

一种热活化延迟荧光黄光材料的合成、性能及高效率电致发光器件
收稿日期: 2018-10-18
网络出版日期: 2018-12-20
基金资助
项目受国家自然科学基金(No.51773029)和河南省自然科学基金(No.182300410230)资助.
A Novel Yellow Thermally Activated Delayed Fluorescence Emitter For Highly Efficient Organic Light-Emitting Diodes
Received date: 2018-10-18
Online published: 2018-12-20
Supported by
Project supported by the National Natural Science Foundation of China (No. 51773029) and Henan Natural Science Foundation (No. 182300410230).
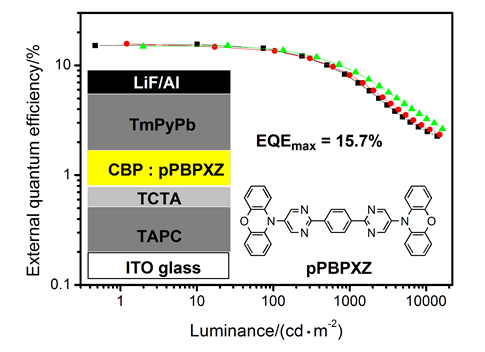
利用吩噁嗪和嘧啶分别作为电子给体和电子受体,通过Buchwald-Hartwig和Suzuki偶联反应成功合成了一种热活化延迟荧光黄光材料pPBPXZ.密度泛函理论计算显示,pPBPXZ分子中吩噁嗪和嘧啶结构单元间的二面角接近90°,而两个嘧啶结构单元与连接二者的苯环间的二面角接近0°;pPBPXZ的最高电子占据轨道主要分布在吩噁嗪结构单元上,最低电子未占轨道主要分布在嘧啶环和苯环上,两种分子轨道只有很小部分重叠.循环伏安、热重和差热测试表明,pPBPXZ具有高的电化学稳定性和热稳定性.在甲苯溶液中,pPBPXZ在360~495 nm显示出了明显的分子内电荷转移跃迁吸收,室温发光峰出现在535 nm.根据低温(77 K)荧光和磷光光谱,计算得到pPBPXZ的最低激发单重态和最低激发三重态能级分别为2.57 eV和2.48 eV,能级差(△EST)仅为0.09 eV.利用pPBPXZ作为发光层客体掺杂材料,制备出了高效率的黄光电致发光器件.器件的发射峰出现在552~560 nm,最大电流效率、功率效率和外量子效率分别达到了49.9 cd/A、49.0 lm/W和15.7%,而且发光效率受pPBPXZ掺杂浓度影响较小.
王志强 , 蔡佳林 , 张明 , 郑才俊 , 吉保明 . 一种热活化延迟荧光黄光材料的合成、性能及高效率电致发光器件[J]. 化学学报, 2019 , 77(3) : 263 -268 . DOI: 10.6023/A18100437
A novel yellow thermally activated delayed fluorescence emitter pPBPXZ was successfully synthesized using phenoxazine as electron-donor and pyrimidine as electron-acceptor by Buchwald-Hartwig and Suzuki coupling reactions. Density functional theory calculations show that pPBPXZ has highly twisted structure with the dihedrals of nearly 90° between phenoxazine and pyrimidine units, while the dihedrals between benzene ring and adjacent pyrimidine rings are almost 0°. The highest occupied molecular orbital (HOMO) is mainly confined on two phenoxazine segments, the lowest unoccupied molecular orbital (LUMO) is mainly located on the central pyrimidine and benzene segments, and there is only a slight overlap between HOMO and LUMO. Cyclic voltammetry investigation show pPBPXZ has reversible redox process, and the HOMO and LUMO energy levels were estimated to be -5.43 eV and -3.23 eV, respectively, from the onsets of oxidation and reduction curves. In diluted toluene solution, pPBPXZ exhibits the absorption band assigned to intramolecular charge-transfer transition and yellow fluorescence with a structureless emission peak at 535 nm. From the onsets of the fluorescence and phosphorescence spectra of pPBPXZ in 2Me-THF at 77 K, the lowest singlet (S1) and the lowest triplet (T1) energy levels are calculated to be 2.57 eV and 2.48 eV, respectively, and thus △EST is only 0.09 eV. The doped electroluminescence devices using pPBPXZ as guest emitter were prepared by vacuum evaporation method. These devices with doping ratios (w) of 6%, 11%, 16% and 23% show yellow emission at 552~560 nm and low turn-on voltages of 3.1~3.3 V. The device with a doping ratio of 11% exhibits the highest maximum forward-viewing efficiencies of a maximum current efficiency of 49.9 cd/A, a maximum power efficiency of 49.0 lm/W and a maximum external quantum efficiency of 15.7% without any light out-coupling enhancement. Particularly, the efficiencies of these devices are not sensitive to the doping ratios of pPBPXZ, which would benefit the further practical application.

