

Ir(III)螯合物催化醚的硅氢化反应中负氢来源的机理研究
收稿日期: 2017-09-03
网络出版日期: 2018-01-09
基金资助
项目受国家自然科学基金(Nos.21572212,21672001,21702041),国家自然科学基金委员会与中国工程物理研究院联合基金(No.U1530262)和深圳超级计算中心和中国科学技术大学计算中心给予计算资源资助.
Hydride Source in Ethers Hydrosilylation Reaction Catalyzed by Brookhart's Ir(Ⅲ) Pincer Complex
Received date: 2017-09-03
Online published: 2018-01-09
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21572212, 21672001, 21702041), Joint Foundation of National Natural Science Foundation of China and Academy of Engineering Physics (No. U1530262), and the supercomputing system of National Supercomputing Center in Shenzhen and USTC.
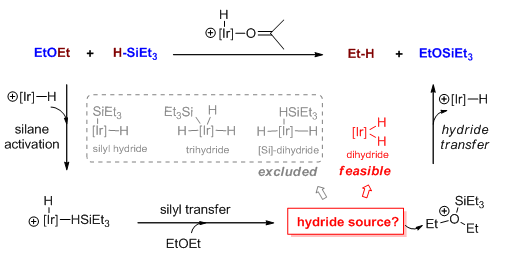
采用DFT方法对Ir(Ⅲ)螯合物催化乙醚硅氢化生成乙烷和乙基硅醚的反应展开理论研究.反应中[H-Ir-H],[H-Ir-Si],[Ir(HSiEt3)]和[Et3Si-Ir-(H)3]化合物均为可能的负氢来源.理论研究表明[H-Ir-H]化合物是最优势的负氢来源.通过扭曲-相互作用能分析,发现其他三种可能的负氢来源不优势的原因在于HSiEt3或SiEt3基团对铱中心的络合.更为重要的,我们发现[H-Ir-H]化合物中适中的Ir-H键解离能,小位阻以及SiEt3对醚的络合而产生的促进作用共同使得[H-Ir-H]化合物上的负氢转移相对优势.
张琪 , 刘奥 , 于海珠 , 傅尧 . Ir(III)螯合物催化醚的硅氢化反应中负氢来源的机理研究[J]. 化学学报, 2018 , 76(2) : 113 -120 . DOI: 10.6023/A17070328
The hydrosilylative reduction with silane is a popular defunctionalization strategy to convert biomass into chemicals and energies because of the mild reaction conditions. Among these, the reduction of C-O bond is particularly important because of its application in sugar biomass reduction. The (C6F5)3 B/silane catalytic system has been frequently used in the reduction of C-O bonds in the past years. However, Brookhart et al. reported alkyl ethers reduction by using Ir(Ⅲ) pincer catalyst and reductant HSiEt3. This work provides a novel hydrosilylation catalyst for C-O reduction and an effective method for sugar biomass deoxygenation. According to the previous mechanistic proposals on similar Ir catalysed hydrosilylation reactions, the iridium dihydride complex, iridium silyl hydride complex, silane adduct iridium complex and iridium silyl trihydride complex might possibly act as the hydride source. We carried out the theoretical study on Brookhart's Ir(Ⅲ) Pincer Complex/HSiEt3 catalyzed hydrosilylation reaction of EtOEt yielding ethane and EtOSiEt3. The density functional theory (DFT) calculations in our study indicate that the iridium dihydride complex is the best hydride source. Our calculation result is consistent well with experimental observations in Brookhart's experiment. For example, the phenomenon that adding iridium dihydride complex into the reaction system increases the reaction rate is understandable because the complex is involved in the rate-determining step. From the Distortion/Interaction analysis, we found that hydride transfer steps on the other three possible hydride sources are disfavoured by the HSiEt3/-SiEt3 group (derived from HSiEt3) bonded with Ir center. The iridium silyl hydride complex is unfavourable because the Ir-H bond is strengthened and the pincer ligand is distorted. For the silane adduct iridium complex, the coordination of HSiEt3 destabilizes iridium complex intermediate for entropy increases and trans effect, and destabilizes the related transition state by damaging its pincer ligand. Further, the corresponding hydride transfer transition state from iridium silyl trihydride is highly unstable and Si-H bond always reform automatically. What's more important, the moderate bond dissociation energy of Ir-hydride, small steric hindrance and the promotion effect of SiEt3 group coordination with ether all facilitate the hydride transfer on the iridium dihydride complex.

Key words: hydrosilylation; iridium; hydride; pincer; DFT
[1] (a) Kerr, R. A.; Service, R. F. Science 2005, 309, 101.
(b) Ra-gauskas, A. J.; Williams, C. K.; Davison, B. H.; Britovsek, G.; Cairney, J.; Eckert, C. A.; Fredrick, W. J. J.; Hallett, J. P.; Leak, D. J.; Liotta, C. L.; Mielenz, J. R.; Murphy, R.; Templer, R.; Tschaplinski, T. Science 2006, 311, 484.
[2] (a) Douvris, C.; Ozerov, O. V. Science 2008, 321, 1188.
(b) Dioumaev, V. K.; Bullock, R. M. Nature 2003, 424, 530.
(c) Fernandez-Alvarez, F. J.; Aitani A. M.; Oro, L. A. Catal. Sci. Technol. 2014, 4, 611.
(d) Sui, Y.-Z.; Zhang, X.-C.; Wu, J.-W.; Li, S.; Zhou, J.-N.; Li, M.; Fang, W.; Chan A. S. C.; Wu, J. Chem.-Eur. J. 2012, 18, 7486.
(e) Addis, D.; Das, S.; Junge K.; Beller, M. Angew. Chem., Int. Ed. 2011, 50, 6004.
(f) Malacea, R.; Poli, R.; Manoury, E. Coord. Chem. Rev. 2010, 254, 729.
(g) Pan, Z.; Liu, M.; Zheng, C.; Gao, D.; Huang, W. Chin. J. Chem. 2017, 35, 1227.
(h) Hu, X.; Hu, F.; Zhang, M.; Liao, Y.; Xu, X.; Yuan, W.; Zhang, X. Chin. J. Org. Chem. 2016, 36, 1895. (扈晓艳, 胡方芝, 张敏敏, 廖益均, 徐小英, 袁伟成, 张晓梅, 有机化学, 2016, 36, 1895.)
(i) Zaranek, M.; Marciniec, B.; Pawluc, P. Org. Chem. Front. 2016, 3, 1337.
(j) Hu, X.; Tian, C.; Maxim, B.; Nie, W. Acta Chim. Sinica 2015, 73, 1025. (胡茜, 田冲, Borzov Maxim, 聂万丽, 化学学报, 2015, 73, 1025.)
[3] (a) Yang, J.; Brookhart, M. Adv. Synth. Catal. 2009, 351, 175.
(b) Caputo, C. B.; Stephan, D. W. Organometallics 2012, 31, 27.
(c) Scott, V. J.; Celenligil-Cetin, R.; Ozerov, O. V. J. Am. Chem. Soc. 2005, 127, 2852.
[4] (a) Gevorgyan, V.; Liu, J.-X.; Rubin, M.; Benson S.; Yamamoto, Y. Tetrahedron Lett. 1999, 40, 8919.
(b) Gevorgyan, V.; Rubin, M.; Benson, S.; Liu, J.-X.; Yamamoto, Y. J. Org. Chem. 2000, 65, 6179.
(c) Blackwell, J. M.; Morrison, D. J.; Piers, W. E. Tetrahedron 2002, 58, 8247.
(d) Nimmagadda, R. D.; McRae, C. Tetrahedron Lett. 2006, 47, 5755.
(e) Mack, D. J.; Guo, B.; Njardarson, J. T. Chem. Commun. 2012, 48, 7844.
(f) Chojnowski, J.; Rubinsztajn, S.; Cella, J. A.; Fortuniak, W.; Cypryk, M.; Kurjata, J.; Kazmierski, K. Organometallics 2005, 24, 6077.
[5] (a) Park, S.; Brookhart, M. Organometallics 2010, 29, 6057.
(b) Parks, D. J.; Piers, W. E. J. Am. Chem. Soc. 1996, 118, 9440.
