

Chinese Journal of Organic Chemistry >
Synthesis and Antiproliferative Activity of 2,4,5,6-Tetrasubstituted Pyrimidine Derivatives Containing Anisole
Received date: 2022-01-28
Revised date: 2022-03-08
Online published: 2022-03-22
Supported by
National Natural Science Foundation of China(U21A20416); Opening Fund from State Key Laboratory of Esophageal Cancer Prevention & Treatment(K2020000X)
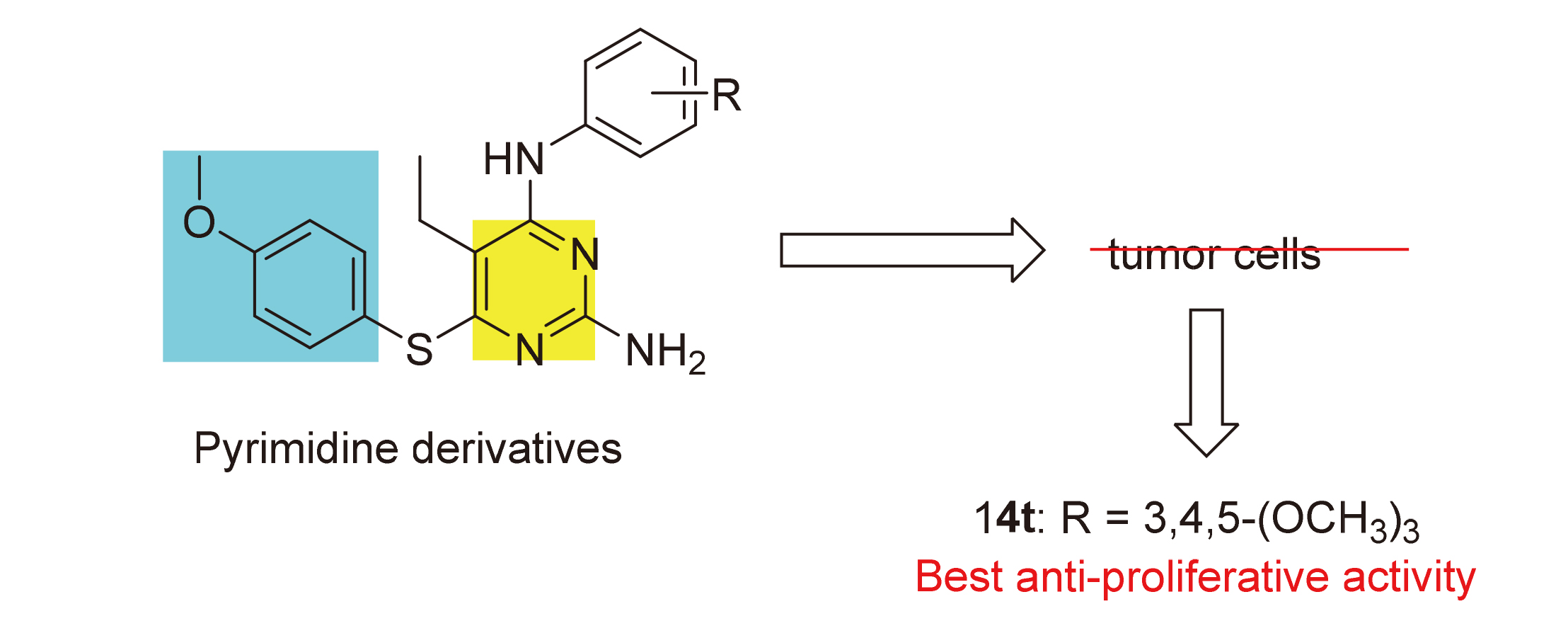
In order to find efficient new anti-tumor drugs, a series of 2,4,5,6-tetrasubstituted pyrimidine derivatives containing anisole structure were designed, synthesized and evaluated for antiproliferative activity against four human cancer cell lines of PC-3, MGC-803, MCF-7 and HGC-27 using methyl thiazolyl tetrazolium (MTT) assay. Most of compounds displayed moderate to excellent antiproliferative activity against the four human tumor cell lines, among which the compound 5-ethyl-6- ((4-methoxyphenyl)thio)-N4-(3,4,5-trimethoxyphenyl)pyrimidine-2,4-diamine (14t) showed the best antiproliferative activity on HGC-27 cells, with IC50 value of (0.98±0.12) μmol•L–1, the antiproliferative activity is significantly better than that of the positive control 5-fluorouracil. Further research on anti-tumor mechanism showed that compound 14t could significantly inhibit the colony formation and migration of HGC-27 cells and induce apoptosis. At the same time, compound 14t can down-regulate the expression of anti-apoptotic protein Bcl-2 and up-regulate the expression of pro-apoptotic proteins Bax and P53.
