

金属/金属氧化物纳米粒子在不对称氢化和氢转移反应中的应用研究进展
收稿日期: 2014-04-25
网络出版日期: 2014-05-07
基金资助
项目受国家自然科学基金(Nos.21372118,21232008)、国家重点基础研究发展计划(No.2010CB833300)、江苏省“333高层次人才培养工程”、江苏省“青蓝工程”和南京农业大学中央高校基本科研业务费-引进人才专项资金(No.KYRC201211)资助.
Recent Progress of Metal/Metal Oxide Nanoparticles for Asymmetric Hydrogenation and Transfer Hydrogenation
Received date: 2014-04-25
Online published: 2014-05-07
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21372118, 21232008), the National Basic Research Program of China(No. 2010CB833300), “333 High Level Talent Project”, “QingLan Project” of JiangSu Province and the Fundamental Research Funds for the Central Universities (NJAU) (No. KYRC201211).
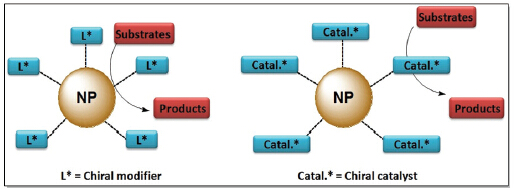
近年来,金属/金属氧化物纳米粒子催化的不对称氢化和氢转移反应已经成为催化领域的前沿和研究热点之一. 金属/金属氧化物纳米粒子的催化模式类似于“纳米反应器”,底物可以通过有机包覆层扩散至催化中心,局部的高催化剂浓度通常可以极大地提高催化反应转换数(TON)和转化频率(TOF). 在以纳米金属为催化活性中心方面,Orito纳米铂体系获得最多的关注,科学家们从手性修饰剂的结构改造、催化剂载体的选择、不同的反应介质、纳米催化剂的形貌和催化反应机理等方面开展了较为系统的研究,并取得重要进展. 此外,纳米钯、铑、钌、铱和铁等金属纳米催化剂也在烯烃、酮和亚胺等化合物的不对称氢化和氢转移反应中表现出良好的催化性能,特别是纳米铱和铁催化剂已获得95%以上的对映选择性. 在金属/金氧化物纳米粒子为催化剂载体方面,其催化不对称氢化及氢转移反应的效率及对映选择性可与均相催化剂相媲美,同时还解决了均相催化剂难于回收再循环的缺陷. 本文简要介绍了近年来手性金属纳米催化剂在不对称氢化和氢转移反应领域的研究进展,讨论了相关反应的催化机理,并对该领域仍存在的问题和未来的发展方向进行了展望.
季益刚 , 吴磊 , 范青华 . 金属/金属氧化物纳米粒子在不对称氢化和氢转移反应中的应用研究进展[J]. 化学学报, 2014 , 72(7) : 798 -808 . DOI: 10.6023/A14040325
Metal/metal oxide nanoparticles for asymmetric hydrogenation and transfer hydrogenation have emerged as a frontier and evolved into a hot topic of asymmetric catalysis in recent years. Their catalytic modes resemble that of “nano-reactor”, where substrates diffuse through organic shells into catalytic active sites. Thus, high local catalyst concentration usually dramatically improves TON and TOF. In the case of nanoparticles as active sites, Orito’s platinum catalytic system received most extensive interests. Achievements have been made in chiral modifier structural modification, catalyst supports, reaction medium, nanoparticle morphology and catalytic mechanism. Moreover, other metal nanoparticles including palladium, rhodium, ruthenium, iridium and iron exhibited favorable catalytic efficiency in the asymmetric hydrogenation and transfer hydrogenation of alkenes, ketones and imines, especially for iridium and iron nanoparticles, ee values over 95% were obtained. In another case of metal/metal oxide nanoparticles as catalyst supports, comparable efficiency and enantioselectivity were observed to homogeneous catalysts, meanwhile, this protocol overcame the drawbacks of homogeneous catalysts with easier recovery and reuse. This review presents a brief overview on the recent progress in the asymmetric hydrogenation and transfer hydrogenation catalyzed by metal/metal oxide nanoparticles, as well as the related catalytic mechanism. However, there are still many challenges in this promising research field of metal/metal oxide nanoparticles for asymmetric catalysis. In addition to the continuous understanding of the catalytic mechanism, it is highly desirable to develop new types of metal/metal oxide nanoparticles with high efficiency, high enantioselectivity, and convenient recyclability.

