Acta Chimica Sinica ›› 2026, Vol. 84 ›› Issue (3): 341-352.DOI: 10.6023/A25110367 Previous Articles Next Articles
Article
赵馨雨a,b, 韩燕楠c, 徐吉磊b,*( ), 安庆大a,*(
), 安庆大a,*( ), 肖作毅a, 苏鑫b, 黄家辉b,*(
), 肖作毅a, 苏鑫b, 黄家辉b,*( )
)
投稿日期:2025-11-12
发布日期:2026-01-20
基金资助:
Zhao Xinyua,b, Han Yannanc, Xu Jileib,*( ), An Qingdaa,*(
), An Qingdaa,*( ), Xiao Zuoyia, Su Xinb, Huang Jiahuib,*(
), Xiao Zuoyia, Su Xinb, Huang Jiahuib,*( )
)
Received:2025-11-12
Published:2026-01-20
Contact:
*E-mail: xujilei@dicp.ac.cn;
anqingda@dlpu.edu.cn;
jiahuihuang@dicp.ac.cn
Supported by:Share
Zhao Xinyu, Han Yannan, Xu Jilei, An Qingda, Xiao Zuoyi, Su Xin, Huang Jiahui. Preparation of Bio-based Polyacid Esters Catalyzed by Supported Ionic Liquids[J]. Acta Chimica Sinica, 2026, 84(3): 341-352.
| Entry | Catalyst | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | Pyrrolidine-YLST-3 | 100 | 79.1 | 79.1 |
| 2 | Pyrrolidine-D113 | 100 | 63.8 | 63.8 |
| 3 | Ethanolamine-YLST-3 | 97.0 | 60.0 | 61.5 |
| 4 | Ethanolamine-D113 | 99.2 | 73.4 | 74.0 |
| 5 | Diethanolamine-YLST-3 | 11.6 | 0 | 0 |
| 6 | Diethanolamine-D113 | 9.9 | 0 | 0 |
| 7b | Pyrrolidine-YLST-3 | 92.7 | 68.9 | 74.4 |
| 8b | Ethanolamine-D113 | 98.7 | 58.8 | 59.5 |
| 9 | YLST-3 | 18.2 | 0 | 0 |
| 10 | Pyrrolidine | 100 | 19.4 | 19.4 |