(c) Skjel, M. K.; Houghton, A. Y.; Kirby, A. E.; Harrison, D. J.; McDonald, R.; Rosenberg, L. Org. Lett. 2010, 12, 376.
(d) Chandrasekhar, S.; Reddy, C. R.; Babu, B. N. J. Org. Chem. 2002, 67, 9080.
[6] (a) Bézier, D.; Park, S.; Brookhart, M. Org. Lett. 2013, 15, 496.
(b) Gevorgyan, V.; Rubin, M.; Liu, J.-X.; Yamamoto, Y. J. Org. Chem. 2001, 66, 1672.
(c) Cheng, C.; Brookhart, M. Angew. Chem., Int. Ed. 2012, 51, 9422.
[7] (a) Park, S.; Bézier, D.; Brookhart, M. J. Am. Chem. Soc. 2012, 134, 11404.
(b) Berkefeld, A.; Piers, W. E.; Parvez, M. J. Am. Chem. Soc. 2010, 132, 10660.
[8] (a) Blackwell, J. M.; Sonmor, E. R.; Scoccitti, T.; Piers, W. E. Org. Lett. 2000, 2, 3921.
(b) Rubin, M.; Schwier, T.; Gevorgyan, V. J. Org. Chem. 2002, 67, 1936.
(c) Ding, S.; Song, L.-J.; Chung, L. W.; Zhang, X.; Sun, J.; Wu, Y.-D. J. Am. Chem. Soc. 2013, 135, 13835.
[9] (a) McLaughlin, M. P.; Adduci, L. L.; Becker, J. J.; Gagné, M. R. J. Am. Chem. Soc. 2013, 135, 1225.
(b) Robert, T.; Oestreich, M. Angew. Chem., Int. Ed. 2013, 52, 5216.
(c) Adduci, L. L.; McLaughlin, M. P.; Bender, T. A.; Becker, J. J.; Gagné, M. R. Angew. Chem., Int. Ed. 2014, 53, 1646.
[10] (a) Yang, J.; White, P. S.; Brookhart, M. J. Am. Chem. Soc. 2008, 130, 17509.
(b) Yang, J.; Brookhart, M. J. Am. Chem. Soc. 2007, 129, 12656.
(c) Park, S.; Brookhart, M. Chem. Commun. 2011, 47, 3643.
[11] Metsänen, T. T.; Hrobarik, P.; Klare, H. F. T.; Kaupp, M.; Oestreich, M. J. Am. Chem. Soc. 2014, 136, 6912.
[12] Herein we examine the first cleavage of EtOEt. The cleavage of the silyl ether could also be achieved (the second cleavage) if lengthen the reaction time.
[13] Yang, J.; White, P. S.; Schauer, C. K.; Brookhart, M. Angew. Chem., Int. Ed. 2008, 47, 4141.
[14] Perutz, R. N.; Sabo-Etienne, S. Angew. Chem., Int. Ed. 2007, 46, 2578.
[15] (a) Lin, Z. Chem. Soc. Rev. 2002, 31, 239.
(b) Chung, L. W.; Lee, H. G.; Lin, Z.; Wu, Y.-D. J. Org. Chem. 2006, 71, 6000.
(c) Lee, T. Y.; Dang, L.; Zhou, Z.; Yeung, C. H.; Lin, Z.; Lau, C. P. Eur. J. Inorg. Chem. 2010, 5675.
[16] Rendler, S.; Oestreich, M. Angew. Chem., Int. Ed. 2008, 47, 5997.
[17] (a) Wang, W.; Gu, P.; Wang, Y.; Wei, H. Organometallics 2014, 33, 847.
(b) Sakata, K.; Fujimoto, H. J. Org. Chem. 2013, 78, 12505.
[18] Cheng, Y.-H.; Zhao, X.; Song, K.-S.; Liu, L.; Guo, Q.-X. J. Org. Chem. 2002, 67, 6638.
[19] Anderson, K. M.; Orpen, A. G. Chem. Commun. 2001, 24, 2682.
[20] The DFT method leads to an overestimation of entropy, resulting in an overestimation of the free energy. This phenomenon has also appeared in previous theoretical calculations. For example:(a) Ding, L.; Ishida, N.; Murakami, M.; Morokuma, K. J. Am. Chem. Soc. 2014, 136, 169.