[1] (a) Noyori, R. Asymmetric Catalysis in Organic Synthesis, Wiley, New York, 1994.
(b) Xie, J. H.; Zhou, Q. L. Acta Chim. Sinica 2012, 70, 1427. (谢建华, 周其林, 化学学报, 2012, 70, 1427.)
(c) Wang, D.; Hou, C. J.; Chen, L.-F.; Liu, X. N.; An, Q. D.; Hu, X. P. Chin. J. Org. Chem. 2013, 33, 1355. (王东, 候传金, 陈丽凤, 刘小宁, 安庆大, 胡向平, 有机化学, 2013, 33, 1355.)
(d) Cui, P. L.; Liu, H. M.; Guo, X. M.; Zhang, D. N.; Wang, Y. E.; Wang, C. Chin. J. Org. Chem. 2013, 33, 436. (崔朋雷, 刘卉敏, 果秀敏, 张冬暖, 王彦恩, 王春, 有机化学, 2013, 33, 436.)
(e) Li, X. N.; Zhang, P. L.; Duan, K.; Wang, J. X. Chin. J. Org. Chem. 2012, 32, 19. (李小娜, 张鹏亮, 段凯, 王家喜, 有机化学, 2012, 32, 19.)
(f) Liu, Y.; Wang, Z.; Ding, K. L. Acta Chim. Sinica 2012, 70, 1464. (刘龑, 王正, 丁奎岭, 化学学报, 2012, 70, 1464.)
[2] Knowles, W. S. Acc. Chem. Res. 1983, 16, 106.
[3] (a) Fan, Q. H.; Li, Y. M.; Chan, A. S. C. Chem. Rev. 2002, 102, 3385.
(b) Handbook of Asymmetric Heterogeneous Catalysis,Eds.: Ding, K.; Uozumi, Y., Wiley-VCH, 2008.
[4] (a) Wang, Z.; Chen, G.; Ding, K. L. Chem. Rev. 2009, 109, 322.
(b) Fan, Q. H.; Ding, K. L. Top. Organomet. Chem. 2011, 36, 207.
(c) Ma, B. D.; Deng, G. J.; Liu, J. ; He, Y. M.; Fan, Q. H. Acta Chim. Sinica 2013, 71, 528. (马保德, 邓国军, 刘继, 何艳梅, 范青华, 化学学报, 2013, 71, 528.)
(d) Liu, H. Q.; Shi, L.; Chen, Q. A.; Wang, L.; Zhou, Y. G. Acta Chim. Sinica 2013, 71, 40. (刘洪强, 时磊, 陈庆安, 王磊, 周永贵, 化学学报, 2013, 71, 40.)
[5] (a) Mallat, T.; Orglmeister, E.; Baiker, A. Chem. Rev. 2007, 107, 4863.
(b) Yasukawa, T.; Miyamura, H.; Kobayashi, S. Chem. Soc. Rev. 2014, 43, 1450.
[6] (a) Astruc, D.; Lu, F.; Aranzaes, J. R. Angew. Chem. Int. Ed. 2005, 44, 7852.
(b) Schätz, A.; Reiser, O.; Stark, W. J. Chem. Eur. J. 2010, 16, 8950.
[7] (a) Barbaro, P.; Santo, V. D.; Liguori, F. Dalton Trans. 2010, 39, 8391.
(b) Ranganath, K. V. S.; Glorius, F. Catal. Sci. Technol. 2011, 1, 13.