| Entry | Catalyst | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | Pyrrolidine-YLST-3 | 100 | 79.1 | 79.1 |
| 2 | Pyrrolidine-D113 | 100 | 63.8 | 63.8 |
| 3 | Ethanolamine-YLST-3 | 97.0 | 60.0 | 61.5 |
| 4 | Ethanolamine-D113 | 99.2 | 73.4 | 74.0 |
| 5 | Diethanolamine-YLST-3 | 11.6 | 0 | 0 |
| 6 | Diethanolamine-D113 | 9.9 | 0 | 0 |
| 7b | Pyrrolidine-YLST-3 | 92.7 | 68.9 | 74.4 |
| 8b | Ethanolamine-D113 | 98.7 | 58.8 | 59.5 |
| 9 | YLST-3 | 18.2 | 0 | 0 |
| 10 | Pyrrolidine | 100 | 19.4 | 19.4 |
| Entry | Solvent | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | ethyl acetate | 100 | 89.9 | 89.9 |
| 2 | tetrahydrofuran | 99.3 | 81.1 | 81.7 |
| 3 | dichloromethane | 100 | 82.3 | 82.3 |
| 4 | chloroform | 100 | 61.9 | 61.9 |
| 5 | acetonitrile | 100 | 79.1 | 79.1 |
| 6 | methanol | 100 | 44.8 | 44.8 |
| 7 | ethanol | 100 | 54.5 | 54.5 |
| 8 | isopropanol | 100 | 54.8 | 54.8 |
| 9 | DMSO | 100 | 6.8 | 6.80 |
| 10 | DMF | 100 | 17.5 | 17.5 |
| Entry | Solvent | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | ethyl acetate | 100 | 89.9 | 89.9 |
| 2 | tetrahydrofuran | 99.3 | 81.1 | 81.7 |
| 3 | dichloromethane | 100 | 82.3 | 82.3 |
| 4 | chloroform | 100 | 61.9 | 61.9 |
| 5 | acetonitrile | 100 | 79.1 | 79.1 |
| 6 | methanol | 100 | 44.8 | 44.8 |
