Acta Chimica Sinica ›› 2026, Vol. 84 ›› Issue (4): 490-497.DOI: 10.6023/A25120391 Previous Articles Next Articles
Article
雷晨a, 赵鑫a, 董婧a, 林文军a, 张蕊a, 王传军a,*( ), 王国强b,*(
), 王国强b,*( )
)
投稿日期:2025-12-02
发布日期:2026-02-14
通讯作者:
王传军, 王国强
基金资助:
Chen Leia, Xin Zhaoa, Jing Donga, Wenjun Lina, Rui Zhanga, Chuanjun Wanga,*( ), Guoqiang Wangb,*(
), Guoqiang Wangb,*( )
)
Received:2025-12-02
Published:2026-02-14
Contact:
Chuanjun Wang, Guoqiang Wang
Supported by:Share
Chen Lei, Xin Zhao, Jing Dong, Wenjun Lin, Rui Zhang, Chuanjun Wang, Guoqiang Wang. Biomass-derived Carbon-supported Nickel-iron Phosphide Catalyst for Electrocatalytic Synthesis of Ammonia from Nitrogen-containing Pollutants[J]. Acta Chimica Sinica, 2026, 84(4): 490-497.
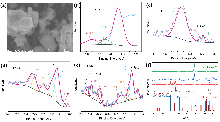
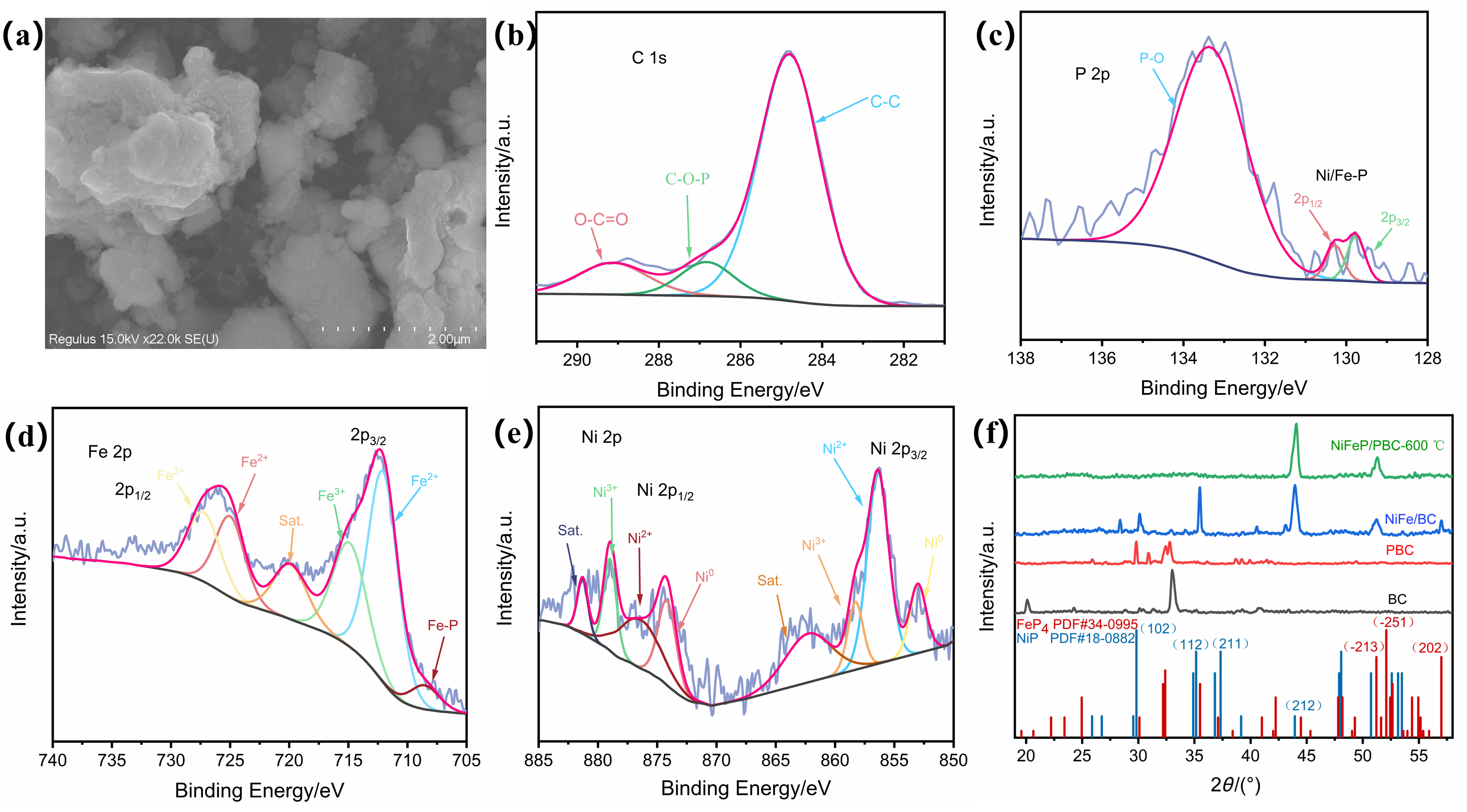
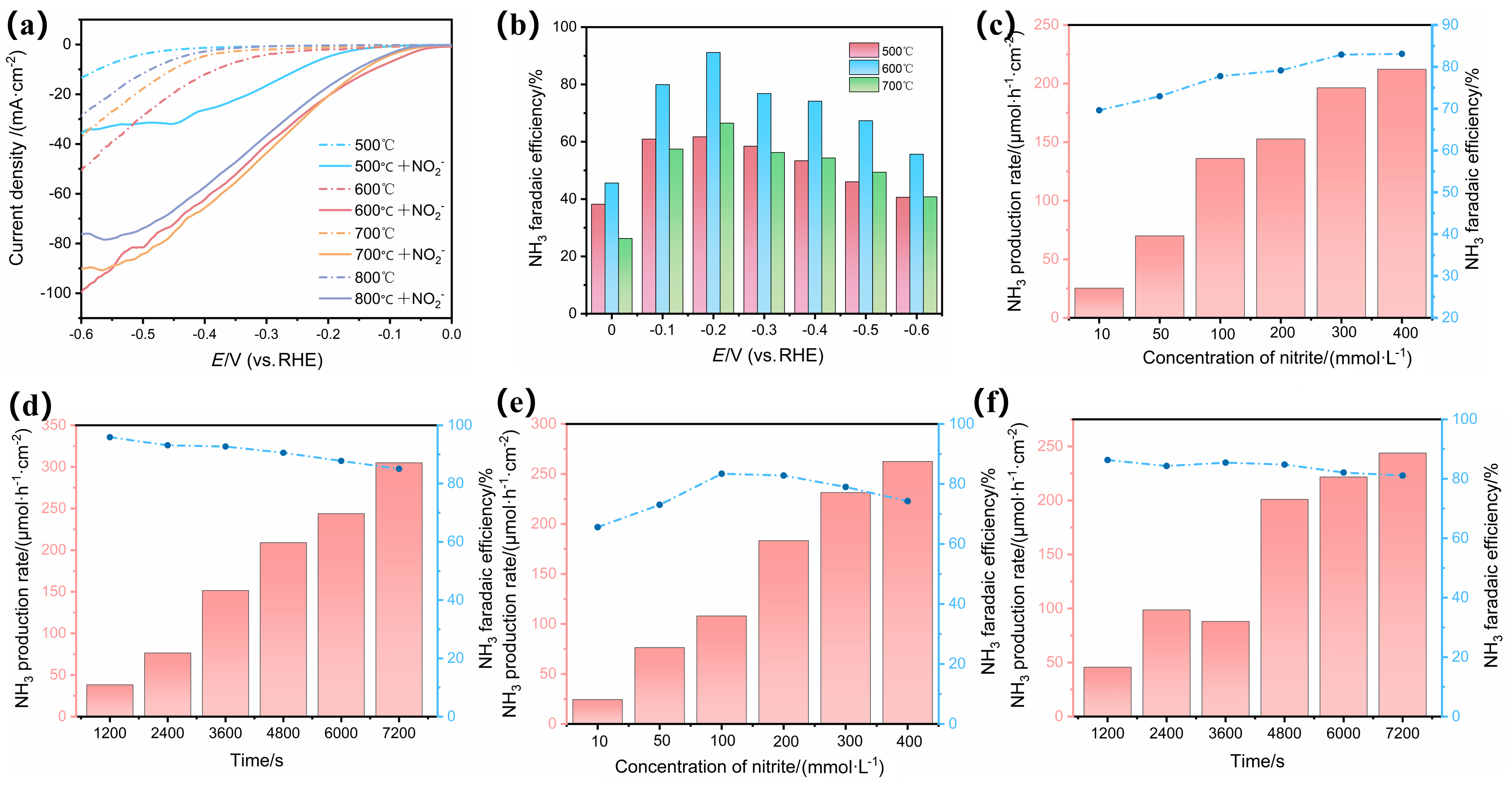
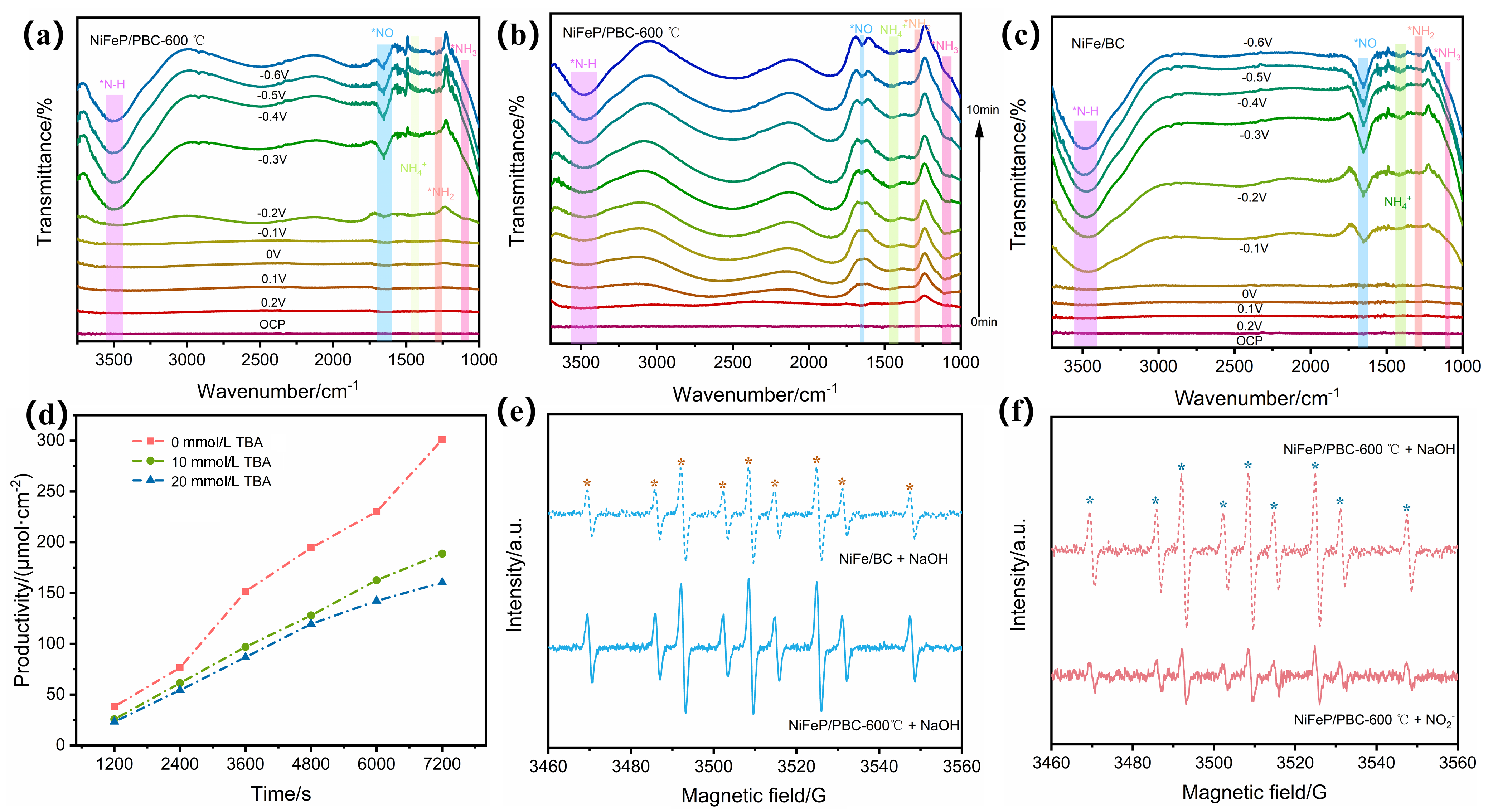
| 催化剂 | 电解质 | 氨产率 | 法拉第效率/% | 电压a | 参考文献 |
|---|---|---|---|---|---|
| Cu@Cu2O MSs | 0.1 mol•L−1 Na2SO4 | 0.3 mol•h−1•g−1 | 80.6 | -1.10 V | [ |
| Cu/Ti3C2 | 0.1 mol•L−1 KOH | 3.0 μmol•h−1•cm−2 | 7.3 | -0.50 V | [ |
| NiFe-LDH | 0.25 mol•L−1 Li2SO4 | 0.1 mmol•h−1•cm−2 | 82 | -0.70 V | [ |
| Cu10Fe1 纳米合金 | 0.5 mol•L−1 Na2SO4 | 0.2 mmol•h−1•cm−2 | 93.7 | -0.35 V | [ |
| CoP | 0.2 mol•L−1 Na2SO4 | 47.2 μmol•h−1•cm−2 | 88.3 | -0.20 V | [ |
| CoS1-x | 0.2 mol•L−1 Na2SO4 | 44.7 μmol•h−1•cm−2 | 53.6 | -0.40 V | [ |
