

八面体纳米笼构筑的金属-有机框架化合物
收稿日期: 2012-08-12
网络出版日期: 2012-09-24
基金资助
项目受国家重点基础研究发展计划(973计划, Nos. 2011CB932504, 2012CB821705);国家自然科学基金(Nos. 91022007, 21003128和20901078);福建省纳米材料重点实验室(No. 2006L2005)和中国科学院重要方向性课题的资助.
Construction of Two Octahedral Cage-based Metal-organic Frameworks
Received date: 2012-08-12
Online published: 2012-09-24
Supported by
Project supported by the 973 Program (Nos. 2011CB932504, 2012CB821705), National Natural Science Foundation of China (Nos. 91022007, 21003128, and 20901078), Fujian Key Laboratory of Nanomaterials (No. 2006L2005), and Key Projects from CAS.
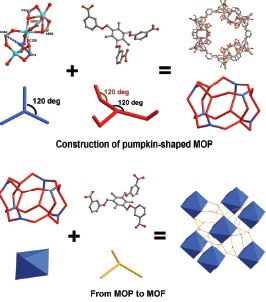
利用半刚性的三足羧酸配体1,3,5-tris[3-(carboxyphenyl)oxamethyl]-2,4,6-trimethylbenzene acid (H3TBTC)与金属离子自组装成功合成了两例八面体纳米笼构建的金属-有机框架化合物: {[Zn3TBTC2(DMA)(H2O)]·3DMA·3H2O}n (1), {[Cd3TBTC2(DMA)2(H2O)2]·2DMA·2H2O}n (2). TBTC3-配体在化合物中呈现出cis,cis,cis-和cis,trans,trans-两种构型. cis,cis,cis-构型的TBTC3-配体与Zn3(COO)6次级结构基元(secondary building units, SBUs)构筑了畸变的八面体金属-有机纳米笼, 因而化合物的结构可以看成是八面体金属-有机纳米笼作为超分子构建单元(supramolecular building blocks, SBBs), 在空间上与cis,trans,trans-构型的配体相连, 最终形成具有(3,18)-连接的三维无限网络. 荧光测试结果表明, 化合物1~2的荧光都是基于配体的发射. 相对于配体的发射光谱, 化合物1~2的发射光谱展现出蓝移的现象. 气体吸附测试结果表明, 化合物1~2具有选择性吸附二氧化碳的能力.
林祖金 , 林熙 , 曹荣 . 八面体纳米笼构筑的金属-有机框架化合物[J]. 化学学报, 2012 , 70(19) : 2012 -2015 . DOI: 10.6023/A12080546
Two isostructural octahedral metal-organic frameworks formulated as {[Zn3TBTC2(DMA)(H2O)]·3DMA·3H2O}n (1), {[Cd3TBTC2(DMA)2(H2O)2]·2DMA·2H2O}n (2) have been successfully prepared via solvothermal synthesis based on a predesigned semi-rigid ligand, 1,3,5-tris[3-(carboxyphenyl)oxamethyl]-2,4,6-trimethylbenzene acid (H3TBTC). In the structure, the semi-rigid ligand adopts cis,cis,cis- and cis,trans,trans-conformations. Six cis,cis,cis- conformational TBTC3- ligands connect six M3(COO)6 (M=Zn(II) or Cd(II) secondary building units (SBUs) to form a pumpkin-shaped metal- organic nanocage. Viewing only the SBUs as vertexes, the pumpkin-shaped nanocage can be seen as a slightly distorted octahedral nanocage. Acting as supramolecular building blocks (SBBs), every octahedral nanocage connects 18 cis,trans,trans-conformational TBTC3- ligands, while every cis,trans,trans-conformational TBTC3- ligand connects three octahedral cages. Thus, the framework can be simplified as a (3,18)-connected network with point symbol of {433.684.836}{43}6. Moreover, upon removal of free guest solvent molecules, compounds 1~2 have metal clusters locating in the tunnel walls, which may have strong interaction with molecules with high quadrapole moment, and they show preferential adsorption of CO2 over N2/H2. The resulting materials have potential for separation of CO2 from other gas molecules such as N2 and H2. The solid state photoluminescent spectra of 1~2 as well as the free H3TBTC ligand were examined at room temperature. When excited at 320 nm, the free ligand shows a maximum emission at 434 nm, which can be ascribed to the π-π* transition of the aromatic rings. The strongest emission peak for compounds 1~2 are observed at about 424 nm. By comparison of the emission energies and profiles of the free H3TBTC ligand and compounds 1~2, the luminescent behavior of compounds 1~2 are considered to be originated from H3TBTC ligands. The blue-shifted bands of their emissions may result from the fact that the ligands are not allowed to relax along the torsional mode upon photoexcitation.

[1] (a) Farrusseng, D.; Aguado, S.; Pinel, C. Angew. Chem., Int. Ed. 2009, 48, 7502; (b) Lin, J.-B.; Xue, W.; Zhang, J.-P.; Chen, X.-M. Chem. Commun. 2011, 47, 926; (c) Cui, Y.; Yue, Y.; Qian, G.; Chen, B. Chem. Rev. 2012, 112, 1126; (d) Zhong, D.; Lu, T. Sci. China Chem. 2011, 54, 1395.
[2] Mal, P.; Breiner, B.; Rissanen, K.; Nitschke, J. R. Science 2009, 324, 1697.
[3] Perry, J. J.; Perman, J. A.; Zaworotko, M. J. Chem. Soc. Rev. 2009, 38, 1400.
[4] (a) Seidel, S. R.; Stang, P. J. Acc. Chem. Res. 2002, 35, 972; (b) Tranchemontagne, D. J.; Ni, Z.; O'Keeffe, M.; Yaghi, O. M. Angew. Chem., Int. Ed. 2008, 47, 5136.
