

Chinese Journal of Organic Chemistry >
Design, Synthesis and Antifungal Activities of Novel Quinazolinone Derivatives
Received date: 2022-01-14
Revised date: 2022-03-02
Online published: 2022-04-11
Supported by
National Natural Science Foundation of China(21702173); Science Technology Program of Sichuan Province(2021YJ0481)
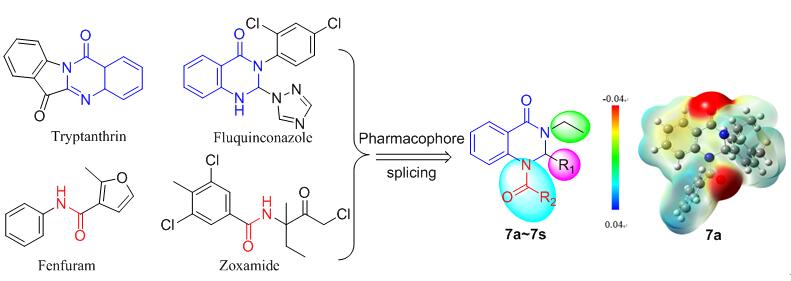
Plant pathogenic fungi have caused enormous yield and economic losses in agricultural production. In order to discover effective antifungal agents, a series of 19 novel quinazolinone derivatives 7 were designed and synthesized based on molecular splicing approach. The classical pharmacophore of amide was introduced into N-1 of quinazolinone skeleton. The structures were confirmed by 1H NMR, 13C NMR and HRMS. Preliminary in vitro antifungal test showed that the target compounds exhibited moderate to favorable activities against six kind of plant pathogenic fungi at the concentration of 50 mg/L. Compound 7a had significant inhibitory effects on Fusarium graminearum and Rhizoctonia solani with EC50 of 12.727 and 12.413 mg/L, respectively, which were better than chlorothalonil (14.323 and 13.339 mg/L). Molecular electrostatic potential of 7a indicated that the amide moieties located in the negative potential region and might generate hydrogen bond with target amino acid residue. The above results provided an effective strategy for the development of quinazolinone antifungal agents.
Wei Chen , Simin Lei , Yuxin Lan , Haojian Xu , Pingbin Yu , Rui Zhang , Run Wu , Yang Chen . Design, Synthesis and Antifungal Activities of Novel Quinazolinone Derivatives[J]. Chinese Journal of Organic Chemistry, 2022 , 42(7) : 2164 -2171 . DOI: 10.6023/cjoc202201020
| [1] | Cui, Z. M.; Zhou, B. H.; Fu, C.; Chen, L.; Fu, J.; Cao, F. J.; Yang, X. J.; Zhou, L. J. Agric. Food Chem. 2020, 68, 15418. |
| [2] | Kang, Z. S. Plant Prot. 2010, 36, 9. (in Chinese) |
| [2] | ( 康振生, 植物保护, 2010, 36, 9.) |
| [3] | Guo, S.; He, F.; Song, B. W. Food Energy Secur. 2021, 10, e293. |
| [4] | Fan, Z.; Shi, J.; Luo, N.; Bao, X. Mol. Diversity 2019, 23, 615. |
| [5] | Liu, Z.; Cao, J.; Yan, X.; Cheng, W.; Wang, X.; Yang, R.; Guo, Y. Chem. Biodiversity 2020, 17, e2000763. |
| [6] | Gupta, T.; Rohilla, A.; Pathak, A.; Akhtar, M. J.; Haider, M. R.; Yar, M. S. Synth. Commun. 2018, 48, 1099. |
| [7] | Peng, J. W.; Yin, X. D.; Li, H.; Ma, K. Y.; Zhang, Z. J.; Zhou, R.; Wang, Y. L.; Hu, G. F.; Liu, Y. Q. J. Agric. Food Chem. 2021, 69, 4604-. |
| [8] | Wang, J.; Battini, N.; Ansari, M. F.; Zhou, C. H. Chin. J. Chem. 2021, 39, 1093. |
| [9] | Liu, Y.; Chen, J.; Xie, D.; Song, B.; Hu, D. J. Agric. Food Chem. 2021, 69, 12135. |
| [10] | Yang, X.; Wang, X.; Wu, M. Chin. J. Org. Chem. 2014, 34, 1015. (in Chinese) |
| [10] | ( 杨绪红, 王翔, 吴鸣虎, 有机化学, 2014, 34, 1015.) |
