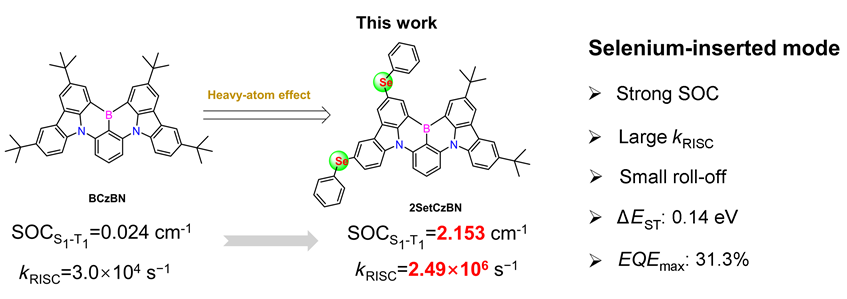
1 引言
2 结果与讨论
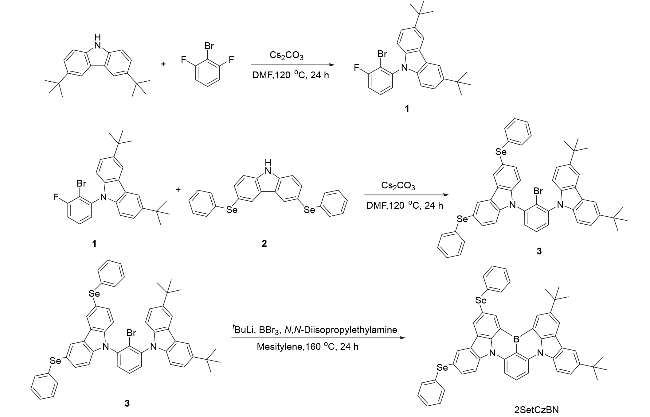
2.1 合成与结构
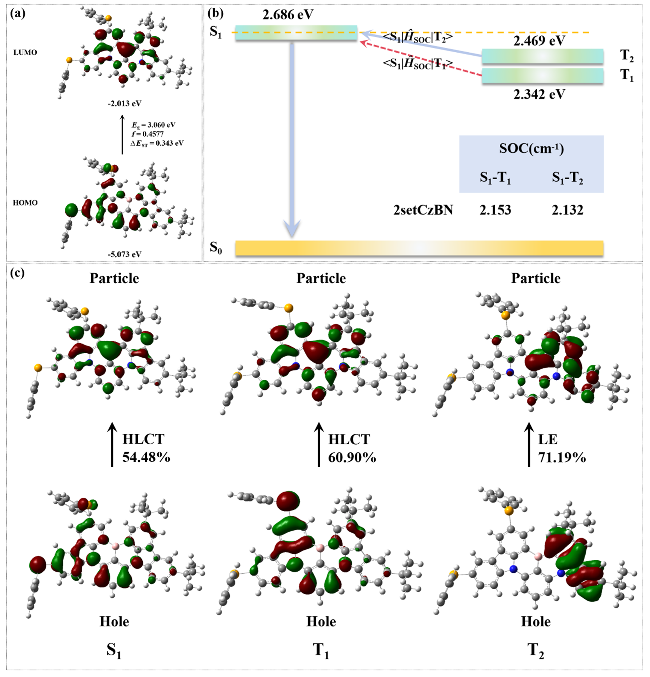
图1 (a) 2SetCzBN分子在溶剂中HOMO和LUMO的能量和分布. (b)激发态的绝热激发能及自旋轨道耦合常数(SOC). (c)单重态和三重态的跃迁属性(箭头右方为LE占比)Figure 1 (a) The energy and distribution of HOMO and LUMO of 2SetCzBN molecules in solvents. (b) The adiabatic excitation energy and spin orbit coupling constant (SOC) of the excited state. (c) The transition properties of singlet and triplet states (The right side of the arrow indicates the proportion of LE) |
2.2 热稳定性与电子轨道能级
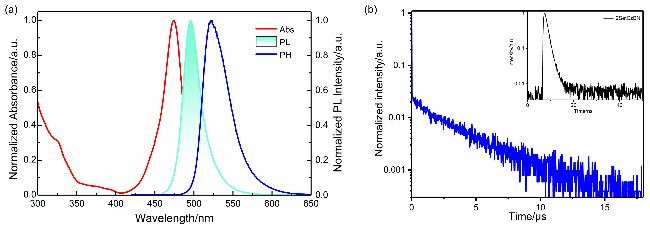
图2 (a) 2SetCzBN在稀释甲苯溶液(10−5 mol/L)中的紫外-可见吸收光谱、荧光光谱(298 K)和磷光光谱(77 K). (b)在300 K真空下, 2SetCzBN薄膜的瞬态光致发光(PL)衰减曲线Figure 2 (a) UV/vis absorption, fluorescence (298 K), and phosphorescence (77 K) spectra of 2SetCzBN in toluene (10−5 mol/L). (b) Transient photoluminescence (PL) decay curve for the 2SetCzBN in film via vacuum processes at 300 K |
表1 2SetCzBN的热力学与电化学数据Table 1 Thermodynamic and electrochemical data of 2SetCzBN |
| 化合物 | Tda/°C | HOMOb/eV | LUMOb/eV | Egc/eV |
|---|---|---|---|---|
| 2SetCzBN | 388 | -5.33 | -2.79 | 2.54 |
a Td: decomposition temperature; b HOMO was calculated from cyclic voltammetry (CV) data, EHOMO=-(Eox-EFc/Fc++4.8) eV; LUMO was calculated from the HOMO and Eg; c Eg was calculated from the corresponding absorption cutoff. |
2.3 光物理性能
表2 2SetCzBN的光物理数据Table 2 Summary of the photophysical properties of 2SetCzBN |
| 化合物 | λabsa/nm | λema/nm | FWHMa/nm | S1b/eV | T1b/eV | ΔESTc/eV |
