

Chinese Journal of Organic Chemistry >
Progress of Trifluoromethylation Using Trifluoroacetic Acid and Its Derivatives as CF3-Sources
Received date: 2018-10-25
Revised date: 2018-12-06
Online published: 2018-12-21
Supported by
Project supported by the National Natural Science Foundation of China (Nos.21372176,21672162).
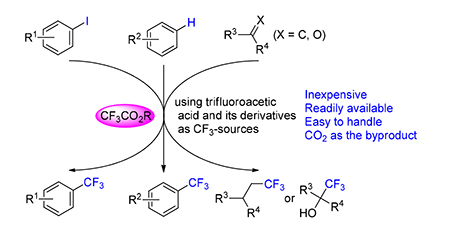
Organic molecules containing trifluoromethyl groups often exhibit unique physical and chemical properties, and have found extensive applications. Trifluoroacetic acid and its derivatives have advantages including low cost, ready availability and high stability. Furthermore, CO2 is the byproduct. Therefore, trifluoroacetic acid and its derivatives are ideal trifluoromethylating reagents, and have great application potentials. The progress of trifluoromethylation reactions using trifluoroacetic acid and its derivatives as CF3-sources is reviewed, including the reactions of C-X (X=Br and I), C-H, and C=X (X=C, O). The mechanisms are also introduced.
Ji Xiaoming , Shi Guangfa , Zhang Yanghui . Progress of Trifluoromethylation Using Trifluoroacetic Acid and Its Derivatives as CF3-Sources[J]. Chinese Journal of Organic Chemistry, 2019 , 39(4) : 929 -939 . DOI: 10.6023/cjoc201810033
[1] (a) Hagmann, W. K. J. Med. Chem. 2008, 51, 4359.
(b) Li, Y.; Wu, Y.; Li, G.-S.; Wang, X.-S. Adv. Synth. Catal. 2014, 356, 1412.
[2] Jeschke, P. ChemBioChem 2004, 5, 570.
[3] (a) O'Hagan, D.; Harper, D. B. J. Fluorine Chem. 1999, 100, 127.
(b) Vaillancourt, F. H.; Yeh, E.; Vosburg, D. A.; GarneauTsodikova, S.; Walsh, C. T. Chem. Rev. 2006, 106, 3364.
[4] (a) Müller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
(b) Schlosser, M. Angew. Chem., Int. Ed. 2006, 45, 5432.
[5] Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
[6] (a) Ismail, F. M. D. J. Fluorine Chem. 2002, 118, 27.
(b) Bégué, J.-P.; Bonnet-Delpon, D. J. Fluorine Chem. 2006, 127, 992.
(c) Petrov, V. A. Fluorinated Heterocyclic Compounds:Synthesis, Chemistry, and Applications, John Wiley & Sons, Inc, Hoboken, NJ, USA, 2009.
(d) Ojima, I. Fluorine in Medicinal Chemistry and Chemical Biology, John Wiley & Sons, Ltd, Chichester, UK., 2009.
(e) Zhu, W.; Wang, J.; Wang, S. J. Fluorine Chem. 2014, 167, 37.
(f) Zhang, J.; Jin, C.; Zhang, Y. Chin. J. Org. Chem. 2014, 34, 662(in Chinese). (张霁, 金传飞, 张英俊, 有机化学, 2014, 34, 662.)
[7] (a) Penning, T. D.; Talley, J. J.; Bertenshaw, S.R.; Carter, J. S.; Collins, P. W.; Docter, S.; Graneto, M. J.; Lee, L. F.; Malecha, J. W.; Miyashiro, J. M.; Rogers, R. S.; Rogier, D. J.; Yu, S. S.; Anderson, G. D.; Burton, E. G.; Cogburn, J. N.; Gregory, S. A.; Koboldt, C. M.; Perkins, W. E.; Seibert, K. A.; Veenhuizen, W. Y.; Zhang, Y.; Isakson, P.C. J. Med. Chem. 1997, 40, 1347.
(b) Chakraborti, A. K.; Garg, S. K.; Kumar, R.; Motiwala, H. F.; Jadhavar, P. S. Curr. Med. Chem. 2010, 17, 1563.
[8] (a) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
(b) Prakash, S. G. K.; Yudin, A. K. Chem. Rev. 1997, 97, 757.
[9] (a) Ye, Y.; Ball, N. D.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2010, 132, 14682.
(b) Wang, X.; Truesdale, L.; Yu, J.-Q. J. Am. Chem. Soc. 2010, 132, 3648.
(c) Zhang, X.; Dai, H.; Wasa, M.; Yu, J.-Q. J. Am. Chem. Soc. 2012, 134, 11948.
(d) Mu, X.; Chen, S.; Zhen, X.; Liu, G. Chem.-Eur. J. 2011, 17, 6039.
(e) Zhang, L.-S.; Chen, K.; Chen, G.; Li, B.-J.; Luo, S.; Guo, Q.-Y.; Wei, J.-B.; Shi, Z.-J. Org. Lett. 2013, 15, 10.
(f) Miura, M.; Feng, C.-G.; Ma, S.; Yu, J.-Q. Org. Lett. 2013, 15, 5258.
(g) Culkin, D. A.; Hartwig, J. F. Organometallics 2004, 23, 3398.
(h) Grushin, V. V. Acc. Chem. Res. 2010, 43, 160.
(i) Hughes, R. P.; Meyer, M. A.; Tawa, M. D.; Ward, A. J.; Williamson, A.; Rheingold, A. L.; Zakharov, L. N. Inorg. Chem. 2004, 43, 747.
(j) Ball, N. D.; Gary, J. B.; Ye, Y.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2011, 133, 7577.
(k) Ball, N. D.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2010, 132, 2878.
(l) Cho, E. J.; Senecal, T. D.; Kinzel, T.; Zhang, Y.; Watson, D. A.; Buchwald, S. L. Science 2010, 328, 1679.
[10] (a) Shimizu, R.; Egami, H.; Nagi, T.; Chae, J.; Hamashima, Y.; Sodeoka, M. Tetrahedron Lett. 2011, 51, 5947.
(b) Zhang, C.-P.; Wang, Z.-L.; Chen, Q.-Y.; Zhang, C.-T.; Gu, Y.-C.; Xiao, J.-C. Angew. Chem., Int. Ed. 2011, 50, 1896.
(c) Liu, T.; Shen, Q. Org. Lett. 2016, 13, 2342.
(d) Chu, L.; Qing, F.-L. J. Am. Chem. Soc. 2012, 134, 1298.
(e) Cai, S.; Chen, C.; Sun, Z.; Xi, C. Chem. Commun. 2013, 49, 4552.
(f) He, Z.; Tan, P.; Hu, J. Org. Lett. 2016, 18, 72.
(g) Li, X.; Zhao, J.; Zhang, L.; Hu, M.; Wang, L.; Hu J. Org. Lett. 2015, 17, 298.
(h) Gao, X.; Xiao, Y.-L.; Wan, X.; Zhang X. Angew. Chem., Int. Ed. 2018, 57, 3187.
(i) Xu, X.; Chen, H.; He, J.; Xu, H. Chin. J. Chem. 2017, 35, 1665.
[11] (a) Hafner, A.; Stefan, B. Angew. Chem., Int. Ed. 2012, 51, 3713.
(b) Ye, Y.; Lee, S. H.; Sanford, M. S. Org. Lett. 2011, 13, 5464.
(c) Loy, R. N.; Sanford, M. S. Org. Lett. 2011, 13, 2548.
(d) Seo, S.; Taylor, J. B.; Greaney, M. F. Chem. Commun. 2013, 49, 6385.
(e) Liu, Y.-R.; Tu, H.-Y.; Zhang, X.-G. Synthesis 2015, 47, 3460.
[12] Kino, T.; Nagase, Y.; Ohtsuka, Y.; Yamamoto, K.; Uraguchi, D.; Tokuhisa, K.; Yamakawa, T. J. Fluorine Chem. 2010, 131, 98.
[13] (a) Nagib, D. A.; MacMillan, D. W. C. Nature 2011, 480, 224.
(b) Iqbal, N.; Choi, S.; Ko, E.; Cho, E. J. Tetrahedron Lett. 2012, 53, 2005.
(c) Kamigata, N.; Fukushima, T.; Yoshida, M. Chem. Lett. 1990, 649.
(d) Kamigata, N.; Ohtsuka, T.; Fukushima, T.; Yoshida, M.; Shimizu, T. J. Chem. Soc. Perkin Trans. 11994, 1339.
(e) Xie, J.; Yuan, X.; Abdukader, A.; Zhu, C.; Ma, J. Org. Lett. 2014, 16, 1768.
[14] Mejía, E.; Togni, A. ACS Catal. 2012, 2, 521.
[15] (a) Studer, A. Angew. Chem., Int. Ed. 2012, 51, 8950.
(b) Parsons, A. T.; Buchwald, S. L. Nature 2011, 480, 184.