[5] (a) Wang, X.-S.; Ma, S.; Forster, P. M.; Yuan, D.; Eckert, J.; López, J. J.; Murphy, B. J.; Parise, J. B.; Zhou, H.-C. Angew. Chem., Int. Ed. 2008, 47, 7263; (b) Zhao, D.; Yuan, D.; Sun, D.; Zhou, H.-C. J. Am. Chem. Soc. 2009, 131, 9186; (c) Farha, O. K.; Özgür Yazayd?n, A.; Eryazici, I.; Malliakas, C. D.; Hauser, B. G.; Kanatzidis, M. G.; Nguyen, S. T.; Snurr, R. Q.; Hupp, J. T. Nat. Chem. 2010, 2, 944; (d) Yan, Y.; Telepeni, I.; Yang, S.; Lin, X.; Kockelmann, W.; Dailly, A.; Blake, A. J.; Lewis, W.; Walker, G. S.; Allan, D. R.; Barnett, S. A.; Champness, N. R.; Schroder, M. J. Am. Chem. Soc. 2010, 132, 4092; (e) Yuan, D.; Zhao, D.; Sun, D.; Zhou, H.-C. Angew. Chem., Int. Ed. 2010, 49, 5357.
[6] (a) Perry, J. J.; Kravtsov, V. C.; McManus, G. J.; Zaworotko, M. J. J. Am. Chem. Soc. 2007, 129, 10076; (b) Zou, Y.; Park, M.; Hong, S.; Lah, M. S. Chem. Commun. 2008, 2340; (c) Li, C.; Qiu, W.; Shi, W.; Song, H.; Bai, G.; He, H.; Li, J.; Zaworotko, M. J. CrystEngComm 2012, 14, 1929.
[7] (a) Liu, T.-F.; Lü, J.; Guo, Z.; Proserpio, D. M.; Cao, R. Cryst. Growth Des. 2010, 10, 1489; (b) Lin, Z.-J.; Liu, T.-F.; Xu, B.; Han, L.-W.; Huang, Y.-B.; Cao, R. CrystEngComm 2011, 13, 3321; (c) Lin, Z.-J.; Liu, T.-F.; Zhao, X.-L.; Lü, J.; Cao, R. Cryst. Growth Des. 2011, 11, 4284; (d) Liu, T.-F.; Lü, J.; Tian, C.; Cao, M.-N; Lin, Z.; Cao, R. Inorg. Chem. 2011, 50, 2264; (e) Lin, Z.-J.; Liu, T.-F.; Huang, Y.-B.; Lü, J.; Cao, R. Chem.-Eur. J. 2012, 18, 7896.
[8] Guo, Z.; Cao, R.; Wang, X.; Li, H.; Yuan, W.; Wang, G.; Wu, H.; Li, J. J. Am. Chem. Soc. 2009, 131, 6894.
[9] Liu, T.-F.; Lu, J.; Lin, X.; Cao, R. Chem. Commun. 2010, 46, 8439.
[10] (a) Qin, L.; Hu, J.-S.; Li, Y.-Z.; Zheng, H.-G. Cryst. Growth Des. 2011, 11, 3115; (b) Fang, Q. R.; Shi, X.; Wu, G.; Tian, G.; Zhu, G. S.; Li, Y. F.; Wang, L. F.; Wang, C. L.; Chen, Y.; Zhang, Z. D.; Guo, Z.; Shang, T. C.; Cai, X. H.; Qiu, S. L. Acta Chim. Sinica 2002, 60, 2087. (方千荣, 石鑫, 吴刚, 田歌, 朱广山, 李亚峰, 王立锋, 王春雷, 陈悦, 张震东, 郭卓, 商铁存, 蔡晓慧, 裘式纶, 化学学报, 2002, 60, 2087.)
[11] Liu, T.; Lu, J.; Shi, L.; Guo, Z.; Cao, R. CrystEngComm 2009, 11, 583.
[12] (a) Chen, Y. F.; Lee, J. Y.; Babarao, R.; Li, J. W. J. Phys. Chem. C 2010, 114, 6602; (b) Lin, Z.-J.; Yang, Z.; Liu, T.-F.; Huang, Y.-B.; Cao, R. Inorg. Chem. 2012, 51, 1813.
[13] (a) Lan, Y.-Q.; Jiang, H.-L.; Li, S.-L.; Xu, Q. Adv. Mater. 2011, 23, 5015; (b) Qin, J.-S.; Du, D.-Y.; Li, W.-L.; Zhang, J.-P.; Li, S.-L.; Su, Z.-M.; Wang, X.-L.; Xu, Q.; Shao, K.-Z.; Lan, Y.-Q. Chem. Sci. 2012, 3, 2114.
[14] (a) Wang, J.-J.; Cao, P.-X.; Gao, L.-J.; Fu, F.; Zhang, M.-L.; Ren, Y.-X.; Hou, X.-Y. Chin. J. Struct. Chem. 2011, 30, 1787; (b) Li, Y. J.; Song, H. H.; Wang, J. Y.; Han, Z. G. Acta Chim. Sinica 2007, 65, 2401. (李亚娟, 宋会花, 王继业, 韩占刚, 化学学报, 2007, 65, 2401.)
[15] (a) Allendorf, M. D.; Bauer, C. A.; Bhakta, R. K.; Houk, R. J. T. Chem. Soc. Rev. 2009, 38, 1330; (b) Kang, W.; Deng, Z. P.; Gao, S. Chinese J. Inorg. Chem. 2011, 27, 131. (康伟, 邓兆鹏, 高山, 无机化学学报, 2011, 27, 131.)
/
| 〈 |
|
〉 |