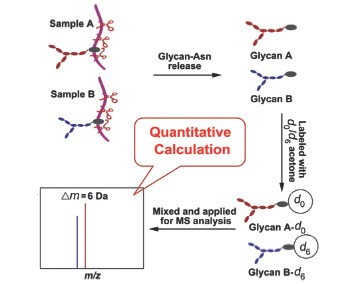
| [1] Apweiler, R.; Hermjakob, H.; Sharon, N. Acta Biochim. Biophys. Sin. 1999, 1473, 4. [2] (a) Powlesland, A. S.; Quintero-Martinez, A.; Lim, P. G.; Pipirou, Z.; Taylor, M. E.; Drickamer, K. Methods Enzymol. 2010, 480, 165;(b) Wu, A. M.; Liu, J.-H.; Gong, Y.-P.; Li, C.-C.; Chang, E.-T. FEBS Lett. 2010, 584, 3561. [3] (a) Katsuhiro, O.; Tetsuya, K.; Takashi, N.; Seiichi, O. Int. J. Biochem. 1987, 23, 569;(b) Wu, A. M.; Wu, J. H.; Singh, T.; Liu, J. H.; Tsai, M. S.; Gilboa-Garber, N. Biochimie 2006, 88, 1479. [4] (a) Brockhausen, I. BBA-Gen. Subjects 1999, 1473, 67;(b) Pinho, S.; Marcos, N. T.; Ferreira, B.; Carvalho, A. S.; Oliveira, M. J.; Santos-Silva, F.; Harduin-Lepers, A.. Reis, C. A. Cancer Lett. 2007, 249, 157;(c) Wang, F.-L.; Cui, S.-X.; Sun, L.-P.; Qu, X.-J.; Xie, Y.-Y.; Zhou, L.; Mu, Y.-L.; Tang, W.; Wang, Y.-S. Cancer Detect. Prev. 2009, 32, 437. [5] (a) Kim, Y.-G.; Jeong, H.-J.; Jang, K.-S.; Yang, Y.-H.; Song, Y.-S.; Chung, J.; Kim, B.-G. Anal. Biochem. 2009, 391, 151;(b) Di Michele, M.; Marcone, S.; Cicchillitti, L.; Della Corte, A.; Ferlini, C.; Scambia, G.; Donati, M. B.; Rotilio, D. J. Proteomics 2010, 73, 879. [6] (a) Miyoshi, E.; Shinzaki, S.; Moriwaki, K.; Matsumoto, H. Methods. Enzymol. 2010, 478, 153;(b) Hirota, M.; Mogaki, M.; Pour, P. M.; Chaney, W. G. Exp. Mol. Pathol. 1993, 58, 169. [7] (a) Wang, Z. F.; Zhang, Y.; Lin, X.; Huang, L. J. Acta Chim. Sinica 2007, 65, 2761. (王仲孚, 张英, 林雪, 黄琳娟, 化学学报, 2007, 65, 2761.);(b) Qiu, J.; Zhang, Y.; Lu, H. J.; Yang, P. Y. Acta Chim. Sinica 2011, 69, 2123. (仇娟, 张莹, 陆豪杰, 杨芃原, 化学学报, 2011, 69, 2123.);(c) Kuang, M.; Zhang, Y.; Yang, P. Y.; Lu, H. J. Acta Chim. Sinica 2013, 71, 1007. (匡敏, 张莹, 杨芃原, 陆豪杰, 化学学报, 2013, 71, 1007.)[8] (a) Bowman, M. J.; Zaia, J. Anal. Chem. 2010, 82, 3023;(b) Xia, B.; Feasley, C. L.; Sachdev, G. P.; Smith, D. F.; Cummings, R. D. Anal. Biochem. 2009, 387, 162;(c) Yuan, J.; Hashii, N.; Kawasaki, N.; Itoh, S.; Kawanishi, T.; Hayakawa, T. J. Chromatogr. A 2005, 1067, 145;(d) Prien, J. M.; Prater, B. D.; Qin, Q.; Cockrill, S. L. Anal. Chem. 2010, 82, 1498;(e) Botelho, J. C.; Atwood Iii, J. A.; Cheng, L.; Alvarez-Manilla, G.; York, W. S.; Orlando, R. Int. J. Mass Spectrom. 2008, 278, 137;(f) Zhang, P.; Zhang, Y.; Xue, X.; Wang, C.; Wang, Z.; Huang, L. Anal. Biochem. 