[16] (a) Ji, Y.; Brueckl, T.; Baxter, R. D.; Fujiwara, Y.; Seiple, I. B.; Su, S.; Blackmond, D. G.; Baran, P. S. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 14411.
(b) Fujiwara, Y.; Dixon, J. A.; O'Hara, F.; Funder, E. D.; Dixon, D. D.; Rodriguez, R. A.; Baxter, R. D.; Herle, B.; Sach, N.; Collins, M. R.; Ishihara, Y.; Baran, P. S. Nature 2012, 492, 95.
(c) Wu, X.; Chu,L.; Qing, F.-L. Tetrahedron Lett. 2013, 54, 249.
(d) Yang, Y.-D.; Iwamoto, K.; Tokunaga, E.; Shibata, N. Chem. Commun. 2013, 49, 5510.
(e) Fennewald, J. C.; Lipshutz, B. H. Green Chem. 2014, 16, 1097.
(f) Cui, L.; Matusaki, Y.; Tada, N.; Miura, T.; Uno, B.; Itoh, A. Adv. Synth. Catal. 2013, 355, 2203.
[17] (a) Umemoto, T. Chem. Rev. 1996, 96, 1757.
(b) Prakash, G. K. S.; Hu, J. Acc. Chem. Res. 2007, 40, 921.
(c) Prakash, G. K. S.; Yudin, A. K. Chem. Rev. 1997, 97, 757.
(d) Shibata, N.; Matsnev, A.; Cahard, D. Beilstein J. Org. Chem. 2010, 6, 65.
(e) Chu, L.; Qing, F.-L. Acc. Chem. Res. 2014, 47, 1513.
[18] Shi, G.; Shao, C.; Pan, S.; Yu, J.; Zhang, Y. Org. Lett. 2015, 17, 38.
[19] (a) Lopez, S. E.; Salazar, J. J. Fluorine Chem. 2013, 156, 73.
(b) Rui, S.; Lei, L. Sci. China Chem. 2011, 54, 1670.
(c) Rodríguez, N.; Goossen, L. J. Chem. Soc. Rev. 2011, 40, 5030.
[20] Wang, J.; Sanchez-Rosello, M.; Acena, J. L.; del Pozo, C.; Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432.
[21] Matsui, K.; Tobita, E.; Ando, M.; Kondo, K. Chem. Lett. 1981, 12, 1719.
[22] Hünig, S.; Bau, R.; Kemmer, M.; Meixner, H.; Metzenthin, T.; Peters, K.; Sinzger, K.; Gulbis, J. Eur. J. Org. Chem. 1998, 2, 335.
[23] Austin, N. E.; Avenell, K. Y.; Boyfield, I.; Branch, C. L.; Hadley, M. S.; Jeffrey, P.; Johnson, C. N.; Macdonald, G. J.; Nash, D. J.; Riley, G. J.; Smith, A. B.; Stemp, G.; Thewlis, K. M.; Vong, A. K. K.; Wood, M. D. Bioorg. Med. Chem. Lett. 2001, 11, 685.
[24] Langlois, B. R.; Roques, N. J. Fluorine Chem. 2007, 128. 1318.
[25] McReynolds, K. A.; Lewis, R. S.; Ackerman, L. K. G.; Dubinina, G. G.; Brennessel, W. W.; Vicic, D. A. J. Fluorine Chem. 2010, 131, 1108.
[26] Li, Y.; Chen, T.; Wang, H.; Zhang, R.; Jin, K.; Wang, X.; Duan, C. Synlett 2011, 1713.
[27] Schareina, T.; Wu, X.-F.; Zapf, A.; Cotte, A.; Gotta, M.; Beller, M. Top Catal. 2012, 55, 426.
[28] Chen, M.; Buchwald, S. L. Angew. Chem., Int. Ed. 2013, 52, 11628.
[29] Maleckis, A.; Sanford, M. S. Organometallics 2014, 33, 2653.
[30] Lin, X.; Hou, C.; Li, H.; Weng, Z. Chem.-Eur. J. 2016, 22, 2075.
[31] Torikai, K.; Koga, R.; Liu, X.; Umehara, K.; Kitano, T.; Watanabe, K.; Oishi, T.; Noguchi, H.; Shimohigashi, Y. Bioorg. Med. Chem. 2017, 25, 5216.
[32] (a) Giri, R.; Shi, B.-F.; Engle, K. M.; Maugel, N.; Yu, J.-Q. Chem. Soc. Rev. 2009, 38, 3242.