| 7 | ethanol | 100 | 54.5 | 54.5 |
| 8 | isopropanol | 100 | 54.8 | 54.8 |
| 9 | DMSO | 100 | 6.8 | 6.80 |
| 10 | DMF | 100 | 17.5 | 17.5 |
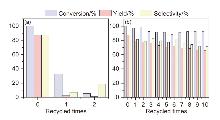
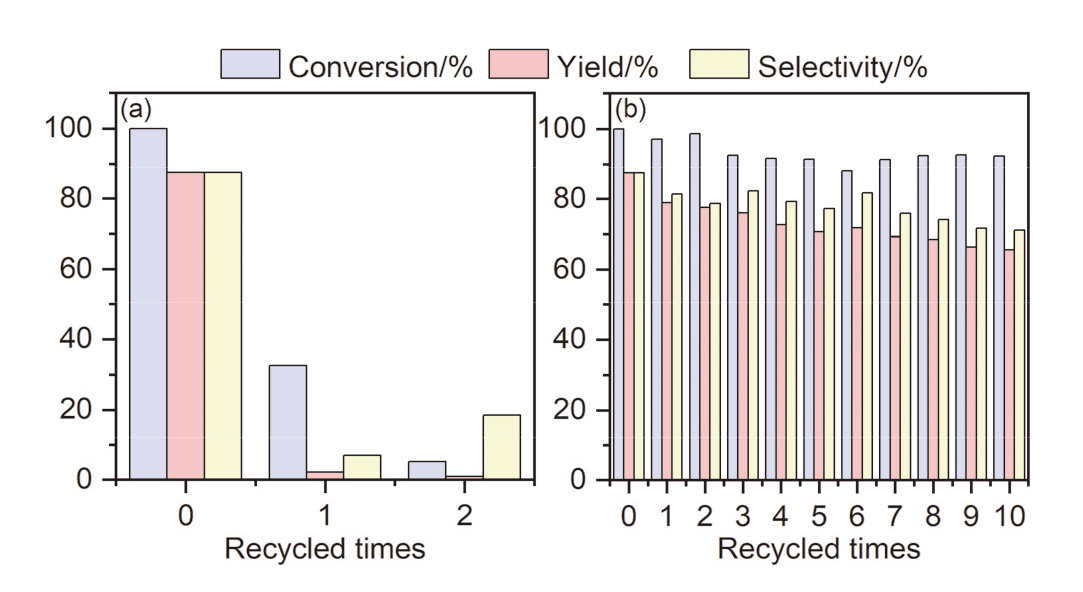
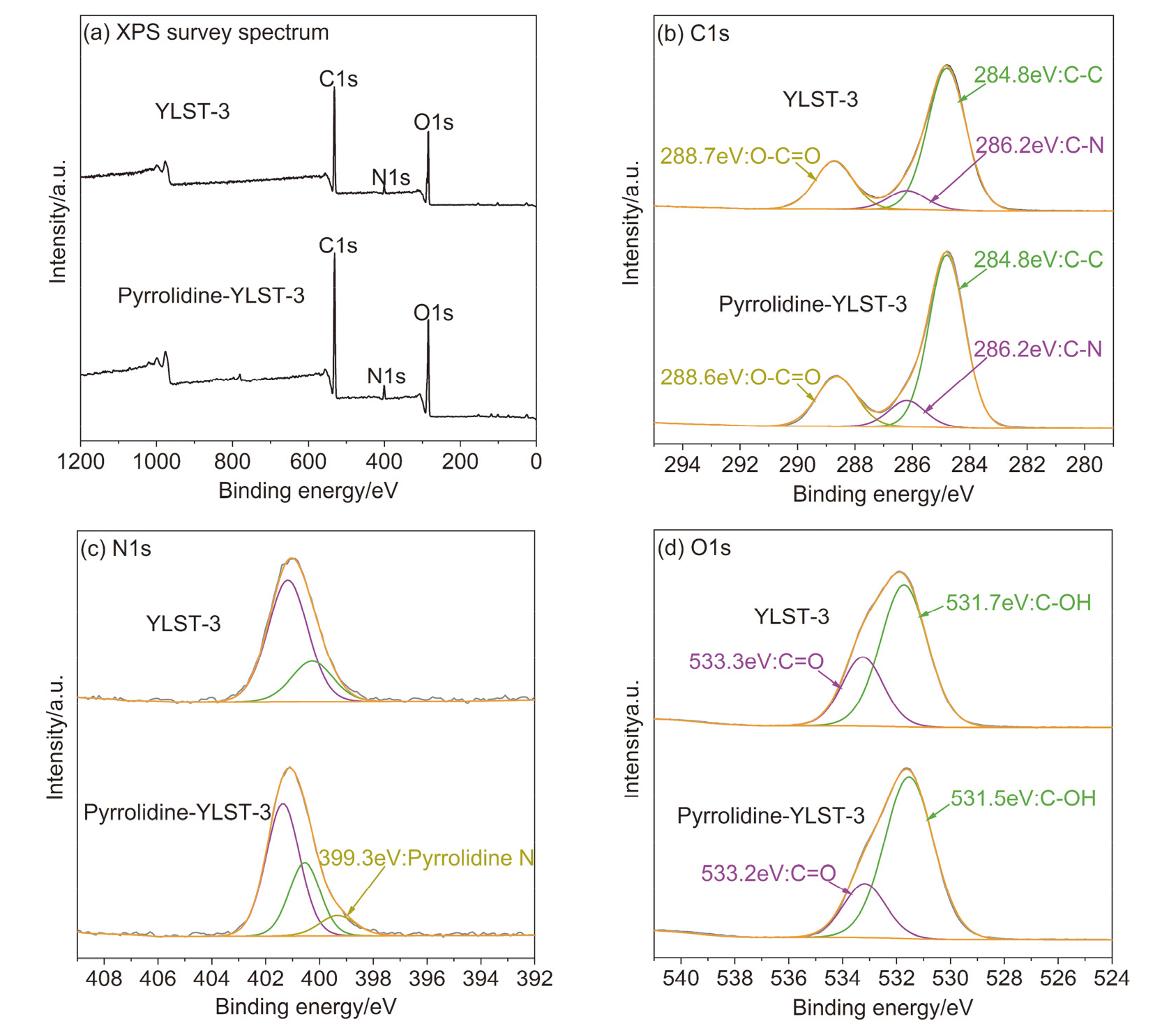
| Entry | Catalyst | Elemental content/% | ||||
|---|---|---|---|---|---|---|
| N | C | H | O | |||
| 1 | Pyrrolidine-YLST-3 | 7.0 | 54.6 | 8.4 | 30.0 | |
| 2 | used catalyst | 4.8 | 54.5 | 7.2 | 33.5 | |
| 3 | regenerated catalyst | 5.9 | 52.1 | 7.6 | 34.4 | |
| Entry | Catalyst | Elemental content/% | ||||
|---|---|---|---|---|---|---|
| N | C | H | O | |||
| 1 | Pyrrolidine-YLST-3 | 7.0 | 54.6 | 8.4 | 30.0 | |
| 2 | used catalyst | 4.8 | 54.5 | 7.2 | 33.5 | |
| 3 | regenerated catalyst | 5.9 | 52.1 | 7.6 | 34.4 | |
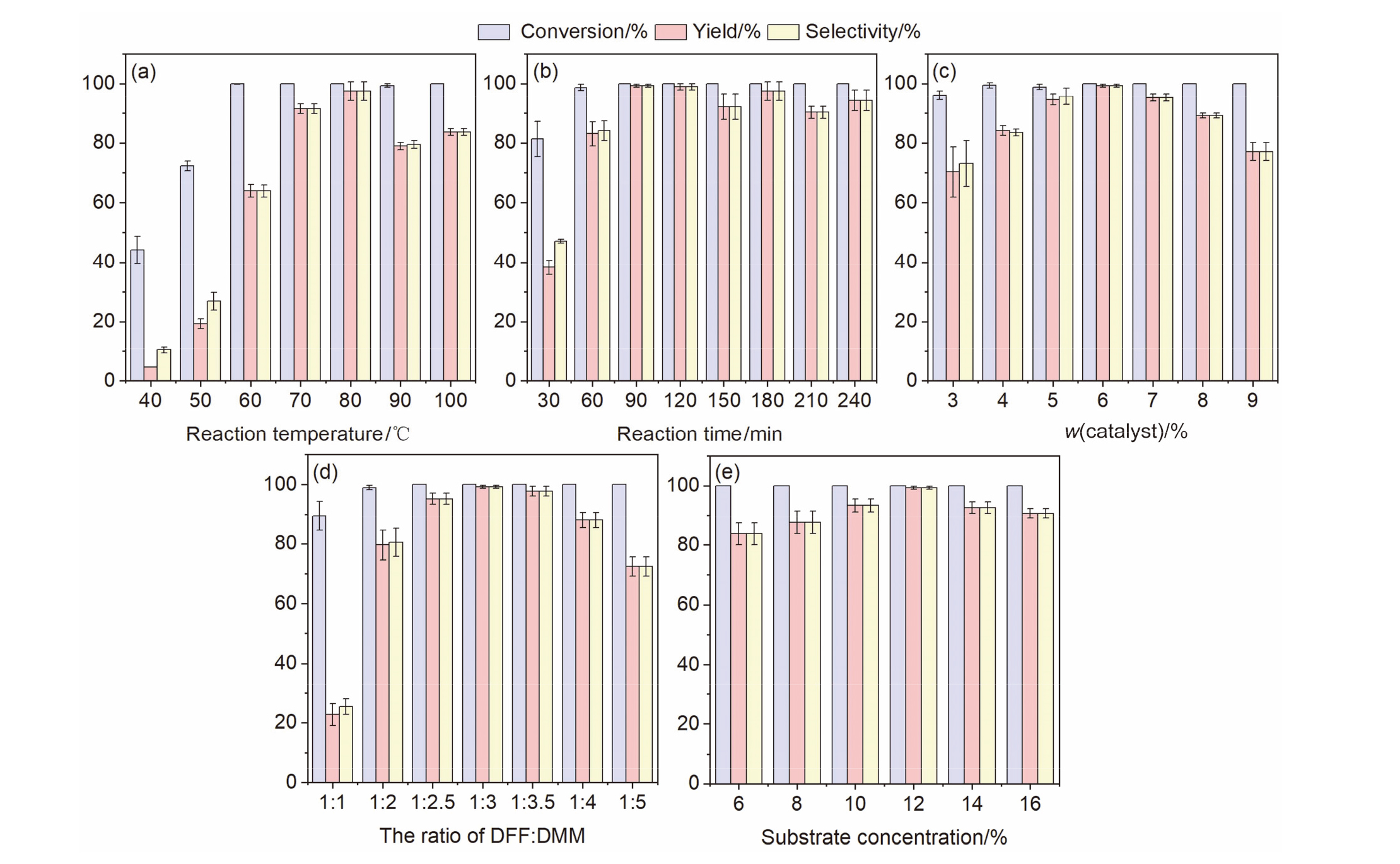
| Entry | Substrate 1 | Substrate 2 | Product | Temp./℃ | Time/min | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|---|---|---|---|
| 1a |  | 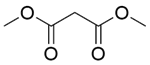 | 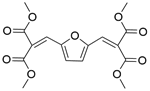 | 80 | 90 | 100 | 99.2 | 99.2 |
| 2a |  |  |  | 60 | 120 | 100 | 65.3 | 65.3 |
| 3a |  | 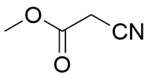 | 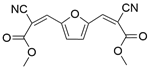 | 80 | 30 | 100 | 61.9 | 61.9 |
