[1] Jiang H.-L.; Luo X.-H.; Wang X.-Z.; Yang J.-L.; Yao, X.-J; Crews P.; Valeriote F. A.; Wu Q.-X. Fitoterapia,2012, 83, 1275-1280.
[2] (a) Ma C.; Li Y.; Niu S.; Zhang H.; Liu X.; Che, Y. J. Nat. Prod.,2011, 74, 32-37.
(b) Li, C.-S.; An, C.-Y.; Li, X.-M.; Gao, S.-S.; Cui, C.-M.; Sun, H.-F.; Wang, B.-G. J. Nat. Prod., 2011, 74, 1331-1334.
(c) Zheng, B.; Wei, M.; Lin, W.; Pan, C. Chin. J. Org. Chem., 2025, 45, 2815-2824 (in Chinese). (郑彬, 韦敏, 林雯雯, 潘成学, 有机化学, 2025, 45, 2815-2824.)
[3] (a) Rahbæk L.; Breinholt J.; Frisvad J. C.; Christophersen, C. J. Org. Chem.,1999, 64, 1689-1692.
(b) Dai, X.; Wong, A.; Virgil, S. C. J. Org. Chem., 1998, 63, 2597-2600.
[4] (a) Kacker I. K.; Zaheer, S. H. J. Ind. Chem. Soc.,1951, 28, 344-349.
(b) Amin, A. H.; Mehta, D. R.; Samarth, S. S. Progress in Drug Research, 1970, 14, 218-225.
[5] (a) Alvi S.; Sil A.; Maity S.; Singh V.; Guchhait B.; Ali R. ACS Omega,2024, 9, 9098-9108.
(b) D. de Química, U. A. de Barcelona, Org. Lett., 2010, 12, 3148-3151.
(c) Lu, Q.; Liu, K.; Zhang, H.; Du, Z.; Wang, X.; Wang, F. ACS. Nano., 2009, 3, 3861-3868.
[6] (a) Ghosh, M. K.; Mittal, K. L. Polyimides, Fundamentals and Applications, Marcel Dekker, New York, 1996.
(b) Tomi kawa, M.; Yoshida, S.; Okamoto, N. Polym. J., 2009, 41, 604-608.
[7] (a) Dhokale B.; Coe-Sessions K.; Wenzel M. J.; Davies A. E.; Kelsey T.; Brant J. A.; Oliveira L. D. S.; Parkinson B. A.; Hobert, J. O. J. Am. Chem. Soc.,2024, 146, 33048-33055.
(b) Guan, Q.; Zhou, L.-L.; Dong, Y.-B. Chem. Soc. Rev., 2022, 51, 6307-6416.
(c) Li, Y.; Chen, W.; Xing, G.; Jiang, D.; Chen, L. Chem. Soc. Rev., 2020, 49, 2852-2868.
(d) Geng, K.; He, T.; Liu, R.; Dalapati, S.; Tan, K. T.; Li, Z.; Tao, S.; Gong, Y.; Jiang, Q.; Jiang, D. Chem. Rev., 2020, 120, 16, 8814-8933.
[8] Davies J. A.; Ronson T. K.; Nitschke, J. R. J. Am. Chem. Soc.,2024, 146, 5215-5223.
[9] Gao Z.; Qian J.; Yang H.; Zhang J.; Jiang G. Org. Lett.,2021, 23, 1731-1737.
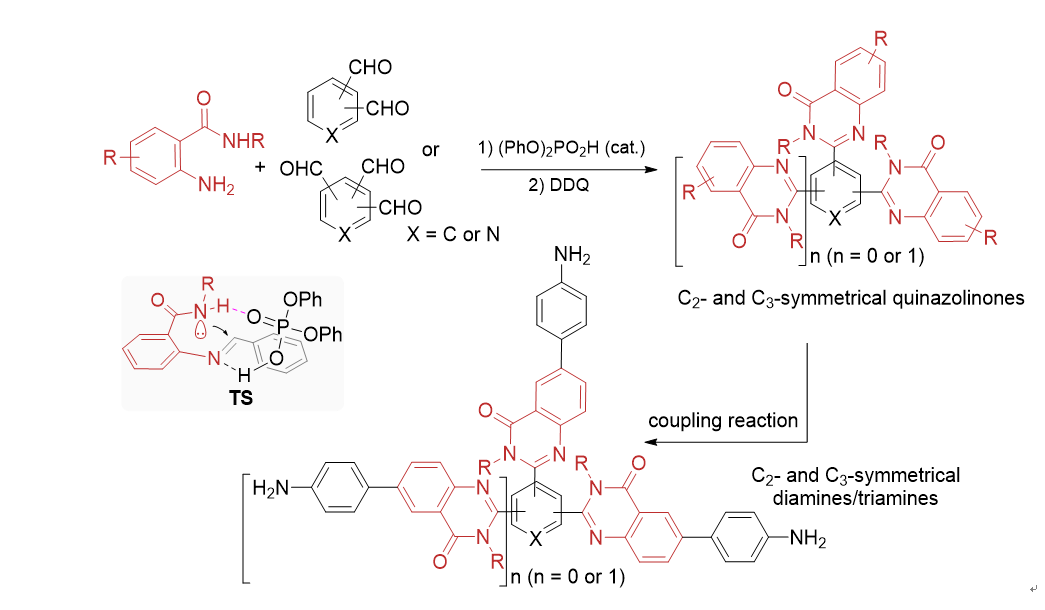
[10] The details for optimization of reaction conditions, please see the supporting information.
[11] (a) Liu T.; Fan L.; Yang H.; Wang L.; Fan X.; Sun L. Artificial Photosynthesis,2025, 1, 14-26.
(b) Gorczyński, A.; Harrowfield, J. M.; Patroniak, V.; Stefankiewicz, A. R. Chem. Rev., 2016, 116, 14620-14674.
(c) Eryazici, I.; Moorefield, C. N.; Newkome, G. R. Chem. Rev., 2008, 108, 1834-1895.