| [11] | Li, X. Q.; Gan, Y. Y.; Meng, J.; Li, W.; Chen, J.; Qi, Y. Y.; Tian, K.; Ouyang, G. P.; Wang, Z. C. J. Heterocycl. Chem. 2018, 55, 1382. |
| [12] | Shi, J.; Ding, M.; Luo, N.; Wan, S.; Li, P.; Li, J.; Bao, X. J. Agric. Food Chem. 2020, 68, 9613. |
| [13] | Chalkha, M.; Moussaoui, A. E.; Hadda, T. B.; Berredjem, M.; Bouzina, A.; Almalki, F. A.; Saghrouchni, H.; Bakhouch, M.; Saadi, M.; Ammari, L. E.; Abdellatiif, M. H.; Yazidi, M. E. J. Mol. Struct. 2021, 1252, 131818. |
| [14] | Liu, X. H.; Qiao, L.; Zhai, Z. W.; Cai, P. P.; Cantrell, C. L.; Tan, C. X.; Weng, J. Q.; Han, L.; Wu, H. K. Pest Manage. Sci. 2019, 75, 2892. |
| [15] | Zhang, R.; Guo, S.; Deng, P.; Wang, Y.; Dai, A.; Wu, J. J. Agric. Food Chem. 2021, 69, 10082. |
| [16] | Noel, R.; Gupat, N.; Pons, V.; Goudet, A.; Garcia-Castillo, M. D.; Michau, A.; Martinez, J.; Buisson, D. A.; JoHannes, L.; Gillet, D.; Barbier, J.; Cintrat, J. C. J. Med. Chem. 2013, 56, 3404. |
| [17] | Takács, A.; Fodor, A.; Németh, J.; Hell, Z. Synth. Commun. 2014, 44, 2269. |
| [18] | Dabiria, M.; Salehi, P.; Mohammadi, Ali. A.; Baghbanzadeh, M.; Kozehgiry, G. J. Chem. Res. 2004, 2004, 570. |
| [19] | Krishnaji, T.; Murthy, V. N.; Raghunadh, A.; Rao, L. V. Russ. J. Org. Chem. 2020, 56, 1468. |
| [20] | Heidary, M.; Khoobi, M.; Ghasemi, S.; Habibi, Z.; Faramarzi, M. Adv. Synth. Catal. 2014, 356, 1789. |
| [21] | Mohammadpoor-Baltork, I.; Khosropour, A. R.; Moghadam, M.; Tangestaninejad, S.; Mirkhani, V.; Baghersad, S.; Mirjafari, A. C. R. Chim. 2011, 14, 944. |
| [22] | Kemnitzer, W.; Sirisoma, N.; Jiang, S.; Kasibhatla, S.; Crogan-Grundy, C.; Tseng, B.; Drewe, J.; Cai, S. X. Bioorg. Med. Chem. Lett. 2010, 20, 1288. |
| [23] | Zuo, H. L.; Lei, S. M.; Zhang, R.; Li, Y. X.; Chen, W. Chem. J. Chin. Univ. 2021, 42, 2766 (In Chinese). |
| [23] | ( 左怀龙, 雷思敏, 张锐, 李玉新, 陈伟, 高等学校化学学报, 2021, 42, 2766.) |
| [24] | Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Petersson, G. A.; Nakatsuji, H.; Li, X.; Caricato, M.; Marenich, A.; Bloino, J.; Janesko, B. G.; Gomperts, R.; Mennucci, B.; Hratchian, H. P.; Ortiz, J. V.; Izmaylov, A. F.; Sonnenberg, J. L.; Williams-Young, D.; Ding, F.; Lipparini, F.; Egidi, F.; Goings, J.; Peng, B.; Petrone, A.; Henderson, T.; Ranasinghe, D.; Zakrzewski, V. G.; Gao, J.; Rega, N.; Zheng, G.; Liang, W.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Throssell, K.; Montgomery, J. A.; Peralta, J. E.; Ogliaro, F.; Bearpark, M.; Heyd, J. J.; Brothers, E.; Kudin, K. N.; Staroverov, V. N.; Keith, T.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J. C.; Iyengar, S. S.; Tomasi, J.; Cossi, M.; Millam, J. M.; Klene, M.; Adamo, C.; Cammi, R.; Ochterski, J. W.; Martin, R. L.; Morokuma, K.; Farkas, O.; Foresman, J. B.; Fox, D. J. Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford CT, 2015. |
| [25] | Sun, C. X.; Zhang, S.; Qian, P.; Li, Y.; Ren, W. S.; Deng, H.; Jiang, L. Pest Manage. Sci. 2021, 77, 5529. |
/
| 〈 |
|
〉 |