| 4a | 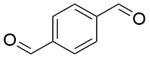 | 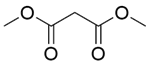 | 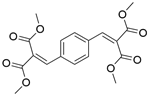 | 100 | 180 | 98.2 | 77.3 | 78.8 |
| 5b | 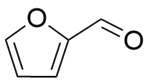 | 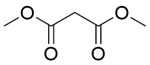 | 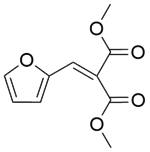 | 50 | 120 | 100 | 96.7 | 96.7 |
| 6b |  | 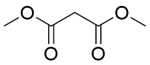 | 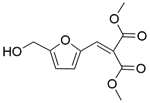 | 70 | 120 | 100 | 82.6 | 82.6 |
| 7b | 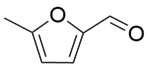 | 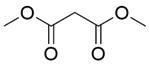 | 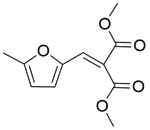 | 70 | 120 | 99.8 | 94.3 | 94.5 |
| 8b | 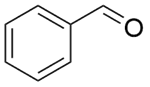 | 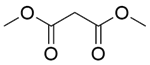 | 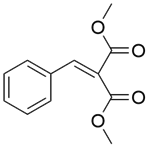 | 70 | 120 | 94.5 | 92.1 | 97.5 |
| 9c | 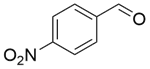 | 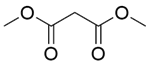 | 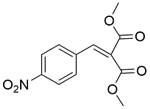 | 80 | 300 | 91.9 | 83.4 | 90.7 |
| 10b | 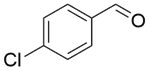 | 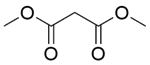 | 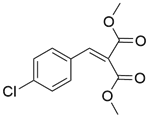 | 80 | 300 | 95.5 | 76.3 | 80.3 |
| 11b | 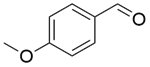 | 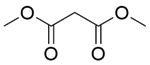 | 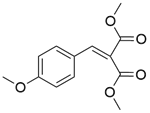 | 80 | 300 | 90.2 | 86.6 | 96.1 |