| NiCoP | 0.1 mol•L−1 Na2SO4 | 5553.4 mg•h−1•cm−2 | 91.3 | -1.20 V | [ |
| 竹笋NC-800 | 0.1 mol•L−1 HCl | 16.28 μg•h−1•mg−1 | 27.5 | -0.35 V | [ |
| 木棉纤维O-KFCNTs | 0.1 mol•L−1 HCl | 25.12 μg•h−1•mg−1 | 9.1 | -0.80 V | [ |
| NiFeP/PBC-600 ℃ | 0.1 mol•L−1 NaNO2 | 304.9 μmol•h−1•cm−2 | 95.9 | -0.20 V | 本工作 |
| 催化剂 | 电解质 | 氨产率 | 法拉第效率/% | 电压a | 参考文献 |
|---|---|---|---|---|---|
| Cu@Cu2O MSs | 0.1 mol•L−1 Na2SO4 | 0.3 mol•h−1•g−1 | 80.6 | -1.10 V | [ |
| Cu/Ti3C2 | 0.1 mol•L−1 KOH | 3.0 μmol•h−1•cm−2 | 7.3 | -0.50 V | [ |
| NiFe-LDH | 0.25 mol•L−1 Li2SO4 | 0.1 mmol•h−1•cm−2 | 82 | -0.70 V | [ |
| Cu10Fe1 纳米合金 | 0.5 mol•L−1 Na2SO4 | 0.2 mmol•h−1•cm−2 | 93.7 | -0.35 V | [ |