[8] Orito, Y.; Imai, S.; Niwa, S. Bull. Chem. Soc. Jpn. 1979, 1118.
[9] Harada, T.; Izumi, Y. Chem. Lett. 1978, 1195.
[10] Studer, M.; Blaser, H. U.; Exner, C. Adv. Synth. Catal. 2003, 345, 45.
[11] (a) Burgi, T.; Baiker, A. Acc. Chem. Res. 2004, 37, 909.
(b) Chen, C. H.; Zhan, E. S.; Li, Y.; Shen, W. J. J. Mol. Catal. A-Chem. 2013, 379, 117.
[12] (a) Griffiths, S. P.; Johnson, P.; Wells, P. B. Appl. Catal. A 2000, 191, 193.
(b) Chen, Z. J.; Li, X. H.; Li, C. Chin. J. Catal. 2011, 32, 155. (陈志坚, 李晓红, 李灿, 催化学报, 2011, 32, 155.)
[13] Li, X.; Shen, Y.; Xing, R.; Liu, Y. M.; Wu, H. H.; He, M. Y.; Wu, P. Catal. Lett. 2008, 122, 325.
[14] Xing, L.; Du, F.; Liang, J. J.; Chen, Y. S.; Zhou, Q. L. J. Mol. Catal. A-Chem. 2007, 276, 191.
[15] Li, B.; Li, X. H.; Wang, H. N.; Wu, P. J. Mol. Catal. A-Chem. 2011, 345, 81.
[16] Schmidt, E.; Vargas, A.; Mallat, T.; Baiker, A. J. Am. Chem. Soc. 2009, 131, 12358.
[17] Nandanan, E.; Gu, H. W.; Shao, H. L.; Jiang, J.; Ying, J. Y. Green Chem. 2011, 13, 3070.
[18] Beier, M. J.; Andanson, J.-M.; Mallat, T.; Krumeich, F.; Baiker, A. ACS Catal. 2012, 2, 337.
[19] Chen, Z. J.; Guan, Z. H.; Li, M. R.; Yang, Q. H.; Li, C. Angew. Chem. Int. Ed. 2011, 50, 4913.
[20] Guan, Z.; Lu, S.; Chen, Z.; Li, C.J. Catal. 2013, 305, 19.
[21] (a) Lavoie, S.; Mahieu, G.; McBreen, P. H. Angew. Chem. Int. Ed. 2006, 45, 7404.
(b) Lavoie, S.; Laliberté, M.-A.; Temprano, I.; McBreen, P. H. J. Am. Chem. Soc. 2006, 128, 7588.
[22] Schmidt, E.; Bucher, C.; Santarossa, G.; Mallat, T.; Gilmour, R.; Baiker, A. J. Catal. 2012, 289, 238.
[23] Meemken, F.; Maeda, N.; Hungerbühler, K.; Baiker, A. Angew. Chem. Int. Ed. 2012, 51, 8212.
[24] Perez, J. R. G.; Malthete, J.; Jacques, J. CR. Acad. Sc. Paris Serie Ⅱ . 1985, 300, 169.
[25] (a) Chen, C. H.; Zhan, E. S.; Li, Y.; Shen, W. J. Acta Chim. Sinica 2013, 71, 1505. (陈春辉, 展恩胜, 李勇, 申文杰, 化学学报, 2013, 71, 1505.)
(b) Chen, C. H.; Zhan, E. S.; Ta, N.; Li, Y.; Shen, W. J. Catal. Sci. Technol. 2013, 3, 2620.
[26] Huck, W.-R.; Mallat, T.; Baiker, A. New J. Chem. 2002, 26, 6.
[27] Nitta, Y.; Watanabe, J.; Okuyama, T.; Sugimura, T. J. Catal. 2005, 236, 164.
[28] Makra, Z.; Szöllösi, G.; Bartók, M. Catal. Today 2012, 181, 56.
[29] Maris, M.; Huck, W.-R.; Mallat, T.; Baiker, A. J. Catal. 2003, 219, 52.
