

Chinese Journal of Organic Chemistry >
Synthesis of Azedarachol and 2α,3α,20R-Trihydroxypregnane-16β-methacrylate
Received date: 2022-03-21
Revised date: 2022-04-27
Online published: 2022-05-07
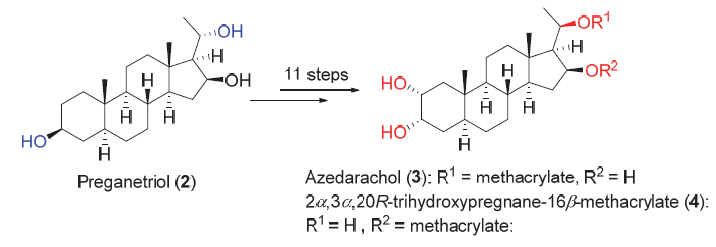
Azedarachol (3) and 2α,3α,20R-trihydroxypregnane-16β-methacrylate (4) were isolated from the root bark of Melia azedarach L. in 1984 by Nakatani and coworkers. Azedarachol (3) shows excellent antifeedant activity against the larvae of Ajrotis sejetum Denis at the concentration of 500 μg/mL, and 2α,3α,20R-trihydroxypregnane-16β-methacrylate (4) inhibits the HGC27 tumor cells with an IC50 value of (11.3±0.5) μg/mL. The noteworthy activities and the high oxidation state structure as well as the scarce of sample suppling in nature resources have attracted considerable attention from the synthetic community. Herein the synthesis of these two natural products from preganetriol (2) is reported. The route features a stereoselective inversion at C(20) and a substrate controlled dihydroxylation of C(23) double bond. 3 and 4 were obtained in an overall yields of 6% (11 steps) and 7.5% (11 steps), respectively.
Ran Gao , Weisheng Tian . Synthesis of Azedarachol and 2α,3α,20R-Trihydroxypregnane-16β-methacrylate[J]. Chinese Journal of Organic Chemistry, 2022 , 42(8) : 2521 -2526 . DOI: 10.6023/cjoc202203040
| [1] | Nakatani, M.; Takao, H.; Miura, I.; Hase, T. Phytochemirtry 1985, 24, 1945. |
| [2] | (a) Wu, S. B.; Ji, Y. P.; Zhu, J. J.; Zhao, Y.; Xia, G.; Hu, Y. H.; Hu, J. F. Steroids 2009, 74, 761. |
| [2] | (b) Wu, S.-B.; Bao, Q-Y.; Wang, W.-X.; Zhao, Y.; Xia, G.; Zhao, Z.; Zeng, H.-Q.; Hu, J.-F. Planta Med. 2011, 77, 922. |
| [3] | (a) Tian, W. S.; Shi, Y. Prog. Chem. 2010, 22, 537. (in Chinese) |
| [3] | (田伟生, 史勇, 化学进展, 2010, 22, 537.) |
| [3] | (b) Tian, W. S. CN 1146457, 1996[Chem. Abstr. 1998, 128, 167599]. |
| [3] | (c) Tian, W. S. Chin. J. Org. Chem. 2000, 18, 11. (in Chinese) |
| [3] | (田伟生, 有机化学, 2000, 18, 11.) |
| [3] | (d) Tian, W. S.; Liu, S. S.; Qiu, B. K.; Wu, X. J. CN 1475494, 2004 [Chem. Abstr. 2004, 142, 374022]. |
| [3] | (e) Gui, J. H.; Wang, D. H.; Tian, W. S. Angew. Chem., nt. Ed. 2011, 50, 7093. |
| [3] | (f) Shi, Y.; Jia, L. Q.; Xiao, Q.; Lan, Q.; Tang, X. H.; Wang, D. H.; Li, M.; Ji, Y.; Zhou, T.; Tian, W. S. Chem.-Asian J. 2011, 6, 786. |
| [3] | (g) Wang, S. S.; Shi, Y.; Tian, W. S. Org. Lett. 2014, 16, 2177. |
| [4] | (a) Zhang, D. S.; Shi, Y.; Tian, W. S. Chin. J. Chem. 2015, 33, 669. |
| [5] | (a) Han, J.; Lin, J. R.; Jin, R. H.; Tian, W. S. Acta Chim. Sinica 2011, 69, 2272. (in Chinese) |
| [5] | (韩军, 林静容, 金荣华, 田伟生, 化学学报, 2011, 69, 2272.) |
| [5] | (b) Han, J. M.S Thesis, Shanghai Normal University, Shanghai, 2011. (in Chinese) |
| [5] | (韩军, 硕士论文, 上海师范大学, 上海, 2011.) |
| [6] | Compound 10/11 cannot be separated by column chromatography, and the proportion of oxidation product 10a/11a obtained by further oxidation is 74%/11%. |
/
| 〈 |
|
〉 |