| CoP | 0.2 mol•L−1 Na2SO4 | 47.2 μmol•h−1•cm−2 | 88.3 | -0.20 V | [ |
| CoS1-x | 0.2 mol•L−1 Na2SO4 | 44.7 μmol•h−1•cm−2 | 53.6 | -0.40 V | [ |
| NiCoP | 0.1 mol•L−1 Na2SO4 | 5553.4 mg•h−1•cm−2 | 91.3 | -1.20 V | [ |
| 竹笋NC-800 | 0.1 mol•L−1 HCl | 16.28 μg•h−1•mg−1 | 27.5 | -0.35 V | [ |
| 木棉纤维O-KFCNTs | 0.1 mol•L−1 HCl | 25.12 μg•h−1•mg−1 | 9.1 | -0.80 V | [ |
| NiFeP/PBC-600 ℃ | 0.1 mol•L−1 NaNO2 | 304.9 μmol•h−1•cm−2 | 95.9 | -0.20 V | 本工作 |
| [1] |
doi: 10.1021/acs.chemrev.9b00538 pmid: 32501681 |
| [2] |
|
| [3] |
doi: 10.1016/j.jece.2024.112892 |
| [4] |
|
| [5] |
doi: 10.1016/j.nanoen.2024.109499 |
| [6] |
doi: 10.1007/s10311-025-01848-1 |
| [7] |
|
| [8] |
doi: 10.6023/A25030091 |
|
(郭超凡, 苏进展, 郭烈锦, 化学学报, 2025, 83, 716.)