| 12c | 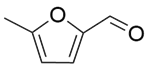 |  | 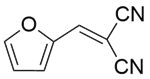 | 40 | 120 | 97.9 | 76.9 | 78.5 |
| 13b | 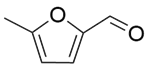 | 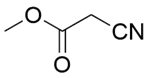 | 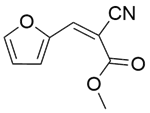 | 40 | 120 | 100 | 97.0 | 97.0 |
| Entry | Substrate 1 | Substrate 2 | Product | Temp./℃ | Time/min | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|---|---|---|---|
| 1a |  |  |  | 80 | 90 | 100 | 99.2 | 99.2 |
| 2a |  |  |  | 60 | 120 | 100 | 65.3 | 65.3 |
| 3a |  |  |  | 80 | 30 | 100 | 61.9 | 61.9 |
| 4a |  |  |  | 100 | 180 | 98.2 | 77.3 | 78.8 |
| 5b |  |  |  | 50 | 120 | 100 | 96.7 | 96.7 |
| 6b |  |  |  | 70 | 120 | 100 | 82.6 | 82.6 |
| 7b |  |  |  | 70 | 120 | 99.8 | 94.3 | 94.5 |
| 8b |  |  |  | 70 | 120 | 94.5 | 92.1 | 97.5 |
| 9c |  |  |  | 80 | 300 | 91.9 | 83.4 | 90.7 |
| 10b |  |  |  | 80 | 300 | 95.5 | 76.3 | 80.3 |
| 11b |  |  |  | 80 | 300 | 90.2 | 86.6 | 96.1 |
| 12c |  |  |  | 40 | 120 | 97.9 | 76.9 | 78.5 |
| 13b |  |  |  | 40 | 120 | 100 | 97.0 | 97.0 |
| [1] |
|
| [2] |
doi: 10.1016/j.gr.2023.09.020 |
| [3] |
|
| [4] |
doi: 10.1002/cptc.v4.8 |
| [5] |
doi: 10.14419/ijac.v3i2 |
| [6] |
doi: 10.1016/j.fuel.2024.133423 |
| [7] |
doi: 10.6023/A24080246 |
|
(鞠嘉浩, 徐吉磊, 王康军, 黄家辉, 化学学报, 2024, 82, 1216.)