(b) Sun, C.-L.; Li, B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293.
(c) Liu, C.; Yuan, J.; Gao, M.; Tang, S.; Li, W.; Shi, R.; Lei, A. Chem. Rev. 2015, 115, 12138.
(d) Song, G.; Li, X. Acc. Chem. Res. 2015, 48, 1007.
(e) Guo, X.-X.; Gu, D.-W.; Wu, Z.; Zhang, W. Chem. Rev. 2015, 115, 1622.
(f) Zheng, C.; You, S.-L. RSC Adv. 2014, 4, 6173.
(g) Ackermann, L. Chem. Rev. 2011, 111, 1315.
(h) Neufeldt, S. R.; Sanford, M. S. Acc. Chem. Res. 2012, 45, 936.
(i) Zhang, Y.; Shi, G.; Yu, J.-Q. Carbon-Carbon σ-Bond Formation via C-H Bond Functionalization in Comprehensive Organic Synthesis, 2nd ed., Vol. 3, Eds.:Molander, G.; Knochel, P., Elsevier, Oxford, 2014, pp. 1101~1209.
[33] Grinberg, V. A.; Polishchuk, V. R.; German, L. S.; Kanevskii, L. S.; Vassiliev, Y. B. Izv. Akad. Nauk SSSR, Ser. Khim. 1978, 3, 673.
[34] Sawada, H.; Nakayama, M.; Yoshida, M.; Yoshida, T.; Kamigata, N. J. Fluorine Chem. 1990, 46, 423.
[35] Lai, C.; Mallouk, T. E. J. Chem. Soc., Chem. Commun. 1993, 17, 1359.
[36] Matsui, M.; Kondoh, S.; Shibata, K.; Muramatsu, H. Bull. Chem. Soc. Jpn. 1995, 68, 1042.
[37] Tanabe, Y.; Matsuo, N.; Ohno, N. J. Org. Chem. 1988, 53, 4583.
[38] Depecker, C.; Marzouk, H.; Trevin, S.; Devynck, J. New J. Chem. 1999, 23, 739.
[39] Beatty, J. W.; Douglas, J. J.; Cole, K. P.; Stephenson, C. R. J. Nat. Commun. 2015, 6, 7919.
[40] Zhong, S.; Hafner, A.; Hussal, C.; Nieger, M.; Brase, S. RSC Adv. 2015, 5, 6255.
[41] Lin, J.; Li, Z.; Kan, J.; Huang, S.; Su, W.; Li, Y. Nat. Commun. 2017, 8, 1.
[42] Yang, B.; Yu, D.; Xu, X.-H.; Qing, F.-L. ACS Catal. 2018, 8, 2839.
[43] (a) Parsons, A. T.; Buchwald, S. L. Angew. Chem., Int. Ed. 2011, 50, 9120.
(b) Xu, J.; Fu, Y.; Luo, D.-F.; Jiang, Y.-Y.; Xiao, B.; Liu, Z.-J.; Gong, T.-J.; Liu, L. J. Am. Chem. Soc. 2011, 133, 15300.
(c) Wang, X.; Ye, Y.; Zhang, S.; Feng, J.; Xu, Y.; Zhang, Y.; Wang, J. J. Am. Chem. Soc. 2011, 133, 16410.
(d) Liu, X.; Xiong, F.; Huang, X.; Xu, L.; Li, P.; Wu, X. Angew. Chem., Int. Ed. 2013, 52, 6962.
[44] Brookes, C. J.; Coe, P. L.; Owen, D. M.; Pedler, A. E.; Tatlow, J. C. J. Chem. Soc., Chem. Commun. 1974, 9, 323.
[45] Barton, D. H. R.; Lacher, B.; Zard, S. Z. Tetrahedron 1986, 42, 2325.
[46] Jablonski, L.; Joubert, J.; Billard, T.; Langlois, B. R. Synlett 2003, 230.
[47] Chang, Y.; Cai, C. J. Fluorine Chem. 2005, 126, 937.
[48] Arai, K.; Watts, K.; Wirth, T. ChemistryOpen 2014, 3, 23.
[49] Kawamura, S.; Sodeoka, M. Angew. Chem., Int. Ed. 2016, 55, 8740.
[50] Valverde, E.; Kawamura, S.; Sekine, D.; Sodeoka, M. Chem. Sci. 2018, 9, 7115.
[51] Zhang, Y.; Li, Z.; Song, H.; Wang, B. Chin. J. Chem. 2018, 36, 635.
/
| 〈 |
|
〉 |