

Chinese Journal of Organic Chemistry >
Design, Synthesis and Anticancer Activity of 2-((Pyridin- 2-ylmethyl)thio)-1H-benzimidazole Derivatives
Received date: 2022-01-02
Revised date: 2022-02-26
Online published: 2022-04-11
Supported by
National Natural Science Foundation of China(21342006); Program for Innovative Research Team of the Ministry of Education(IRT_14R36)
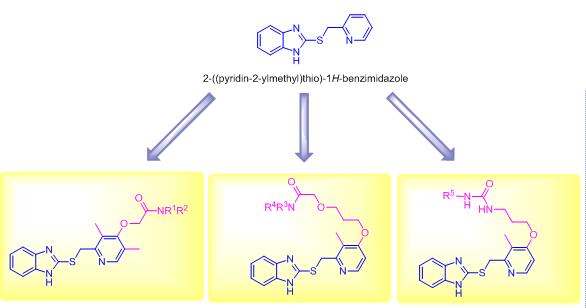
Benzimidazole is an important class of nitrogen-containing heterocyclic compounds, which has diverse biological activities. A series of novel 2-((pyridin-2-ylmethyl)thio)-1H-benzimidazole derivatives were reported and their antiproliferative activities against human lung cancer A549, human colorectal cancer HCT116 and human prostate cancer PC3 cell lines were evaluated with the MTT assay. Some target compounds demonstrated obvious antiproliferative activities against A549, HCT116 and PC3 cancer cell lines. Among them, 2-(3-((2-(((1H-benzimidazol-2-yl)thio)methyl)-3-methylpyridin-4-yl)oxy)propoxy)-1-(4-benzhydrylpiperazin-1-yl)ethanone (7c) showed the most potent antiproliferative activity with IC50 values of 1.14 μmol/L for A549, 1.67 μmol/L for HCT116 and 2.34 μmol/L for PC3, respectively. Preliminary structure-activity relationships were summaried and preliminary cellular mechanism studies elucidated that compound 7c could arrest the cell cycle at G0/G1 phase. The flow cytometry analysis showed that compound 7c could dose-dependently induce A549 cells apoptosis. It is suggested that the 2-((pyridin-2-ylmethyl)thio)-1H-benzimidazole scaffold might be regarded as new scaffold structure for the development of potent cancer chemotherapeutic agents in the drug discovery process.
Key words: benzimidazole derivatives; synthesis; anticancer activity; apoptosis
Jing Zhao , Zhe Jin , Run Wang , Xin'geng Zhang , Yingmei Han , Chun Hu , Xiaoping Liu , Chuanming Zhang , Liping Jin . Design, Synthesis and Anticancer Activity of 2-((Pyridin- 2-ylmethyl)thio)-1H-benzimidazole Derivatives[J]. Chinese Journal of Organic Chemistry, 2022 , 42(7) : 2172 -2183 . DOI: 10.6023/cjoc202201002
| [1] | Shewach, D. S.; Kuchta, R. D. Chem. Rev. 2009, 109, 2859. |
| [2] | Kanwal, A.; Saddique, F. A.; Aslam, S.; Ahmad, M.; Zahoor, A. F.; Mohsin, N. Pharm. Chem. J. 2018, 51, 1068. |
| [3] | Anand, K.; Wakode, S. Int. J. Chem. Stud. 2017, 5, 350. |
| [4] | Hranjec, M.; Kralj, M.; Piantanida, I.; Sedić, M.; Šuman, L.; Pavelić, K.; Karminski-Zamola, G. J. Med. Chem. 2007, 50, 5696. |
| [5] | Piskin, A. K.; Ates-Alagoz, Z.; Atac, F. B.; Musdal, Y.; Buyukbingol, E. Turk. J. Biochem. 2009, 34, 39. |
| [6] | Shaaban, M. A.E.; Kamal, A. M.; Teba, H. E.S. J. Chem. Res. 2016, 40, 228. |
| [7] | Abe, Y.; Matsumoto, S.; Kito, K.; Ueda, N. J. Biol. Chem. 2000, 275, 21525. |
| [8] | Gaudet, S.; Branton, D.; Lue, R. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 5167. |
| [9] | Kim, D.; Li, Y.; Reddy, K.; Lee, M.; Kim, M.; Cho, Y.; Lee, S.; Kim, J.; Bode, A.; Dong, Z. Cancer. Res. 2012, 72, 3060. |
| [10] | Matsuo, Y.; Park, J.; Miyamoto, T.; Yamamoto, S.; Hisada, S.; Alachkar, H.; Nakamura, Y. Sci. Transl. Med. 2014, 6, 251. |