doi: 10.6023/A24080246 |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
doi: 10.3390/polym14183868 |
| [12] |
doi: 10.6023/A24100329 |
|
(辛翠, 蒋俊, 邓紫微, 欧丽娟, 何卫民, 化学学报, 2024, 82, 1109.)
doi: 10.6023/A24100329 |
|
| [13] |
doi: 10.6023/A23120542 |
|
(郭建荣, 张书玉, 贺军辉, 任世学, 化学学报, 2024, 82, 242.)
doi: 10.6023/A23120542 |
|
| [14] |
|
| [15] |
doi: 10.1016/j.enconman.2009.03.013 |
| [16] |
doi: 10.1039/D3GC04663J |
| [17] |
|
| [18] |
doi: 10.3390/ijms26020727 |
| [19] |
doi: 10.1002/vnl.v30.4 |
| [20] |
doi: 10.1177/10915818231158272 |
| [21] |
doi: 10.3390/ijms20184333 |
| [22] |
|
| [23] |
doi: 10.1002/adhm.v4.2 |
| [24] |
doi: 10.1002/ls.161 |
| [25] |
doi: 10.1016/j.jconrel.2023.01.002 |
| [26] |
doi: 10.1038/nbt0892-894 pmid: 1368984 |
| [27] |
doi: 10.3389/fmicb.2017.02184 pmid: 29163455 |
| [28] |
doi: 10.1002/jctb.v50:4 |
| [29] |
doi: 10.1016/j.electacta.2022.140796 |
| [30] |
doi: 10.1016/j.tetlet.2004.09.037 |
| [31] |
doi: 10.1016/j.apcata.2004.10.039 |
| [32] |
doi: 10.2478/pjct-2018-0061 |
| [33] |
doi: 10.3390/catal10121443 |
| [34] |
|
|
(李建, 刘燕, 干丰丰, 杨运信, 分析试验室, 2017, 36, 726.)
|
|
| [35] |
doi: 10.1021/acssuschemeng.8b04310 |
| [36] |
doi: 10.1016/j.gee.2023.02.004 |
| [37] |
doi: 10.6023/A25030099 |
|
(刘珊珊, 董微微, 李珍珍, 张瑶瑶, 李超, 焦林郁, 化学学报, 2025, 83, 479.)
doi: 10.6023/A25030099 |
|
| [38] |
|
| [39] |
doi: 10.2174/1385272827666230406082857 |
| [40] |
doi: 10.1007/s10562-017-2255-6 |
| [41] |
doi: 10.1016/j.jscs.2019.01.001 |
| [42] |
doi: 10.1002/slct.v5.46 |
| [43] |
doi: 10.1002/macp.v205:7 |
| [44] |
doi: 10.1016/S0022-2860(01)00719-0 |
| [45] |
doi: 10.1134/S0022476617050080 |
| [46] |
pmid: 17044721 |
| [47] |
doi: 10.1063/1.3633699 |
| [48] |
doi: 10.1002/aic.v42:1 |
| [49] |
doi: 10.1016/j.tsf.2015.07.051 |
| [50] |
pmid: 15253839 |
| [51] |
doi: 10.1021/acs.joc.2c00047 |
| [52] |
doi: 10.1016/j.electacta.2022.141512 |
| [53] |
doi: 10.1021/acs.iecr.9b01375 |
| [54] |
doi: 10.1016/j.molstruc.2018.04.078 |
| [55] |
doi: 10.1016/j.catcom.2020.105954 |
| [56] |
doi: 10.1039/C6RA25595G |
| [57] |
doi: 10.2174/1570179412666150505185134 |
| [1] | Wang Yike, Chen Liping, Shu Jingli, Zhu Xuehua, Wang Yang. Efficient Anti-Markovnikov Hydrothiolation of Alkenes via Phase-Transfer Catalysis in Aqueous Phase [J]. Acta Chimica Sinica, 2026, 84(3): 299-304. |
| [2] | Yuhan Liu, Pan Gao. Direct Borylation of Organohalides Using Mechanochemically Generated Calcium-Based Heavy Grignard Reagents (R-CaX) [J]. Acta Chimica Sinica, 2024, 82(11): 1114-1119. |