Key words: pyrimidine; anisole; synthesis; antiproliferative activity
Chao Gao , Xiaojie Si , Lingling Chi , Hao Wang , Honglin Dai , Limin Liu , Zhengjie Wang , Yang Zhang , Tao Wang , Yaochuan Zhou , Jiaxin Zheng , Yu Ke , Hongmin Liu , Qiurong Zhang . Synthesis and Antiproliferative Activity of 2,4,5,6-Tetrasubstituted Pyrimidine Derivatives Containing Anisole[J]. Chinese Journal of Organic Chemistry, 2022 , 42(6) : 1677 -1686 . DOI: 10.6023/cjoc202201048
| [1] | Sung, H.; Ferlay, J.; Siegel, R. L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. CA Cancer J Clin. 2021, 71, 209. |
| [2] | Mao, J. J.; Pillai, G. G.; Andrade, C. J.; Ligibel, J. A.; Basu, P.; Cohen, L.; Khan, I. A.; Mustian, K. M.; Puthiyedath, R.; Dhiman, K. S.; Lao, L.; Ghelman, R.; Caceres Guido, P.; Lopez, G.; Gallego-Perez, D. F.; Salicrup, L. A. CA Cancer J Clin. 2022, 72, 144. |
| [3] | Tilaoui, M.; Ait Mouse, H.; Zyad, A. Front Pharmacol. 2021, 12, 719694. |
| [4] | Gu, X.; Huang, Z.; Ren, Z.; Tang, X.; Xue, R.; Luo, X.; Peng, S.; Peng, H.; Lu, B.; Tian, J.; Zhang, Y. J. Med. Chem. 2017, 60, 928. |
| [5] | Meng, Y.; Li, E.; Zhang, Y.; Liu, S.; Bao, C.; Yang, P.; Zhang, L.; Zhang, D.; Wang, J.; Chen, Y.; Li, N.; Xin, J.; Zhao, P.; Ke, Y.; Zhang, Q.; Liu, H. Chin. J. Org. Chem. 2019, 39, 2541. (in Chinese) |
| [5] | ( 孟娅琪, 李二冬, 张洋, 刘栓, 包崇男, 杨鹏, 张路野, 张丹青, 王继宽, 陈雅欣, 栗娜, 辛景超, 赵培荣, 可钰, 张秋荣, 刘宏民, 有机化学, 2019, 39, 2541.) |
| [6] | Ma, L. Y.; Zheng, Y. C.; Wang, S. Q.; Wang, B.; Wang, Z. R.; Pang, L. P.; Zhang, M.; Wang, J. W.; Ding, L.; Li, J.; Wang, C.; Hu, B.; Liu, Y.; Zhang, X. D.; Wang, J. J.; Wang, Z. J.; Zhao, W.; Liu, H. M. J. Med. Chem. 2015, 58, 1705. |
| [7] | Guillemont, J.; Pasquier, E.; Palandjian, P.; Vernier, D.; Gaurrand, S.; Lewi, P. J.; Heeres, J.; de Jonge, M. R.; Koymans, L. M.; Daeyaert, F. F.; Vinkers, M. H.; Arnold, E.; Das, K.; Pauwels, R.; Andries, K.; de Bethune, M. P.; Bettens, E.; Hertogs, K.; Wigerinck, P.; Timmerman, P.; Janssen, P. A. J. Med. Chem. 2005, 48, 2072. |
| [8] | Wang, L.; Tang, J.; Huber, A. D.; Casey, M. C.; Kirby, K. A.; Wilson, D. J.; Kankanala, J.; Xie, J.; Parniak, M. A.; Sarafianos, S. G.; Wang, Z. Eur. J. Med. Chem. 2018, 156, 652. |
| [9] | Kumar, N.; Chauhan, A.; Drabu, S. Biomed. Pharmacother. 2011, 65, 375. |
| [10] | Tale, R. H.; Rodge, A. H.; Hatnapure, G. D.; Keche, A. P. Bioorg. Med. Chem. Lett. 2011, 21, 4648. |
| [11] | Fouda, A. M.; Abbas, H. S.; Ahmed, E. H.; Shati, A. A.; Alfaifi, M. Y.; Elbehairi, S. E. I. Molecules 2019, 24, 1080. |
| [12] | Shaaban, O. G.; Issa, D. A. E.; El-Tombary, A. A.; Abd El Wahab, S. M.; Abdel Wahab, A. E.; Abdelwahab, I. A. Bioorg. Chem. 2019, 88, 102934. |
| [13] | Cassidy, J.; Clarke, S.; Diaz-Rubio, E.; Scheithauer, W.; Figer, A.; Wong, R.; Koski, S.; Rittweger, K.; Gilberg, F.; Saltz, L. Br. J. Cancer 2011, 105, 58. |
| [14] | Passaro, A.; Guerini-Rocco, E.; Pochesci, A.; Vacirca, D.; Spitaleri, G.; Catania, C. M.; Rappa, A.; Barberis, M.; de Marinis, F. Pharmacol. Res. 2017, 117, 406. |