2011, 418, 1;(g) Bowman, M. J.; Zaia, J. Anal. Chem. 2007, 79, 5777;(h) Alvarez-Manilla, G.; Warren, N. L.; Abney, T.; Atwood, J.; Azadi, P.; York, W. S.; Pierce, M.; Orlando, R. Glycobiology 2007, 17, 677. [9] (a) Flory, M. R.; Griffin, T. J.; Martin, D.; Aebersold, R. Trends Biotech. 2002, 20, S23;(b) Goshe, M. B.; Smith, R. D. Curr. Opin. Biotech. 2003, 14, 101;(c) Gouw, J. W.; Krijgsveld, J.; Heck, A. J. R. Mol. Cell. Proteomics 2010, 9, 11;(d) Julka, S.; Regnier, F. J. Proteome Res. 2004, 3, 350. [10] (a) Ong, S.-E.; Blagoev, B.; Kratchmarova, I.; Kristensen, D. B.; Steen, H.; Pandey, A.; Mann, M. Mol. Cell. Proteomics 2002, 1, 376;(b) Krüger, M.; Moser, M.; Ussar, S.; Thievessen, I.; Luber, C. A.; Forner, F.; Schmidt, S.; Zanivan, S.; Fässler, R.; Mann, M. Cell 2008, 134, 353. [11] Gygi, S. P.; Rist, B.; Gerber, S. A.; Turecek, F.; Gelb, M. H.; Aebersold, R. Nat. Biotechnol. 1999, 17, 994. [12] Schmidt, A.; Kellermann, J.; Lottspeich, F. Proteomics 2005, 5, 4. [13] Ross, P. L.; Huang, Y. N.; Marchese, J. N.; Williamson, B.; Parker, K.; Hattan, S.; Khainovski, N.; Pillai, S.; Dey, S.; Daniels, S.; Purkayastha, S.; Juhasz, P.; Martin, S.; Bartlet-Jones, M.; He, F.; Jacobson, A.; Pappin, D. J. Mol. Cell. Proteomics 2004, 3, 1154. [14] Liu, X.; McNally, D. J.; Nothaft, H.; Szymanski, C. M.; Brisson, J.-R.; Li, J. Anal. Chem. 2006, 78, 6081. [15] Liu, X.; Li, X.; Chan, K.; Zou, W.; Pribil, P.; Li, X.-F.; Sawyer, M. B.; Li, J. Anal. Chem. 2007, 79, 3894. [16] Xu, S.; Zhang, P.; Huang, L.; Wang, Z. Chem. J. Chin. Univ. 2010, 31, 1992. (徐莎, 张萍, 黄琳娟, 王仲孚, 高等学校化学学报, 2010, 31, 1992.)[17] Zhai, J.; Liu, X.; Huang, Z.; Zhu, H. J. Am. Soc. Mass Spectr. 2009, 20, 1366. [18] (a) Beardsley, R. L.; Karty, J. A.; Reilly, J. P. Rapid Commun. Mass Spectrom. 2000, 14, 2147;(b) Brancia, F. L.; Oliver, S. G.; Gaskell, S. J. Rapid Commun. Mass Spectrom. 2000, 14, 2070. [19] Fretheim, K.; Iwai, S.; Feeney, R. E. Int. J. Pept. Protein Res. 1979, 14, 451. [20] (a) Huntington, J. A.; Stein, P. E. J. Chromatogr. B 2001, 756, 189;(b) Charlwood, J.; Langridge, J.; Camilleri, P. Rapid Commun. Mass Spectrom. 1999, 13, 1522;(c) Saba, J. A.; Shen, X.; Jamieson, J. C.; Perreault, H. J. Mass Spectrom. 2001, 36, 563. [21] Packer, N. H.; Lawson, M. A.; Jardine, D. R.; Redmond, J. W. Glycoconjugate J. 1998, 15, 737. |