[1] Sun, Y. R.; Giebink, N. C.; Kanno, H.; Ma, B. W.; Thompson, M. E.; Forrest, S. R. Nature 2006, 440, 908.
[2] Reineke, S.; Lindner, F.; Schwartz, G.; Seidler, N.; Walzer, K.; Lussem, B.; Leo, K. Nature 2009, 459, 234.
[3] Helander, M. G.; Wang, Z. B.; Qiu, J.; Greiner, M. T.; Puzzo, D. P.; Liu, Z. W. Science 2011, 332, 944.
[4] Han, T.-H.; Lee, Y.; Choi, M.-R.; Woo, S.-H.; Hong, B. H.; Ahn, J.-H.; Lee, T.-W. Nat. Photonics 2012, 459, 105.
[5] Sasabe, H.; Kido, J. J. Mater. Chem. C 2013, 1, 1699.
[6] Zhang, Z.; Li, W.; Ye, K.; Zhang, H. Acta Chim. Sinica 2016, 74, 179(in Chinese). (张振宇, 李婉君, 叶开其, 张红雨, 化学学报, 2016, 74, 179.)
[7] Yu, Y.; Yang, J.; Ren, Z.; Xie, G.; Li, Q.; Li, Z. Acta Chim. Sinica 2016, 74, 865.
[8] Wang, F.; Cao, X.; Mei, L.; Zhang, X.; Hu, J.; Tao, Y. Chinese J. Chem. 2018, 36, 241.
[9] Liang, X.; Wang, Z.; Wang, L.; Hanif, M.; Hu, D.; Su, S.; Xie, Z.; Gao, Y.; Yang, B.; Ma, Y. Chinese J. Chem. 2017, 35, 1559.
[10] Lin, D.; Song, S.; Chen, Z.; Guo, P.; Chen, J.; Shi, H.; Mai, Y.; Song, H. Chinese J. Org. Chem. 2018, 38, 103(in Chinese). (林丹燕, 宋森川, 陈智勇, 郭鹏然, 陈江韩, 史华红, 麦裕良, 宋化灿, 有机化学, 2018, 38, 103.)
[11] Xu, H.; Chen, R.; Sun, Q.; Huang, W.; Liu, X. Chem. Soc. Rev. 2014, 43, 3259.
[12] Volz, D.; Wallesch, M.; Fléchon, C.; Danz, M.; Verma, A.; Navarro, J. M.; Zink, D. M.; Bräse, S.; Baumann, T. Green Chem. 2015, 17, 1988.
[13] Chi, Y.; Tong, B.; Chou, P.-T. Coord. Chem. Rev. 2014, 281, 1.
[14] Uoyama, H.; Goushi, K.; Shizu, K.; Nomura, H.; Adachi, C. Nature 2012, 492, 234.
[15] Tao, Y.; Yuan, K.; Chen, T.; Xu, P.; Li, H.; Chen, R.; Zheng, C.; Zhang, L.; Huang, W. Adv. Mater. 2014, 26, 7931.
[16] Kim, M.; Jeon, S. K.; Hwang, S. H.; Lee, S. S.; Yu, E.; Lee, J. Y. Chem. Commun. 2016, 52, 339.
[17] Lee, J.; Aizawa, N.; Yasuda, T. Chem. Mater. 2017, 29, 8012.
[18] Tamai, Y.; Ohkita, H.; Benten, H.; Ito, S.; Lee, J.-H.; Kido, J.; Park, J. J. Mater. Chem. C 2013, 1, 432.
[19] Chen, W.-C.; Lee, C.-S.; Tong, Q.-X. J. Mater. Chem. C 2015, 3, 10957.
[20] Chen, Y.-H.; Lin, C.-C.; Huang, M.-J.; Hung, K.; Wu, Y.-C.; Lin, W.-C.; Chen-Cheng, R.-W.; Lin, H.-W.; Cheng, C.-H. Chem. Sci. 2016, 7, 4044.
[21] Pan, Y.; Li, W.; Zhang, S.; Yao, L.; Gu, C.; Xu, H.; Yang, B.; Ma, Y. Adv. Opt. Mater. 2014, 2, 510.
[22] Konidena, R. K.; Thomas, K. R. J.; Dubey, D. K.; Sahoo, S.; Jou, J.-H. Chem. Commun. 2017, 53, 11802.
[23] Chen, W.-C.; Yuan, Y.; Ni, S.-F.; Tong, Q.-X.; Wong, F.-L.; Lee, C.-S. Chem. Sci. 2017, 8, 3599.
