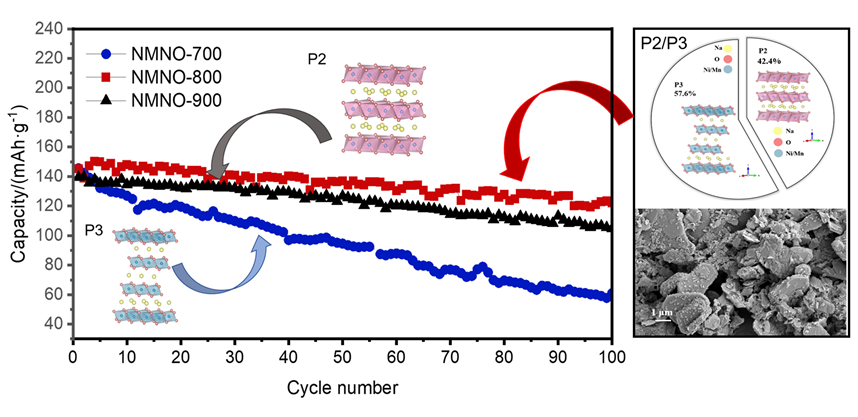
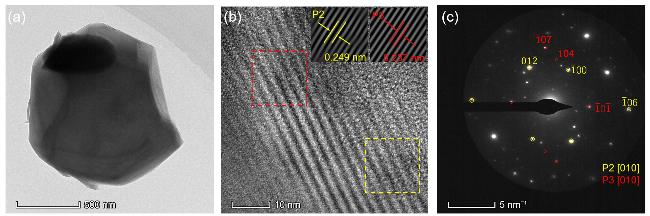
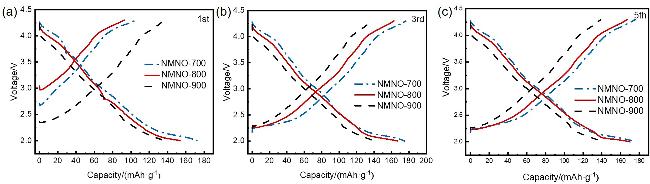
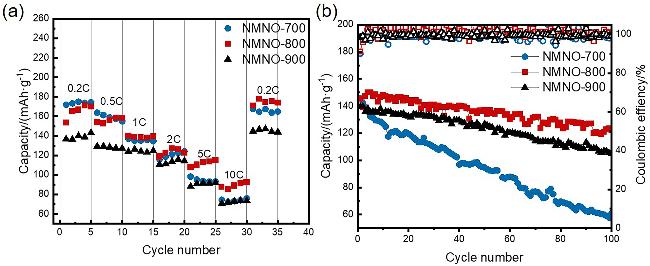
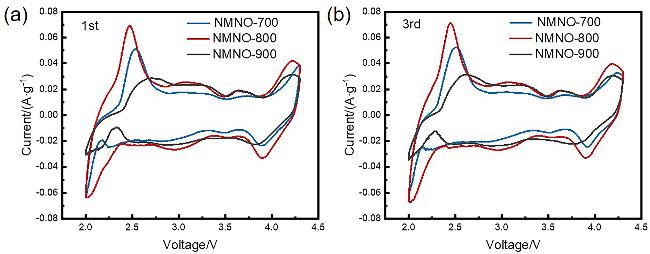
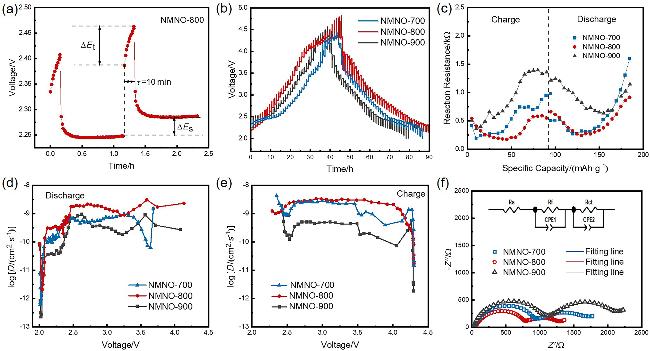
通过调控煅烧温度合成了三种高锰Na
0.67Mn
0.9Ni
0.1O
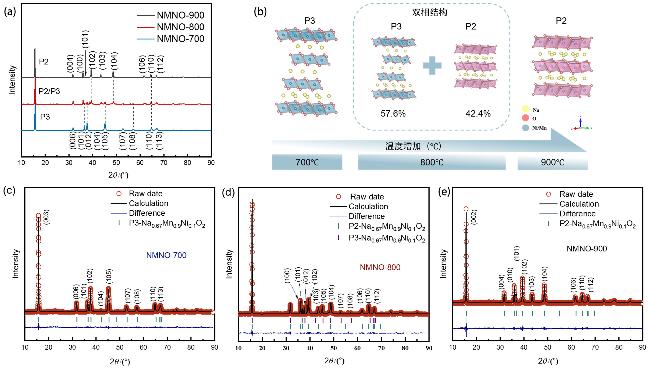
2正极材料, 利用X射线衍射(XRD)分析煅烧温度对材料的晶体结构的影响规律. NMNO-700、NMNO-800和NMNO-900的XRD谱图如
图1(a)所示. NMNO-700是典型的P3相材料(空间群
R3
m); 当温度从700 ℃上升到800 ℃时, NMNO-800中表明P3相的衍射峰(101)、(012)、(105)、(107)和(108)强度降低, 并出现了属于P2相材料的新峰(100)、(102)、(103)、(104)和(106); 当温度达到900 ℃时, NMNO-900中P3结构完全转变为P2结构(空间群
P63/
mmc)
[34-35]. 因此, XRD表明改变煅烧温度可以使得其材料呈P2/P3双相.
图1(b)为不同煅烧温度下制备的三种材料的晶体结构示意图. 对NMNO-700、NMNO-800、NMNO-900结构进行Rietveld精修, 以阐明晶体结构和晶胞参数(
图1(c~e),
表1). 结果表明, NMNO-800为P2/P3双相结构, 其比例分别为42.4%和57.6%. 比较三种样品的晶格参数和结构信息, P3相NMNO-700 (
a=
b=0.2858 nm,
c=1.67923 nm), P2/P3双相NMNO-800 (P2相:
a=
b=0.28771 nm,
c=1.122952 nm; P3相:
a=
b=0.28787(6) nm,
c=1.67589(8) nm)和P2相NMNO-900 (
a=
b=0.28821 nm,
c=1.129538 nm), 清楚地反映了随着煅烧温度的升高, 材料逐渐从P3相转变为P2相. 另外, 纯P2相NMNO-900的
c轴小于NMNO-800和NMNO-700中P3相的
c轴, 这表明P2相材料的层间间距比P3相更窄, 这可能是由于较高的温度促使Ni—O键断裂或重组, 正极材料中的钠层滑移, 过渡金属层的堆叠顺序从ABBCCA (P3相)转变为ABBA (P2相)
[36]. 通过Debye-Scherrer计算了NMNO的晶粒尺寸, 由下式所示: