

Tetraaminopillar[5]arene Dimers: Synthesis, Structure and Properties
Received date: 2022-03-24
Online published: 2022-05-06
Supported by
National Natural Science Foundation of China(21772178); National Natural Science Foundation of China(21971223)
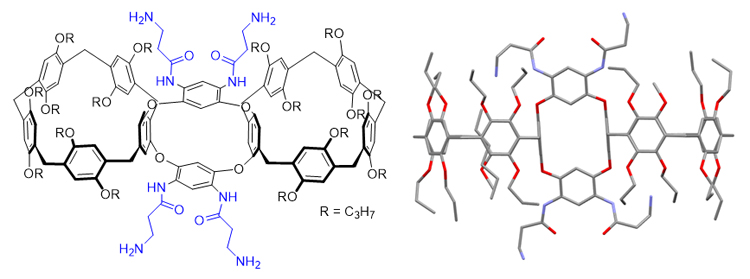
Multi-macrocyclic hosts with fixed conformations provide important platforms for the construction of complicated mechanically inter-locked structures. In order to synthesize rigid multi-macrocycles, a type of oxacalix[4]arene-bridged pillar[5]arene dimers were further derivatized. Firstly, the four NO2 groups in pillar[5]arene dimer 1 were reduced under catalytic hydrogenation conditions using Raney Ni as a catalyst to give tetraamino compound 2. Unfortunately, the aromatic amino groups in compound 2 were unstable in the air and hard for next investigation of molecular recognition. Therefore, further derivatization was used to introduce more stable aliphatic amino groups. After multiple optimization attempts, the “1+4” intermediate product 3 was obtained in a high yield of 72% from reaction of compound 2 and ten equivalents of t-butoxycarbonyl (Boc)-β-alanine 5 in the presence of dicyclohexylcarbodiimide (DCC) at room temperature. The N-Boc protection groups were further removed in the presence of trifluoroacetic acid in CH2Cl2 to afford target compound 4 with four aliphatic amino groups. The structure of 4 was thoroughly confirmed by nuclear magnetic resonance (NMR), high resolution mass spectrometry (HRMS) and X-ray single crystal analysis. X-ray single crystal analysis indicated that the tricyclic host had a shape of binoculars and the conformation was rigid, which could be slightly tuned by different substituents. With a pillar-calix-pillar structure, the host-guest properties of compound 4 were investigated subsequently. Because of its small cavity, oxacalix[4]arene unit played just a role of framework to make two pillar cavities parallel and appropriate for association with guest molecules. The dimers 4 could associate with 1,4-dicyanobutane and form the complex of 2G⸦4 with strong binding ability. The binding constants (K1=3.9×104 L•mol–1, K2=2.4×104 L•mol–1, Ka=9.4×108 L2•mol–2) were determined by ultraviolet and visible (UV-Vis) spectroscopy. The complexation between pillar[5]arene dimer 1 with NO2 group and G was also investigated for comparison. The 1H NMR spectrum and UV-vis spectroscopy studies indicated the binding ability for 1 was not as good as that for 4. This investigation provides a new opportunity for fabricating hierarchical supramolecular systems.
Minggang Wu , Yong Yang , Min Xue . Tetraaminopillar[5]arene Dimers: Synthesis, Structure and Properties[J]. Acta Chimica Sinica, 2022 , 80(8) : 1057 -1060 . DOI: 10.6023/A22030130
| [1] | (a) Diederich, F.; Stang, P. J.; Tykwinski, R. R. Modern Supramolecular Chemistry: Strategies for Macrocycle Synthesis, Wiley-VCH, Berlin, 2008, p. 1. |
| [1] | (b) Steed, J. W.; Atwood, J. L. Supramolecular Chemistry, John Wiley & Sons, Ltd., New York, 2009, p. 1. |
| [1] | (c) Li, J.; Han, Y.; Chen, C. Chin. J. Org. Chem. 2020, 40, 3714. (in Chinese) |
| [1] | (李晶, 韩莹, 陈传峰, 有机化学, 2020, 40, 3714.) |
| [2] | (a) Xue, M.; Yang, Y.; Chi, X.; Yan, X.; Huang, F. Chem. Rev. 2015, 115, 7398. |
| [2] | (b) Xia, D.; Wang, P.; Ji, X.; Khashab, N. M.; Sessler, J. L.; Huang, F. Chem. Rev. 2020, 120, 6070. |
| [2] | (c) Han, X. N.; Han, Y.; Chen, C. F. J. Am. Chem. Soc. 2020, 142, 8262. |
| [3] | (a) Jiang, Y.; Guo, J. B.; Chen, C. F. Chem. Commun. 2010, 46, 5536. |
| [3] | (b) Evans, N. H.; Serpell, C. J.; Beer, P. D. Angew. Chem., Int. Ed. 2011, 50, 2507. |
| [3] | (c) Li, C.; Han, K.; Li, J.; Zhang, H.; Ma, J.; Shu, X.; Chen, Z.; Weng, L.; Jia, X. Org. Lett. 2012, 14, 42. |
| [3] | (d) Hu, W. B.; Xie, C. D.; Hu, W. J.; Zhao, X. L.; Liu, Y. A.; Huo, J. C.; Li, J. S.; Jiang, B.; Wen, K. J. Org. Chem. 2015, 80, 7994. |
| [3] | (e) Zhou, Y.; Jie, K.; Shi, B.; Yao, Y. Chem. Commun. 2015, 51, 11112. |
| [3] | (f) Sun, Y.; Wang, J.; Yao, Y. Chem. Commun. 2017, 53, 165. |
| [3] | (g) Ding, Y.; Alimi, L. O.; Moosa, B.; Maaliki, C.; Jacquemin, J.; Huang, F.; Khashab, N. M. Chem. Sci. 2021, 12, 5315. |
| [4] | (a) Zhang, Z.; Luo, Y.; Chen, J.; Dong, S.; Yu, Y.; Ma, Z.; Huang, F. Angew. Chem., Int. Ed. 2011, 50, 1397. |
| [4] | (b) Song, N.; Chen, D. X.; Qiu, Y. C.; Yang, X. Y.; Xu, B.; Tian, W.; Yang, Y. W. Chem. Commun. 2014, 50, 8231. |
| [4] | (c) Meng, L. B.; Li, D.; Xiong, S.; Hu, X. Y.; Wang, L.; Li, G. Chem. Commun. 2015, 51, 4643. |
| [4] | (d) Huo, B.; Li, B.; Su, H.; Zeng, X.; Xu, K.; Cui, L. Chin. J. Org. Chem. 2019, 39, 1990. (in Chinese) |
| [4] | (霍博超, 李斌, 苏杭, 曾宪强, 徐凯迪, 崔雷, 有机化学, 2019, 39, 1990.) |
| [4] | (e) Xiao, T.; Zhou, L.; Sun, X.-Q.; Huang, F.; Lin, C.; Wang, L. Chin. Chem. Lett. 2020, 31, 1. |
| [5] | (a) Wu, J.; Sun, S.; Feng, X.; Shi, J.; Hu, X. Y.; Wang, L. Chem. Commun. 2014, 50, 9122. |
| [5] | (b) Yue, S. Y.; Zhou, Y. J.; Yao, Y.; Xue, M. Acta Chim. Sinica 2014, 72, 1053. (in Chinese) |
| [5] | (岳诗雨, 周玉娟, 姚勇, 薛敏, 化学学报, 2014, 72, 1053.) |
| [5] | (c) Wang, Q.; Cheng, M.; Zhao, Y.; Wu, L.; Jiang, J.; Wang, L.; Pan, Y. Chem. Commun. 2015, 51, 3623. |
| [5] | (d) Song, N.; Chen, D. X.; Xia, M. C.; Qiu, X. L.; Ma, K.; Xu, B.; Tian, W.; Yang, Y. W. Chem. Commun. 2015, 51, 5526. |
| [5] | (e) Chen, J.; Zhang, Y.; Meng, Z.; Guo, L.; Yuan, X.; Zhang, Y.; Chai, Y.; Sessler, J. L.; Meng, Q.; Li, C. Chem. Sci. 2020, 11, 6275. |
| [6] | (a) Wu, X.; Duan, Q. P.; Ni, M. F.; Hu, X. Y.; Wang, L. Y. Chin. J. Org. Chem. 2014, 34, 437. (in Chinese) |
| [6] | (吴旋, 段群鹏, 倪梦飞, 胡晓玉, 王乐勇, 有机化学, 2014, 34, 437.) |
| [6] | (b) Hu, W. B.; Hu, W. J.; Zhao, X. L.; Liu, Y. A.; Li, J. S.; Jiang, B.; Wen, K. Chem. Commun. 2015, 51, 13882. |
| [6] | (c) Li, S. H.; Zhang, H. Y.; Xu, X.; Liu, Y. Nat. Commun. 2015, 6, 7590. |
| [6] | (d) Zhang, R.; Wang, C.; Sun, J.; Yan, C.; Yao, Y. Chin. J. Org. Chem. 2019, 39, 3483. (in Chinese) |
| [6] | (张润淼, 王陈威, 孙晶, 颜朝国, 姚勇, 有机化学, 2019, 39, 3483.) |
| [6] | (e) Liang, H.; Hua, B.; Xu, F.; Gan, L. S.; Shao, L.; Huang, F. J. Am. Chem. Soc. 2020, 142, 19772. |
| [6] | (f) Dong, J. H.; Li, J. J.; Wang, H.; Liu, B. X.; Peng, B.; Chen, J. Z.; Lin, S. L. Acta Chim. Sinica 2021, 79, 803. (in Chinese) |
| [6] | (董锦辉, 李进杰, 王赫, 刘彬秀, 彭博, 陈健壮, 林绍梁, 化学学报, 2021, 79, 803.) |
| [7] | (a) Li, Z. Y.; Zhang, Y.; Zhang, C. W.; Chen, L. J.; Wang, C.; Tan, H.; Yu, Y.; Li, X.; Yang, H. B. J. Am. Chem. Soc. 2014, 136, 8577. |
| [7] | (b) Chen, J.; Liu, X.; Han, B.; Ding, J.; Zhang, Y.; Lin, Q.; Yao, H.; Wei, T. Chin. J. Org. Chem. 2018, 38, 2741. (in Chinese) |
| [7] | (陈进发, 刘茜, 韩冰冰, 丁金东, 张有明, 林奇, 姚虹, 魏太保, 有机化学, 2018, 38, 2741.) |
| [7] | (c) Li, B.; Wang, B.; Huang, X.; Dai, L.; Cui, L.; Li, J.; Jia, X.; Li, C. Angew. Chem., Int. Ed. 2019, 58, 3885. |
| [7] | (d) Liu, J.; Sun, X. W.; Huang, T. T.; Zhang, Y. M.; Yao, H.; Wei, T. B.; Lin, Q. Chin. J. Chem. 2021, 39, 3421. |
| [8] | (a) Si, W.; Chen, L.; Hu, X. B.; Tang, G.; Chen, Z.; Hou, J. L.; Li, Z. T. Angew. Chem., Int. Ed. 2011, 50, 12564. |
| [8] | (b) Chen, L.; Si, W.; Zhang, L.; Tang, G.; Li, Z. T.; Hou, J. L. J. Am. Chem. Soc. 2013, 135, 2152. |
| [8] | (c) Yan, Z. J.; Wang, D.; Ye, Z.; Fan, T.; Wu, G.; Deng, L.; Yang, L.; Li, B.; Liu, J.; Ma, T.; Dong, C.; Li, Z. T.; Xiao, L.; Wang, Y.; Wang, W.; Hou, J. L. J. Am. Chem. Soc. 2020, 142, 15638. |
| [8] | (d) Yan, Z. J.; Li, Y. W.; Yang, M.; Fu, Y. H.; Wen, R.; Wang, W.; Li, Z. T.; Zhang, Y.; Hou, J. L. J. Am. Chem. Soc. 2021, 143, 11332. |
| [8] | (e) Li, Y. W.; Fu, Y. H.; Hou, J. L. Chin. J. Chem. 2022, 40, 1293. |
| [9] | Wan, K.; Gao, S. C.; Fang, X.; Xu, M. Y.; Yang, Y.; Xue, M. Chem. Commun. 2020, 56, 10155. |
| [10] | Shu, X.; Chen, S.; Li, J.; Chen, Z.; Weng, L.; Jia, X.; Li, C. Chem. Commun. 2012, 48, 2967. |
| [11] | (a) Thordarson, P. Chem. Soc. Rev. 2010, 40, 1305. |
| [11] | (b) Hibbert, D. B.; Thordarson, P. Chem. Commun. 2016, 52, 12792. |
| [12] | Capici, C.; Gattuso, G.; Notti, A.; Parisi, M. F.; Pappalardo, S.; Brancatelli, G.; Geremia, S. J. Org. Chem. 2012, 77, 9668. |
/
| 〈 |
|
〉 |