[24] Wang, Z.; Li, Y.; Cai, X.; Chen, D.; Xie, G.; Liu, K.; Wu, Y.; Lo, C.-C.; Lien, A.; Cao, Y.; Su, S.-J. ACS Appl. Mater. Interfaces 2016, 8, 8627.
[25] Chen, D.-Y.; Liu, W.; Zheng, C.-J.; Wang, K.; Li, F.; Tao, S.-L.; Ou, X.-M.; Zhang, X.-H. ACS Appl. Mater. Interfaces 2016, 8, 16791.
[26] Lee, D. R.; Choi, J. M.; Lee, C. W.; Lee, J. Y. ACS Appl. Mater. Interfaces 2016, 8, 23190.
[27] Cai, X.; Li, X.; Xie, G.; He, Z.; Gao, K.; Liu, K.; Chen, D.; Cao, Y.; Su, S.-J. Chem. Sci. 2016, 7, 4264.
[28] Li, F.; Xie, G.; Gong, S.; Wu, K.; Yang, C. Chem. Sci. 2016, 7, 5441.
[29] Rajamalli, P.; Senthilkumar, N.; Gandeepan, P.; Huang, P.-Y.; Huang, M.-J.; Yang, C.-Y.; Chiu, M.-J.; Chu, L.-K.; Lin, H.-W.; Cheng, C.-H. J. Am. Chem. Soc. 2016, 138, 628.
[30] Liu, X.; Zhan, C.; Zheng, C.; Liu, C.; Lee, C.; Li, F.; Ou, X.; Zhang, X. Adv. Mater. 2015, 27, 2378.
[31] Wang, K.; Zheng, C. J.; Liu, W.; Liang, K.; Shi, Y. Z.; Tao, S. L.; Lee, C. S.; Ou, X. M.; Zhang, X. H. Adv. Mater. 2017, 29, 1701476.
[32] Yang, Z.; Mao, Z.; Xie, Z.; Zhang, Y.; Liu, S.; Zhao, J.; Xu, J.; Chi, Z.; Aldred, M. P. Soc. Rev. 2017, 46, 915.
[33] Liu, Y.; Li, C.; Ren, Z.; Yan, S.; Bryce, M. R. Nat. Rev. Mater. 2018, 3, 18020.
[34] Wong, M. Y.; Zysman-Colman, E. Adv. Mater. 2017, 29, 1605444.
[35] Xiang, Y.; Gong, S.; Zhao, Y.; Yin, X.; Luo, J.; Wu, K.; Lu, Z.; Yang, C. J. Mater. Chem. C 2016, 4, 9998.
[36] Xie, G.; Li, X.; Chen, D.; Wang, Z.; Cai, X.; Chen, D.; Li, Y.; Liu, K.; Cao, Y.; Su, S. J. Adv. Mater. 2016, 28, 181.
[37] Chen, J.-X.; Wang, K.; Zheng, C.-J.; Zhang, M.; Shi, Y.-Z.; Tao, S.-L.; Lin, H.; Liu, W.; Tao, W.-W.; Ou, X.-M.; Zhang, X.-H. Adv. Sci. 2018, 5, 1800436.
[38] Chen, X.; Yang, Z.; Xie, Z.; Zhao, J.; Yang, Z.; Aldred, M. P.; Chi, Z. Mater. Chem. Front. 2018, 2, 1017.
[39] Chen, Y.; Li, S.; Hu, T.; Wei, X.; Li, Z.; Liu, W.; Liu, J.; Wang, R.; Yi, Y.; Zhao, C.; Wang, Y.; Wang, P. J. Mater. Chem. C 2018, 6, 2951.
[40] Xiang, Y.; Zhu, Z.-L.; Xie, D.; Gong, S.; Wu, K.; Xie, G.; Lee, C.-S.; Yang, C. J. Mater. Chem. C 2018, 6, 7111.
[41] Huang, J.; Nie, H.; Zeng, J.; Zhuang, Z.; Gan, S.; Cai, Y.; Guo, J.; Su, S.-J. Angew. Chem. Int. Ed. 2017, 56, 12971.
[42] Li, J.; Ding, D. X.; Tao, Y. T.; Wei, Y. Y.; Chen, R. F.; Xie, L. H.; Haung, W.; Xu, H. Adv. Mater. 2016, 28, 3122.
[43] Pan, K.-C.; Li, S.-W.; Ho, Y.-Y.; Shiu, Y.-J.; Tsai, W.-L.; Jiao, M.; Lee, W.-K.; Wu, C.-C.; Chung, C.-L.; Chatterjee, T.; Li, Y.-S.; Wong, K.-T.; Hu, H.-C.; Chen, C.-C.; Lee, M.-T. Adv. Funct. Mater. 2016, 26, 7560.
/
| 〈 |
|
〉 |