

Chinese Journal of Organic Chemistry >
Research Progress on Asymmetric Synthesis of Flavanones
Received date: 2021-09-21
Revised date: 2021-10-22
Online published: 2021-11-17
Supported by
Joint Special Fund Project for Basic Research of Local Undergraduate Universities (Part of) in Yunnan Province(2019FH001(-087))
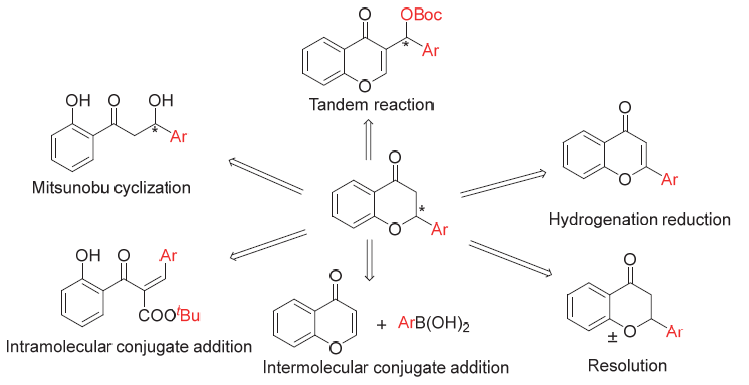
Flavanones and their derivatives are important bioactivenatural products. The skeleton structure exists in many complex natural products and has a wide range of biological activities. The research on flavanones is getting more and more attention. Chiral flavanone itself is an important chemical substance in plants and has potential medicinal value, but its content is small. Many chemists are committed to solve the problem through synthetic methods, which promotes the preparation of enantiomerically enriched flavanones. The preparation process has certain limitations, but for some complex structures of flavanone compounds it is still difficult to achieve, This is going to be a problem to overcome in the future. The asymmetric synthesis methods of flavanones are reviewed in recent years. The synthesis methods include carbonyl reduction and chiral resolution of raceme, formation of carbon-carbon bonds, formation of carbon-heterobonds and other types of synthesis methods.
Key words: flavanones; asymmetric synthesis; review
Lihua Wang , Xushun Gong , Ting Lei , Shizhi Jiang . Research Progress on Asymmetric Synthesis of Flavanones[J]. Chinese Journal of Organic Chemistry, 2022 , 42(3) : 758 -769 . DOI: 10.6023/cjoc202109030
| [1] | Veitch, N. C.; Grayer, R. J. Nat. Prod. Rep. 2011, 28, 1626. |
| [2] | Amado, N. G.; Fonseca, B. F.; Cerqueira, D. M.; Neto, V. M.; Abreu, J. G. Life Sci. 2011, 89(15-16), 545. |
| [3] | Crozier, A.; Jaganath, I. B.; Clifford, M. N. Nat. Prod. Rep. 2009, 26(8), 1001. |
| [4] | Nicolaou, K. C.; Pfefferkorn, J. A.; Roecker, A. J.; Cao, G. Q.; Barluenga, S.; Mitchell, H. J. J. Am. Chem. Soc. 2000, 122, 9939. |
| [5] | Marais, J. P. J.; Ferreira, D.; Slade, D. Phytochem. Lett. 2005, 66(18), 2145. |
| [6] | Gladiali, S.; Alberico, E. Chem. Soc. Rev. 2006, 35(3), 226. |
| [7] | Wang, D. S.; Chen, Q. A.; Lu, S. M.; Zhou, Y. G. Chem. Rev. 2011, 112(4), 2557. |
| [8] | Zhao, D. B.; Beiring, B.; Glorius, F. Angew. Chem., Int. Ed. 2013, 52(32), 8454. |
| [9] | Huddleston, R. R.; Krische, M. J. Org. Lett. 2003, 5(7), 1143. |
| [10] | Corey, E. J.; Mitra, R. B. J. Am. Chem. Soc. 1962, 84(15), 2938. |
| [11] | Rákosi, M.; Tökés, A. L.; Bognár, R. Tetrahedron Lett. 1970, 11(26), 2305. |
| [12] | Pellissier, H. Adv. Synth. Catal. 2011, 353(10), 1613. |
| [13] | Lemke, M. K.; Schwab, P.; Fischer, P.; Tischer, S.; Witt, M.; Noehringer, L.; Rogachev, V.; Jäger, A.; Kataeva, O.; Fröhlich, R.; Metz, P. Angew. Chem., Int. Ed. 2013, 52(44), 11651. |
| [14] | Patel, R.; Hanson, R.; Goswami, A. J. Ind. Microbiol. Biotechnol. 2003, 30, 252. |
| [15] | Lzumi, T.; Hino, T.; Kasahara, A. J. Chem. Soc., erkin Trans. 1 1992, (10), 1265. |
| [16] | Lzumi, T.; Suenaga, K. J. Heterocycl. Chem. 1997, 34, 1535. |
| [17] | Kasashi, K.; Kakuda, H.; Goto, M.; Kawabata, S.; Kometani, T. Tetrahedron: Asymmetry. 2003, 14(11), 1529. |
| [18] | Richard, G.; Button.; Peter, J.; Taylo J. Chem. Soc., erkin Trans. 2 1992, 1571. |