|---|---|---|---|---|---|---|
| 2SetCzBN | 474 | 496 | 26 | 2.61 | 2.47 | 0.14 |
a Measured in toluene at 298 K; b calculated from the onset of fluorescence at room temperature and phosphorescence spectrum at 77 K; c ΔEST=ES1-ET1. |
表3 2SetCzBN的动力学参数Table 3 Kinetic parameters of 2SetCzBN |
| 化合物 | ФPL/% | Фd/% | τp/ns | τd/μs | Kp/108 s−1 | Kd/105 s−1 | KrS/107 s−1 | KIC/107 s−1 | KISC/108 s−1 | KRISC/106 s−1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2SetCzBN | 70 | 63.98 | 1.30 | 3.27 | 7.69 | 2.14 | 4.63 | 1.98 | 7.03 | 2.49 | |
2.4 电致发光性能
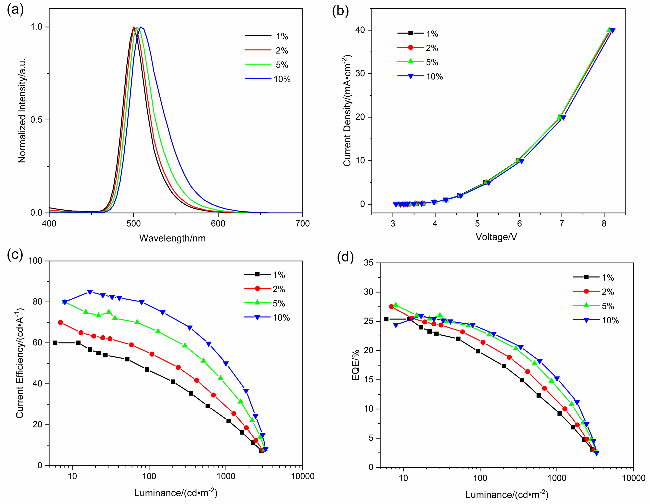
图3 基于2SetCzBN的二元器件电致发光性能. (a) 2SetCzBN的归一化EL光谱曲线. (b) 2SetCzBN的电流密度-电压特性曲线. (c) 2SetCzBN的电流效率(CE)-亮度特性曲线. (d) 2SetCzBN的外量子效率(EQE)-亮度特性曲线Figure 3 EL performance of 2SetCzBN based binary OLED. (a) Normalized EL spectra curves of the 2SetCzBN. (b) Current density-voltage characteristics of 2SetCzBN. (c) Current efficiency (CE)-luminance characteristics of the 2SetCzBN. (d) External quantum efficiency (EQE)-luminance characteristics of the 2SetCzBN |
表4 基于2SetCzBN的二元OLED的EL性能Table 4 EL performance of 2SetCzBN based binary OLED |
| 发光材料 | Dop (w/%) | Vona/V | EQEb/% | CEmaxc/(cd•A−1) | λem/nm | FWHM/nm | ||
|---|---|---|---|---|---|---|---|---|
| Max | @103 cd/m2 | |||||||
| 2SetCzBN | 1 2 5 10 | 3.2 3.2 3.1 3.1 | 25.4 27.5 27.7 25.9 | 9.8 11.7 13.9 15.2 | 60 70 80 85 | 500 500 504 508 | 32 32 36 44 | |
a Turn-on voltage at 1 cd/m2; b maximum external quantum efficiency; c maximum current efficiency. |