| [15] | Finlay, M. R.; Anderton, M.; Ashton, S.; Ballard, P.; Bethel, P. A.; Box, M. R.; Bradbury, R. H.; Brown, S. J.; Butterworth, S.; Campbell, A.; Chorley, C.; Colclough, N.; Cross, D. A.; Currie, G. S.; Grist, M.; Hassall, L.; Hill, G. B.; James, D.; James, M.; Kemmitt, P.; Klinowska, T.; Lamont, G.; Lamont, S. G.; Martin, N.; McFarland, H. L.; Mellor, M. J.; Orme, J. P.; Perkins, D.; Perkins, P.; Richmond, G.; Smith, P.; Ward, R. A.; Waring, M. J.; Whittaker, D.; Wells, S.; Wrigley, G. L. J. Med. Chem. 2014, 57, 8249. |
| [16] | Cross, D. A.; Ashton, S. E.; Ghiorghiu, S.; Eberlein, C.; Nebhan, C. A.; Spitzler, P. J.; Orme, J. P.; Finlay, M. R.; Ward, R. A.; Mellor, M. J.; Hughes, G.; Rahi, A.; Jacobs, V. N.; Red Brewer, M.; Ichihara, E.; Sun, J.; Jin, H.; Ballard, P.; Al-Kadhimi, K.; Rowlinson, R.; Klinowska, T.; Richmond, G. H.; Cantarini, M.; Kim, D. W.; Ranson, M. R.; Pao, W. Cancer Discovery 2014, 4, 1046. |
| [17] | Jabbour, E. Am. J. Hematol. 2016, 91, 59. |
| [18] | Kategaya, L.; Di Lello, P.; Rougé, L.; Pastor, R.; Clark, K. R.; Drummond, J.; Kleinheinz, T.; Lin, E.; Upton, J.-P.; Prakash, S.; Heideker, J.; McCleland, M.; Ritorto, M. S.; Alessi, D. R.; Trost, M.; Bainbridge, T. W.; Kwok, M. C. M.; Ma, T. P.; Stiffler, Z.; Brasher, B.; Tang, Y.; Jaishankar, P.; Hearn, B. R.; Renslo, A. R.; Arkin, M. R.; Cohen, F.; Yu, K.; Peale, F.; Gnad, F.; Chang, M. T.; Klijn, C.; Blackwood, E.; Martin, S. E.; Forrest, W. F.; Ernst, J. A.; Ndubaku, C.; Wang, X.; Beresini, M. H.; Tsui, V.; Schwerdtfeger, C.; Blake, R. A.; Murray, J.; Maurer, T.; Wertz, I. E. Nature 2017, 550, 534. |
| [19] | Di Lello, P.; Pastor, R.; Murray, J. M.; Blake, R. A.; Cohen, F.; Crawford, T. D.; Drobnick, J.; Drummond, J.; Kategaya, L.; Kleinheinz, T.; Maurer, T.; Rouge, L.; Zhao, X.; Wertz, I.; Ndubaku, C.; Tsui, V. J. Med. Chem. 2017, 60, 10056. |
| [20] | Modranka, J.; Drogosz-Stachowicz, J.; Pietrzak, A.; Janecka, A.; Janecki, T. Eur. J. Med. Chem. 2021, 219, 113429. |
| [21] | Barghash, R. F.; Eldehna, W. M.; Kovalova, M.; Vojackova, V.; Krystof, V.; Abdel-Aziz, H. A. Eur. J. Med. Chem. 2022, 227, 113952. |
| [22] | Li, J.; Hu, X.; Luo, T.; Lu, Y.; Feng, Y.; Zhang, H.; Liu, D.; Fan, X.; Wang, Y.; Jiang, L.; Wang, Y.; Hao, X.; Shi, T.; Wang, Z. Eur. J. Med. Chem. 2021, 226, 113817. |
| [23] | Ren, Y.; Ruan, Y.; Cheng, B.; Li, L.; Liu, J.; Fang, Y.; Chen, J. Bioorg. Med. Chem. 2021, 46, 116376. |
| [24] | Ma, J.; Liang, W.; Qiang, Y.; Li, L.; Du, J.; Pan, C.; Chen, B.; Zhang, C.; Chen, Y.; Wang, Q. Cell Commun. Signal. 2021, 19, 122. |
| [25] | Fu, S.; Chen, X.; Lo, H. W.; Lin, J. Cancer Lett. 2019, 448, 11. |
| [26] | Legraverend, M.; Bisagni, E. Tetrahedron Lett. 1985, 26, 2001. |
| [27] | Legraverend, M.; Ngongo-Tekam, R. M.; Bisagni, E.; Zerial, A. J Med. Chem. 1985, 28, 1477. |
| [28] | Jansa, P.; Holy, A.; Dracinsky, M.; Kolman, V.; Janeba, Z.; Kostecka, P.; Kmonickova, E.; Zidek, Z. Med. Chem. Res. 2014, 23, 4482. |
/
| 〈 |
|
〉 |