| [19] | Nibbs, A. E.; Scheidt, K. A. Eur. J. Inorg. 2012, 3, 449. |
| [20] | Solladié, G.; Gehrold, N.; Maignan, J. Tetrahedron: Asymmetry 1999, 10(14), 2739. |
| [21] | Chen, J.; Chen, J.; Lang, F.; Zhang, X. Y.; Cun, L. F.; Zhu, J.; Deng, J. G.; Liao, J. J. Am. Chem. Soc. 2010, 41(33), 4552. |
| [22] | Shintani, R.; Tsutsumi, Y.; Nagaosa, M.; Nishimura, T.; Hayashi, T. J. Am. Chem. Soc. 2009, 131(38), 13588. |
| [23] | Takaya, Y.; Ogasawara, M.; Hayashi, T.; Sakai, M.; Miyaura, N. J. Am. Chem. Soc. 1998, 120(22), 5579. |
| [24] | Shintani, R.; Takeda, M.; Nishimura, T.; Hayashi, T. Angew. Chem., Int. Ed. 2010, 49(23), 3969. |
| [25] | Han, F. Z.; Chen, G. H.; Zhang, X. Y.; Liao, J. Eur. J. Org. Chem. 2011, 16, 2928. |
| [26] | Korenaga, T.; Hayashi, K.; Akaki, Y.; Maenishi, R.; Sakai, T. Org. Lett. 2011, 13(8), 2022. |
| [27] | Cisak, A.; Mielczarek, C. J. Chem. Soc., erkin Trans 2. 1992, 1603. |
| [28] | Mino, T.; Hashimoto, M.; Uehara, K.; Naruse, Y.; Kobayashi, S.; Sakamoto, M.; Fujita, T. Tetrahedron Lett. 2012, 53(34), 4562. |
| [29] | He, Q. J.; So, C. M.; Bian, Z. X.; Hayashi, T.; Wang, J. Chem. Asian J. 2015, 10(3), 540. |
| [30] | Holder, J. C.; Marziale, A. N.; Gatti, M.; Mao, B.; Stoltz, B. M. Chemistry 2013, 19(1), 74. |
| [31] | Tamura, M.; Ogata, H.; Ishida, Y.; Takahashi, Y. Tetrahedron Lett. 2017, 58(40), 3808. |
| [32] | Meng, L.; Wang, J. ChemInform 2016, 47(5), 656. |
| [33] | Biddle, M. M.; Lin, M.; Scheidt, K. A. J. Am. Chem. Soc. 2007, 129(13), 3830. |
| [34] | Farmer, R. L.; Biddle, M. M.; Nibbs, A. E.; Huang, X.; Bergan, R. C.; Scheidt, K. A. ACS Med. Chem. Lett. 2010, 1(8), 400. |
| [35] | Wang, L. J.; Liu, X. H.; Dong, Z. H.; Fu, X.; Feng, X. M. Angew. Chem., Int. Ed. 2008, 47(45), 8670. |
| [36] | Feng, Z.; Zeng, M.; Xu, Q. L.; You, S. L. Sci. Bull. 2010, 55(17), 1723. |
| [37] | Wang, H. F.; Xiao, H.; Wang, X. W.; Zhao, G. Tetrahedron 2011, 67(30), 5389. |
| [38] | Dittmer, C.; Raabe, G. Hintermann Eur. J. Inorg 2007, 5886. |
| [39] | Miles, C. O.; Main, L.; Nicholson, B. K. Aust. J. Chem. 1989, 42, 1103. |
| [40] | Hintermann, L.; Dittmer, C. Eur. J. Inorg. 2012, 28, 5573. |
| [41] | Zhang, Y. L.; Wang, Y. Q. Tetrahedron Lett. 2014, 55(21), 3255. |
| [42] | Nising, C. F.; Bräse, S. Chem. Soc. Rev. 2012, 41, 988. |
| [43] | Reyes, E.; Talavera, G.; Vicario, J. L.; Badía, D.; Carrillo, L. Angew. Chem., nt. Ed. 2009, 48, 5701. |
| [44] | McDonald, B. R.; Nibbs, A. E.; Scheidt, K. A. Org. Lett. 2015, 17(1), 98. |
| [45] | Sharma, A.; Singh, M.; Rai, N. N.; Sawant, D. J. Org. Chem. 2013, 9, 1235. |
| [46] | Singh, A. K.; Mangawa, S. K.; Kumar, A.; Dixit, A. K.; Awasthi, S. K. ChemistrySelect 2017, 2(34), 11160. |
| [47] | Hodgetts, K. J. Tetrahedron Lett. 2001, 42, 3763. |
| [48] | Noda, Y.; Watanabe, M. Helv. Chim. Acta 2002, 85(10), 3473. |
| [49] | Wang, H. F.; Cui, H. F.; Chai, Z.; Li, P.; Zheng, C. W.; Yang, Y. Q.; Zhao, G. Chemistry 2009, 15(48), 13299. |
| [50] | Zhong, N. J; Liu, L.; Wang, D.; Chen, Y. J. Chem. Commun. (Camb.) 2013, 49(35), 3697. |
| [51] | Wen, G. F.; Su, Y. P.; Zhang, G. X.; Lin, Q. Q.; Zhu, Y. J.; Zhang, Q. Q.; Fang, X. Q. Org. Lett. 2016, 18(16), 3980. |
| [52] | Reddi, Y.; Sunoj, R. B. ACS Catal. 2015, 5(3), 1596. |
/
| 〈 |
|
〉 |