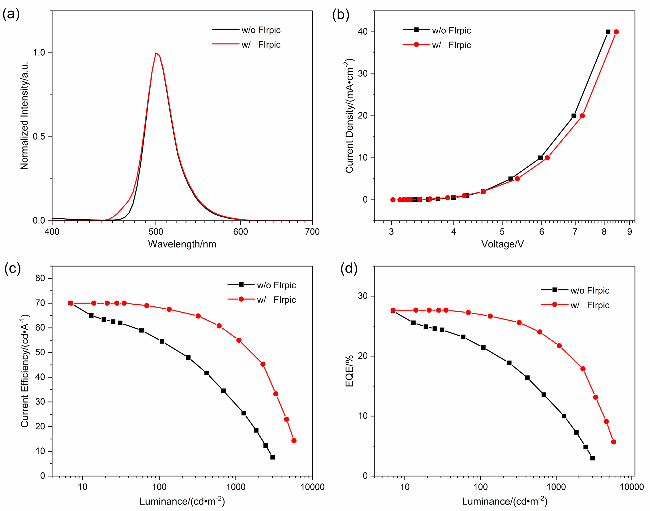
表5 基于2SetCzBN的三元TSF-OLED与二元OLED的EL性能Table 5 EL performance of 2SetCzBN based ternary TSF-OLED and binary OLED |
| 发光材料 | Dop (w/%) | Von/V | EQE/% | CE/(cd•A−1) | λem/nm | FWHM/nm | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Max | @103 cd/m2 | Max | @103 cd/m2 | |||||||
| 2SetCzBN 2SetCzBN:Firpic | 2 2 | 3.2 3.1 | 27.5 27.6 | 11.7 22.2 | 70.0 70.0 | 29.7 56.2 | 500 500 | 32 32 | ||
图4 掺杂浓度为2%时, 基于2SetCzBN的三元敏化器件与未敏化器件电致发光性能. (a) 2SetCzBN的归一化EL光谱曲线. (b) 2SetCzBN的电流密度-电压特性曲线. (c) 2SetCzBN的电流效率(CE)-亮度特性曲线. (d) 2SetCzBN的外量子效率(EQE)-亮度特性曲线Figure 4 EL performance of 2SetCzBN based ternary TSF-OLED and binary OLED with doping concentrations of 2% (w). (a) Normalized EL spectra curves of the 2SetCzBN. (b) Current density-voltage characteristics of 2SetCzBN. (c) Current efficiency (CE)-luminance characteristics of the 2SetCzBN. (d) External quantum efficiency (EQE)-luminance characteristics of the 2SetCzBN |
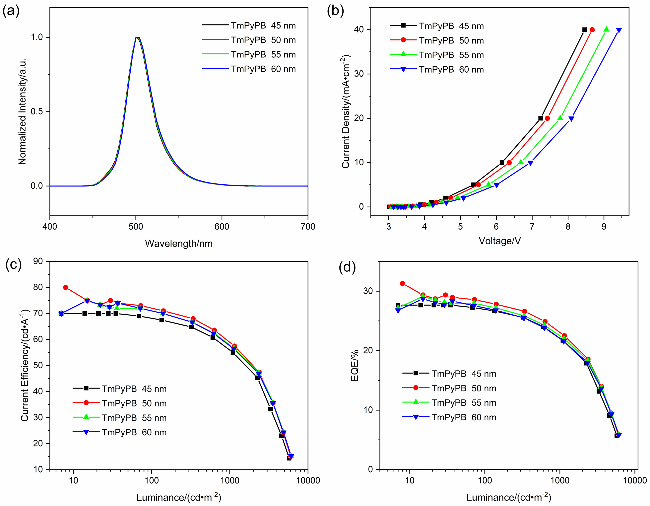
图5 掺杂浓度为2%时, 基于2SetCzBN的三元敏化器件在不同电子传输层厚度下的电致发光性能. (a) 2SetCzBN的归一化EL光谱曲线. (b) 2SetCzBN的电流密度-电压特性曲线. (c) 2SetCzBN的电流效率(CE)-亮度特性曲线. (d) 2SetCzBN的外量子效率(EQE)-亮度特性曲线Figure 5 EL performance of 2SetCzBN based ternary TSF-OLED at different ETL thicknesses with doping concentrations of 2% (w). (a) Normalized EL spectra curves of the 2SetCzBN. (b) Current density-voltage characteristics of 2SetCzBN. (c) Current efficiency (CE)-luminance characteristics of the 2SetCzBN. (d) External quantum efficiency (EQE)-luminance characteristics of the 2SetCzBN |