| [3] | Li Liu, Gang Zheng, Guoqiang Fan, Hongguang Du, Jiajing Tan. Research Progress in Organic Reactions Involving 4-Acyl/Carbamoyl/Alkoxycarbonyl Substituted Hantzsch Esters [J]. Acta Chimica Sinica, 2023, 81(6): 657-668. |
| [4] | Luyao Yu, Zhen Ren, Yusen Yang, Min Wei. Directed Preparation of Biomass-based Polyester Monomers by Catalytic Conversion [J]. Acta Chimica Sinica, 2023, 81(2): 175-190. |
| [5] | Jingpeng Li, Qi Yang, Zhou Zhang, Guiyun Zeng, Teng Liu, Chao Huang. Highly Selective Synthesis of (Z)-N-vinyl Ring N,O-Acetal Derivatives by Multi-component Continuous Flow [J]. Acta Chimica Sinica, 2022, 80(11): 1463-1468. |
| [6] | Lingling Li, Yu Liu, Shuyan Song, Hongjie Zhang. Synthesis of Cu Single Atom with Adjustable Coordination Environment and Its Catalytic Hydrogenation Performance※ [J]. Acta Chimica Sinica, 2022, 80(1): 16-21. |
| [7] | Wei Zheyu, Chang Yalin, Yu Han, Han Sheng, Wei Yongge. Application of Anderson Type Heteropoly Acids as Catalysts in Organic Synthesis [J]. Acta Chimica Sinica, 2020, 78(8): 725-732. |
| [8] | Liu Jianguo, Zhang Mingyue, Wang Nan, Wang Chenguang, Ma Longlong. Research Progress of Covalent Organic Framework Materials in Catalysis [J]. Acta Chimica Sinica, 2020, 78(4): 311-325. |
| [9] | Dong Kui, Liu Qiang, Wu Li-Zhu. Cross-Coupling Hydrogen Evolution Reactions [J]. Acta Chimica Sinica, 2020, 78(4): 299-310. |
| [10] | Ye Wenbo, Yan Zicong, Wan Changfeng, Hou Haoqing, Wang Zhiyong. A New Decarboxylation/Methylation Process of Cinnamic Acids [J]. Acta Chim. Sinica, 2018, 76(2): 99-102. |
| [11] | Guo Xiaoling, Chen Xiao, Su Dangsheng, Liang Changhai. Preparation of Ni/C Core-shell Nanoparticles through MOF Pyrolysis for Phenylacetylene Hydrogenation Reaction [J]. Acta Chim. Sinica, 2018, 76(1): 22-29. |
| [12] | Zhang Wen-Qiang, Li Qiu-Yan, Yang Xinyu, Ma Zheng, Wang Huanhuan, Wang Xiao-Jun. Benzothiadiazole Conjugated Metalorganic Framework for Organic Aerobic Oxidation Reactions under Visible Light [J]. Acta Chim. Sinica, 2017, 75(1): 80-85. |
| [13] | Pei Pengkun, Zhang Fan, Yi Hong, Lei Aiwen. Visible Light Promoted Benzylic Csp3-H Bond Activation and Functionalization [J]. Acta Chim. Sinica, 2017, 75(1): 15-21. |
| [14] | Huang Gang, Chen Yuzhen, Jiang Hailong. Metal-Organic Frameworks for Catalysis [J]. Acta Chimica Sinica, 2016, 74(2): 113-129. |
| [15] | Wang Changan, Wang Wei. Advances in Porous Organic Catalysis [J]. Acta Chim. Sinica, 2015, 73(6): 498-529. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||