doi: 10.6023/A25030091 |
|
| [9] |
|
| [10] |
doi: 10.1016/j.jechem.2024.10.054 |
| [11] |
doi: 10.1016/j.apenergy.2017.01.071 |
| [12] |
doi: 10.1007/s10311-022-01402-3 |
| [13] |
doi: 10.1021/acs.energyfuels.9b02924 |
| [14] |
|
| [15] |
doi: 10.1016/j.matchemphys.2024.129165 |
| [16] |
|
| [17] |
doi: 10.1016/j.jpba.2022.115102 |
| [18] |
doi: 10.1007/s12649-020-01109-y |
| [19] |
doi: 10.1021/acsomega.0c02673 |
| [20] |
doi: 10.1186/s40643-023-00671-2 |
| [21] |
|
| [22] |
|
| [23] |
doi: 10.1002/cjoc.v42.24 |
| [24] |
|
| [25] |
doi: 10.1016/j.nanoen.2023.108415 |
| [26] |
doi: 10.1002/adfm.v32.35 |
| [27] |
doi: 10.1016/j.jeurceramsoc.2023.09.086 |
| [28] |
doi: 10.1007/s12598-020-01513-2 |
| [29] |
|
| [30] |
doi: 10.1002/cplu.v86.1 |
| [31] |
doi: 10.1039/D2CC02463B |
| [32] |
doi: 10.1016/j.nanoen.2023.108840 |
| [33] |
doi: 10.1039/D2QI00002D |
| [34] |
doi: 10.1021/acs.inorgchem.2c01112 |
| [35] |
doi: 10.1039/D5RA00391A |
| [36] |
doi: 10.3390/catal10030353 |
| [37] |
doi: 10.1039/C9CC90001B |
| [1] | Jianrong Guo, Shuyu Zhang, Junhui He, Shixue Ren. Preparation and Application of Biodegradable Films Based on Biomass [J]. Acta Chimica Sinica, 2024, 82(2): 242-256. |
| [2] | Jiahao Ju, Jilei Xu, Kangjun Wang, Jiahui Huang. One-step Preparation of 5-(Methoxymethyl)-2-furaldehyde from Fructose and Regeneration of Resin Catalyst [J]. Acta Chimica Sinica, 2024, 82(12): 1216-1225. |
| [3] | Lujie Liu, Jian Zhang, Liang Wang, Fengshou Xiao. Heterogeneous Catalysts for Selective Hydrogenolysis of Biomass-derived Polyols★ [J]. Acta Chimica Sinica, 2023, 81(5): 533-547. |
| [4] | Luyao Yu, Zhen Ren, Yusen Yang, Min Wei. Directed Preparation of Biomass-based Polyester Monomers by Catalytic Conversion [J]. Acta Chimica Sinica, 2023, 81(2): 175-190. |
| [5] | Zhaowei Tian, Weimin Da, Lei Wang, Yusen Yang, Min Wei. Structural Design and Research Progress of Heterogeneous Catalysts for the Preparation of Second Generation Biodiesel [J]. Acta Chimica Sinica, 2022, 80(9): 1322-1337. |
| [6] | Heng Shu, Yide-Rigen Bao, Yong Na. Photocatalytic Oxidation of 5-Hydroxymethylfurfural Selectively into 2,5-Diformylfuran with CdS Nanotube [J]. Acta Chimica Sinica, 2022, 80(5): 607-613. |
| [7] | Wang Wenbin, Wen Qunlei, Liu Youwen, Zhai Tianyou. Research Progress of Surface and Interface Chemistry Regulate Two-dimensional Materials for Electrocatalytic Biomass Conversion [J]. Acta Chimica Sinica, 2020, 78(11): 1185-1199. |
| [8] | Li Cui, Zhang Qi, Fu Yao. Transition Metal Catalyzed Deoxydehydration of Alcohols [J]. Acta Chim. Sinica, 2018, 76(7): 501-514. |
| [9] | Zhao Jing, Gong Junwei, Li Yiju, Cheng Kui, Ye Ke, Zhu Kai, Yan Jun, Cao Dianxue, Wang Guiling. Self N-Doped Porous Interconnected Carbon Nanosheets Material for Supercapacitors [J]. Acta Chim. Sinica, 2018, 76(2): 107-112. |
| [10] | Wang Yujue, Hu Min, Wang Yu, Qin Yanhong, Chen Hongyang, Zeng Limin, Lei Jianrong, Huang Xiaofeng, He Lingyan, Zhang Ruiqin, Wu Zhijun. Characterization and Influence Factors of PM2.5 Emitted from Crop Straw Burning [J]. Acta Chim. Sinica, 2016, 74(4): 356-362. |
| [11] | Li Jiang, Huang Yaobing, Guo Qingxiang, Fu Yao. Production of Acetic Acid from Lignocellulosic Biomass in the Presence of Mineral Acid and Oxygen under Hydrothermal Condition [J]. Acta Chim. Sinica, 2014, 72(12): 1223-1227. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||