

“陈试剂”作为三氟甲基源机理的理论研究
收稿日期: 2023-09-30
网络出版日期: 2023-11-30
基金资助
国家重点研发计划(2021YFF0701700); 国家自然科学基金(22122104); 国家自然科学基金(22193012); 国家自然科学基金(21933004); 中国科学院稳定支持基础研究领域青年团队计划(YSBR-052); 中国科学院稳定支持基础研究领域青年团队计划(YSBR-095)
Computational Study on the Mechanism of Chen’s Reagent as Trifluoromethyl Source
Received date: 2023-09-30
Online published: 2023-11-30
Supported by
Ministry of Science and Technology of China(2021YFF0701700); National Natural Science Foundation of China(22122104); National Natural Science Foundation of China(22193012); National Natural Science Foundation of China(21933004); CAS Project for Young Scientists in Basic Research(YSBR-052); CAS Project for Young Scientists in Basic Research(YSBR-095)
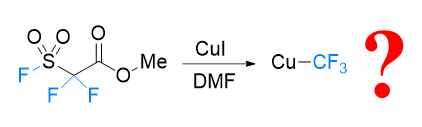
FSO2CF2CO2Me (MFSDA)是陈庆云院士课题组于1989年开发的一种十分有效的三氟甲基化试剂, 在学术界和工业界有着广泛地应用, 是我国有机氟化学领域早期最重要的研究成果之一, 该试剂也被命名为“陈试剂”. 此研究采用密度泛函理论(DFT)方法研究了碘化亚铜介导“陈试剂”形成三氟甲基亚铜的反应机理. 计算结果揭示了反应中碘化亚铜催化剂的双重作用: 一方面碘负离子通过进攻“陈试剂”中具有亲电性的甲基启动反应; 另一方面一价铜阳离子络合物捕获原位生成二氟卡宾形成二氟卡宾亚铜中间体. 发现与过去普遍假设的机理不同, (1)关键的脱羧过程并不需要一价铜阳离子参与, (2)三氟甲基是通过二氟卡宾亚铜中间体与氟离子络合后形成, 而非二氟卡宾直接捕获氟离子. 研究结果丰富了对“陈试剂”形成三氟甲基源的认识, 为进一步开发基于“陈试剂”的新反应提供了理论依据.
关键词: 密度泛函理论(DFT)方法; 氟磺酰二氟乙酸甲酯; 陈试剂; 三氟甲基化试剂; 反应机理
黄广龙 , 薛小松 . “陈试剂”作为三氟甲基源机理的理论研究[J]. 化学学报, 2024 , 82(2) : 132 -137 . DOI: 10.6023/A23090434
In 1989, Qing-Yun Chen’s research group at the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences reported the development of methyl fluorosulfonyldifluoroacetate (FSO2CF2CO2Me or MFSDA) as trifluoromethylation reagent. This reagent is now known as Chen’s reagent, which is perhaps the first well-recognized and widely used trifluoromethylation reagent originate from China. Despite the widespread use of Chen’s reagent in both academia and industry, the detailed mechanism underlying the conversion of Chen’s reagent into a trifluoromethyl source has remained elusive. In this contribution, we conducted a thorough investigation into the reaction mechanism, employing density functional theory (DFT) calculations. Geometry optimizations and frequency analyses were performed using the PBE0/def2-SVP level of theory. To ensure accurate electronic energy calculations, single-point energy calculations were conducted at the ωB97X-D/def2-TZVPP level of theory. The solvent effects were considered using the solvation model density (SMD) model during both geometry optimizations and single-point energy calculations. Furthermore, Gibbs free energies were corrected with GoodVibes, employing Truhlar et al.’s quasi-harmonic treatment by setting all positive frequencies less than 100 to 100 cm–1. Concentration corrections were applied from 1 atm to 1 mol/L. Our calculations reveal the detailed mechanism governing the generation of copper(I) trifluoromethyl from Chen’s reagent in the presence of a CuI catalyst. An in-depth understanding of such mechanistic details would be helpful for future development of new reaction and application with Chen’s reagent.