(b) Sugiyama, A.; Ohnishi, Y.-Y.; Nakaoka, M.; Nakao, Y.; Sato, H.; Sakaki, S.; Nakao, Y.; Hiyama, T. J. Am. Chem. Soc. 2008, 130, 12975.
(c) Yu, Z.-X.; Houk, K. N. J. Am. Chem. Soc. 2003, 125, 13825.
(d) Hermans, J.; Wang, L. J. Am. Chem. Soc. 1997, 119, 2707.
(e) Tanaka, R.; Yamashita, M.; Chung, L. W.; Morokuma, K.; Nozaki, K. Organometallics 2011, 30, 6742.
(f) Dub, P. A.; Ikariya, T. J. Am. Chem. Soc. 2013, 135, 2604.
(g) Strajbl, M.; Sham, Y. Y.; Villà, J.; Chu, Z.-T.; Warshel, A. J. Phys. Chem. B 2000, 104, 4578.
(h) Hermans, J.; Wang, L. J. Am. Chem. Soc. 1997, 119, 2707.
[21] Gorelsky, S. I.; Lapointe, D.; Fagnou, K. J. Am. Chem. Soc. 2008, 130, 10848.
[22] Zhang, Q.; Yu, H.-Z.; Shi, J. Acta Phys.-Chim. Sin. 2013, 29, 2321. (张琪, 于海珠, 石景, 物理化学学报, 2013, 29, 2321.)
[23] Zhang, Q.; Yu, H.-Z.; Fu, Y. Organometallics 2016, 35, 2473.
[24] Hydride is dissociated in the calculation of Ir-H(2) bond dissociation energy. See reference herein:Qi, X.-J.; Liu, L.; Fu, Y.; Guo, Q.-X. Organometallics 2006, 25, 5879.
[25] Gaussian 09, revision D.01; Gaussian, Inc., Wallingford, CT, 2013.
[26] Zhao, Y.; Truhlar, D. G. Theor. Chem. Acc. 2008, 120, 215.
[27] (a) Kundu, S.; Choi, J.; Wang, D. Y.; Choliy, Y.; Emge, T. J.; Krogh-Jespersen, K.; Goldman, A. S. J. Am. Chem. Soc. 2013, 135, 5127.
(b) Haibach, M. C.; Guan, C.; Wang, D. Y.; Li, B.; Lease, N.; Steffens, A. M.; Krogh-Jespersen, K.; Goldman, A. S. J. Am. Chem. Soc. 2013, 135, 15062.
(c) Tian, Y.; Fu, Y.; Zhang, Q.; Yu, H.-Z.; Shi, J. Acta Chim. Sinica 2014, 72, 935. (田燕, 傅尧, 张琪, 于海珠, 石景, 化学学报, 2014, 72, 935.)
(d) Liu, D.-J.; Yu, H.-Z.; Fu, Y. Acta Chim. Sinica 2013, 71, 1385. (刘丁嘉, 于海珠, 傅尧, 化学学报, 2013, 71, 1385.)
[28] Zhang, Q.; Yu, H.-Z.; Fu, Y. Org. Chem. Front. 2014, 1, 614.
[29] Fukui, K. J. Phys. Chem. 1970, 74, 4161.
[30] Fukui, K. Acc. Chem. Res. 1981, 14, 363.
[31] Marenich, A. V.; Cramer, C. J.; Truhlar, D. G. J. Phys. Chem. B 2009, 113, 6378.
[32] Ehlers, A. W.; Bohme, M.; Dapprich, S.; Gobbi, A.; Hollwarth, A.; Jonas, V.; Kohler, K. F.; Stegmann, R.; Veldkamp, A.; Frenking, G. Chem. Phys. Lett. 1993, 208, 111.
[33] Hollwarth, A.; Bohme, M.; Dapprich, S.; Ehlers, A. W.; Gobbi, A.; Jonas, V.; Kohler, K. F.; Stegmann, R.; Veldkamp, A.; Frenking, G. Chem. Phys. Lett. 1993, 208, 237.
/
| 〈 |
|
〉 |