| [11] | Savarino, E.; Marabotto, E.; Zentilin, P.; Furnari, M.; Bodini, G.; Pellegatta, G.; Lorenzon, G.; Coletta, M. D.; Ghisa, M.; Coppo, C.; Marinelli, C.; Savarino, V. Expert Opin. Drug Saf. 2018, 17, 785. |
| [12] | Gillies, R. J.; Pilot, C.; Marunaka, Y.; Fais, S. Biochim. Biophys. Acta, Rev. Cancer 2019, 1871, 273. |
| [13] | Ikemura, K.; Hiramatsu, S.; Okuda, M. Front. Pharmacol. 2017, 8, 911. |
| [14] | Lugini, L.; Federici, C.; Borghi, M.; Azzarito, T.; Marino, M. L.; Cesolini, A.; Spugnini, E. P.; Fais, S. J. Enzyme Inhib. Med. Chem. 2016, 31, 538. |
| [15] | Zeng, X.; Liu, L.; Zheng, M.; Sun, H.; Xiao, J.; Lu, T.; Huang, G.; Chen, P.; Zhang, J.; Zhu, F.; Li, H.; Duan, Q. Oncotarget 2016, 7, 22460. |
| [16] | Zheng, M.; Luan, S.; Gao, S.; Cheng, L.; Hao, B.; Li, J.; Chen, Y.; Hou, X.; Chen, L.; Li, H. Oncotarget 2017, 8, 39143. |
| [17] | Ng, H.-L.; Chen, S.; Chew, E.-H.; Chui, W.-K. Eur. J. Med. Chem. 2016, 115, 63. |
| [18] | Wu, P.; Nielsen, T. E.; Clausen, M. H. Trends Pharmacol. Sci. 2015, 36, 422. |
| [19] | Viegas-Junior, C.; Danuello, A.; Bolzani, V.; Barreiro, E.; Fraga, C. Curr. Med. Chem. 2007, 14, 1829. |
| [20] | Sobhy, M. K.; Mowafy, S.; Lasheen, D. S.; Farag, N. A.; Abouzid, K. A. Bioorg. Chem. 2019, 89, 102988. |
| [21] | Sanmartín, C.; Plano, D.; Domínguez, E.; Font, M.; Calvo, A.; Prior, C.; Encío, I.; Palop, J. A. Molecules 2009, 14, 3313. |
| [22] | Zhivotova, T. S.; Gazaliev, A. M.; Fazylov, S. D.; Aitpaeva, Z. K.; Turdybekov, D. M. Russ. J. Org. Chem. 2006, 42, 448. |
| [23] | Seto, M.; Miyamoto, N.; Aikawa, K.; Aramaki, Y.; Kanzaki, N.; Iizawa, Y.; Baba, M.; Shiraishi, M. Bioorg. Med. Chem. 2005, 13, 363. |
| [24] | Liu, D.; Tian, Z.; Yan, Z.; Wu, L.; Ma, Y.; Wang, Q.; Liu, W.; Zhou, H.; Yang, C. Bioorg. Med. Chem. 2013, 21, 2960. |
| [25] | Diaz-Moralli, S.; Tarrado-Castellarnau, M.; Miranda, A.; Cascante, M. Pharmacol. Ther. 2013, 138, 255. |
| [26] | Liu, X. P.; Xu, H. L.; Sun, R; Li, X.; Hu, B. H.; Hu, C. Lat. Am. J. Pharm. 2015, 34, 126. |
| [27] | Reddy, G. M.; Bhaskar, B. V.; Reddy, P. P.; Sudhakar, P.; Babu, J. M.; Vyas, K.; Reddy, P. R.; Mukkanti, K. J. Pharm. Biomed. Anal. 2007, 4, 1262. |
| [28] | Mosmann, T. J. Immunol. Methods 1983, 65, 55. |
| [29] | Liu, C.; Zhang, M.; Zhang, Z.; Zhang, S. B.; Yang, S.; Zhang, A.; Yin, L.; Swarts, S.; Vidyasagar, S.; Zhang, L.; Okunieff, P. Bioorg. Med. Chem. 2016, 24, 4263. |
| [30] | Xie, Q.; Wang, J.; Liu, M.; Wang, X.; Jiao, D.; Ma, Q.; Jin, Z.; Meng, Q.; Hu, C. Med. Chem. Res. 2019, 28, 1000. |
| [31] | Zhang, L.; Deng, X.-S.; Zhang, C.; Meng, G.-P.; Wu, J.-F.; Li, X.-S.; Zhao, Q.-C.; Hu, C. Med. Chem. Res. 2017, 26, 2180. |
| [32] | Zhang, C.; Tan, X.; Feng, J.; Ding, N.; Li, Y.; Jin, Z.; Meng, Q.; Liu, X.; Hu, C. Molecules 2019, 24, 2108. |
| [33] | Hou, S.; Liang, S.; Zhang, C.; Han, Y.; Liang, J.; Hu, H.; Zhang, X.; Hu, C.; Liu, X.; Zhang, H. Molecules 2021, 26, 3496. |
| [34] | Yan, Q.; Li, R.; Xin, A.; Han, Y.; Zhang, Y.; Liu, J.; Li, W.; Di, D. Bioorg. Med. Chem. 2017, 25, 6542. |
| [35] | Xu, Y.; Jing, D.; Chen, R.; Rashid, H. U.; Jiang, J.; Liu, X.; Wang, L.; Xie, P. Bioorg. Med. Chem. 2018, 26, 4136. |
/
| 〈 |
|
〉 |