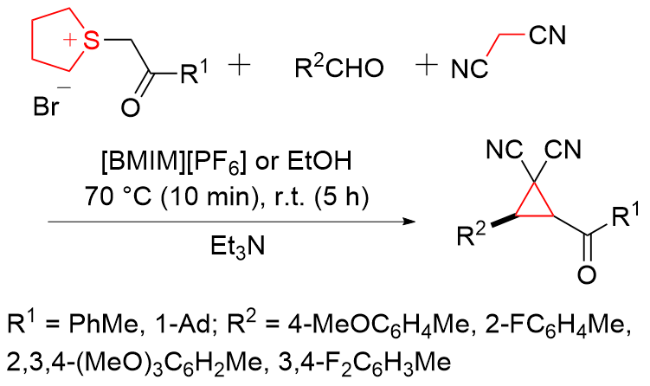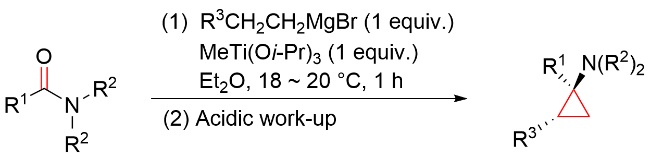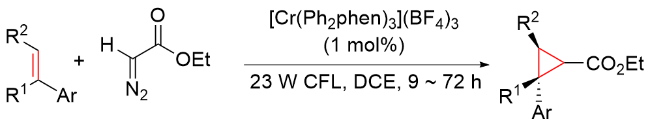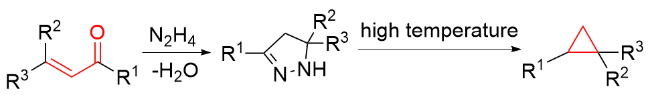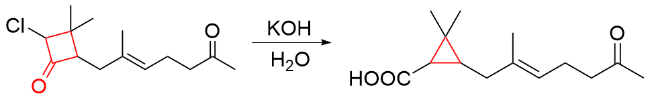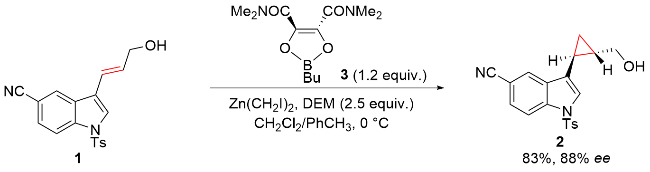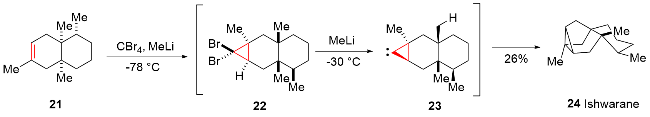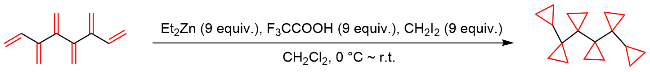该反应对于底物烯烃结构以及所含官能团等表现出一些独有的特性
[34]. 例如, 当烯烃含有杂原子官能团, 如羟基、乙酸根和甲氧基等, 这些官能团有很强的诱导效应, 有机锌试剂会在离官能团较近的一侧进行反应, 这样可以提高反应的活性或者立体选择性; 环状烯醇参与反应时, 立体化学结果和环大小有关: 五元环、六元环和七元环具有很高的顺式非对映选择性, 而更大的环一般得到的是反式非对映选择性产物. 此外, 碘或氯甲基碘化钐(Sm/Hg/CH
2I
2)在多个双键存在时, 对烯丙醇位的双键具有很好的化学选择性, 而二烃基(碘甲基)铝(
i-Bu
3Al/CH
2I
2)在烯丙醇位双键存在时对无官能团烯烃有很好的选择性. 当然该反应也存在难以解决的痼疾, 例如常规采用的二碘甲烷具有自燃性, 且与锌反应经常表现出反应延迟并突然放热的现象, 这在大规模的生产中尤为明显, 给安全生产带来巨大压力. 1982年, Repič等
[35]发现采用超声波激活锌, 可使反应顺利进行, 且在一定程度上减缓了延迟放热效应. 另外, 由于类碳锌的亲电性质, 溶剂的选择对反应有很大的影响, 随着溶剂碱度的增加, 环丙烷化速率降低. 为提高反应效率, 研究人员在Et
2Zn-CH
2I
2体系中添加有机酸RCOOH, 发现反应效果提高显著
[3]. 例如以甲基苯乙烯为底物参与环丙烷化时, 所加入RCOOH的酸性越强, 底物的转化率就越高, 添加CF
3COOH后的反应效果最明显, 在反应1 h后甲基苯乙烯的转化率便达到了100%. 但CF
3COOH催化的反应存在成本过高, 环境危害较大等问题. 为解决这一问题, Liu等
[36]采用湿浸渍法制备了一系列高活性的固体酸催化剂, 其中5% HPW/MCM-41在用于双环戊二烯的环丙烷化中转化率和双环产物的选择率分别为93.4%和55.7%, 均优于CF
3COOH催化.