[30] Watson, D. J.; Jesudason, R. J.; Beaumont, S. K.; Kyriakou, G.; Burton, J. W.; Lambert, R. M. J. Am. Chem. Soc. 2009, 131, 14584.
[31] Ma, H.; Chen, H.; Zhang, Q.; Li, X. J. Mol. Catal. A-Chem. 2003, 196, 131.
[32] Sonderegger, O. J.; Ho, G. M.-W.; Bürgi, T.; Baiker, A. J. Catal. 2005, 230, 499.
[33] Wang, J.; Feng, J.; Qin, R.; Fu, H.; Yuan, M.; Chen, H.; Li, X. Tetrahedron: Asymmetry 2007, 18, 1643.
[34] Jiang, H. Y.; Yang, C. F.; Li, C.; Fu, H. Y.; Chen, H.; Li, R. X.; Li, X. J. Angew. Chem. Int. Ed. 2008, 47, 1.
[35] Jiang, H. Y.; Sun, B.; Zheng, X. X.; Chen, H. Appl. Catal. A- Gen. 2012, 421, 86.
[36] (a) Johnson, N. B.; Lennon, I. C.; Moran, P. H.; Ramsden, J. A. Acc. Chem. Res. 2007, 40, 1291.
(b) Sun, Q.; Meng, X. J.; Liu, X.; Zhang, X. M.; Yang, Y.; Yang, Q. H.; Xiao, F. S. Chem. Commun. 2012, 48, 10505.
[37] Groves, J. T.; Myers, R. S. J. Am. Chem. Soc. 1983, 105, 5791.
[38] Gopalaiah, K. Chem. Rev. 2013, 113, 3248.
[39] (a) Mikhailine, A.; Lough, A. J.; Morris, R. H. J. Am. Chem. Soc. 2009, 131, 13943.
(b) Sonnenberg, J. F.; Coombs, N.; Dube, P. A.; Morris, R. H. J. Am. Chem. Soc. 2012, 134, 5893.
[40] Li, Y. Y.; Yu, S.; Wu, X.; Xiao, J.; Shen, W. Y.; Dong, Z. R.; Gao, J. X. J. Am. Chem. Soc. 2014, 136, 4031.
[41] Wu, L. Ph.D. Dissertation, Institute of Chemistry, Chinese Academy of Sciences (CAS), Beijing, 2007. (吴磊, 博士论文, 中国科学院化学研究所, 北京, 2007.)
[42] Li, H.; Luk, Y. Y.; Mrksich, M. Langmuir 1999, 15, 4957.
[43] Daniel, M. C.; Astruc, D. Chem. Rev. 2004, 104, 293.
[44] Belser, T.; Stöhr, M.; Pfaltz, A. J. Am. Chem. Soc. 2005, 127, 8720.
[45] Polshettiwar, V.; Luque, R.; Fihri, A.; Zhu, H.; Bouhrara, M.; Basset, J. M. Chem. Rev. 2011, 111, 3036.
[46] Hu, A. G.; Yee, G. T.; Lin, W. B. J. Am. Chem. Soc. 2005, 127, 12486.
[47] Hu, A. G.; Liu, S.; Lin, W. B. RSC Adv. 2012, 2, 2576.
[48] Gao, X.; Liu, R.; Zhang, D.; Wu, M.; Cheng, T.; Liu, G. Chem. Eur. J. 2014, 20, 1515.
[49] Fraile, J. M.; García, J. I.; Mayoral, J. A. Chem. Rev. 2009, 109, 360.
[50] Wu, L.; He, Y. M.; Fan, Q. H. Adv. Synth. Catal. 2011, 353, 2915.
[51] Gleeson, O.; Davies, G. L.; Peschiulli, A.; Tekoriute, R.; Gun'ko, Y. K.; Connon, S. J. Org. Biomol. Chem. 2011, 9, 7929.
/
| 〈 |
|
〉 |