| [1] | (a) Chen, Q.-Y.; Zhu, S. Z. Sci. China Chem. B 1986, 6, 561. (in Chinese) |
| [1] | (陈庆云, 朱仕正, 中国科学(B辑), 1986, 6, 561.) |
| [1] | (b) Chen, Q.-Y.; Wu, S.-W. J. Chem. Soc., Chem. Commun. 1989, 705. |
| [2] | (a) Jiang, X. K. Acta Chim. Sinica 1957, 5, 330. (in Chinese) |
| [2] | (蔣錫夔, 化学学报, 1957, 5, 330.) |
| [2] | (b) Olah, G. A.; Iyer, P. S.; Prakash, G. K. S. Synthesis 1986, 513. |
| [2] | (c) England, D. C. US 2852554, 1958. |
| [2] | (d) England, D. C.; Dietrich, M. A.; Lindsey, R. V. J. Am. Chem. Soc. 1960, 82, 6181. |
| [2] | (e) Dmitriev, M. A.; Sokol'skii, G. A.; Knunyants, I. L. Bull. Acad. Sci. USSR, Div. Chem. Sci. 1960, 9, 792. |
| [2] | (f) Dmitriev, M. A.; Sokol'skii, G. A.; Knunyants, I. L. Bull. Acad. Sci. USSR, Div. Chem. Sci. 1960, 9, 966. |
| [2] | (g) Zhao, G.; Wu, H.; Xiao, Z.; Chen, Q.-Y.; Liu, C. RSC Adv. 2016, 6, 50250. |
| [2] | (h) Zhang, C.-P.; Chen, Q.-Y; Guo, Y.; Xiao, J.-C.; Gu, Y.-C. Coord. Chem. Rev. 2014, 261, 28. |
| [3] | Chen, Q.-Y.; Yang, G.-Y.; Wu, S.-W. J. Fluorine Chem. 1991, 55, 291. |
| [4] | Zhao, S.; Guo, Y.; Han, E.-J.; Luo, J.; Liu, H.-M.; Liu, C.; Xie, W.; Zhang, W.; Wang, M. Org. Chem. Front. 2018, 5, 1143. |
| [5] | Chen, Q.-Y. J. Fluorine Chem. 1995, 72, 241. |
| [6] | (a) Eusterwiemann, S.; Martinez, H.; Dolbier, W. R. J. Org. Chem. 2012, 77, 5461. |
| [6] | (b) Thomoson, C. S.; Martinez, H.; Dolbier, W. R. J. Fluorine Chem. 2013, 150, 53. |
| [6] | (c) Yu, W.; Xu, X.-H.; Qing, F.-L. Org. Lett. 2016, 18, 5130. |
| [6] | (d) Zhao, G.; Wu, H.; Xiao, Z.; Chen, Q.-Y.; Liu, C. RSC Adv. 2016, 6, 50250. |
| [6] | (e) Liu, Y.; Wu, H.; Guo, Y.; Xiao, J.-C.; Chen, Q.-Y.; Liu, C. Angew. Chem., Int. Ed. 2017, 56, 15432. |
| [7] | (a) Tomashenko, O. A.; Grushin, V. V. Chem. Rev. 2011, 111, 4475. |
| [7] | (b) Clarke, S. L.; McGlacken, G. P. Chem. Eur. J. 2016, 22, 1. |
| [7] | (c) Xie, Q.; Hu, J. Chin. J. Chem. 2020, 38, 202. |
| [7] | (d) Chen, Q. Chin. J. Org. Chem. 2001, 21, 805. (in Chinese) |
| [7] | (陈庆云, 有机化学, 2001, 21, 805.) |
| [8] | (a) Wiemers, D. M.; Burton, D. J. J. Am. Chem. Soc. 1986, 108, 832. |
| [8] | (b) Dubinina, G. G.; Furutachi, H.; Vicic, D. A. J. Am. Chem. Soc. 2008, 130, 8600. |
| [8] | (c) Morimoto, H.; Tsubogo, T.; Litvinas, N. D.; Hartwig, J. F. Angew. Chem., Int. Ed. 2011, 50, 3793. |
| [8] | (d) Litvinas, N. D.; Fier, P. S.; Hartwig, J. F. Angew. Chem., Int. Ed. 2012, 51, 536. |
| [8] | (e) Ni, C.; Hu, J. Synthesis 2014, 46, 842. |
| [8] | (f) Konovalov, A. I.; Lishchynskyi, A.; Grushin, V. V. J. Am. Chem. Soc. 2014, 136, 13410. |
| [8] | (g) de Salinas, S. M.; Mudarra, á. L.; Odena, C.; Belmonte, M. M.; Benet-Buchholz, J.; Maseras, F.; Pérez-Temprano, M. H. Chem. Eur. J. 2019, 25, 9390. |
| [8] | (h) Yu, W.; Ouyang, Y.; Xu, X.-H.; Qing, F.-L. Chin. J. Chem. 2018, 36, 1024. |
| [8] | (i) Zhang, W.; Lin, J.-H.; Wu, W.; Cao, Y.-C.; Xiao, J.-C. Chin. J. Chem. 2020, 38, 169. |
| [9] | Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Petersson, G. A.; Nakatsuji, H.; Li, X.; Caricato, M.; Marenich, A. V.; Bloino, J.; Janesko, B. G.; Gomperts, R.; Mennucci, B.; Hratchian, H. P.; Ortiz, J. V.; Izmaylov, A. F.; Sonnenberg, J. L.; Williams-Young, D.; Ding, F.; Lipparini, F.; Egidi, F.; Goings, J.; Peng, B.; Petrone, A.; Henderson, T.; Ranasinghe, D.; Zakrzewski, V. G.; Gao, J.; Rega, N.; Zheng, G.; Liang, W.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Throssell, K.; Montgomery Jr., J. A.; Peralta, J. E.; Ogliaro, F.; Bearpark, M. J.; Heyd, J. J.; Brothers, E. N.; Kudin, K. N.; Staroverov, V. N.; Keith, T. A.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A. P.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Millam, J. M.; Klene, M.; Adamo, C.; Cammi, R.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Farkas, O.; Foresman, J. B.; Fox, D. J. Gaussian 16, Revision A.03, Gaussian, Inc., Wallingford CT, 2016. |
| [10] | Marenich, A. V.; Cramer, C. J.; Truhlar, D. G. J. Phys. Chem. B 2009, 113, 6378. |
| [11] | (a) Zhao, Y.; Truhlar, D. G. J. Chem. Phys. 2006, 125, 194101. |
| [11] | (b) Quintal, M. M.; Karton, A.; Iron, M. A.; Boese, A. D.; Martin, J. M. L. J. Phys. Chem. A 2006, 110, 709. |
| [12] | Weigend, F.; Ahlrichs, R. Phys. Chem. Chem. Phys. 2005, 7, 3297. |
| [13] | Chai, J.-D.; Head-Gordon, M. Phys. Chem. Chem. Phys. 2008, 10, 6615. |
| [14] | Weigend, F. Phys. Chem. Chem. Phys. 2006, 8, 1057. |
| [15] | Funes-Ardoiz, I.; Paton, R. S., GoodVibes v2. 0.2, 2016, (http://doi.org/10.5281/zenodo.595246). |
| [16] | Legault, C. Y., CYLview, 1.0b, Université de Sherbrooke, 2009. (http://www.cylview.org). |
/
| 〈 |
|
〉 |