

Chinese Journal of Organic Chemistry >
Direct Synthesis of 3-Sulfonylquinolines from N-Propargylanilines with Sulfonyl Chlorides
Received date: 2022-03-28
Revised date: 2022-04-26
Online published: 2022-05-07
Supported by
Scientific Research Fund of Hunan Provincial Education Department(19C0783); Science and Technology Innovation Program of Hunan Province(2021RC5028)
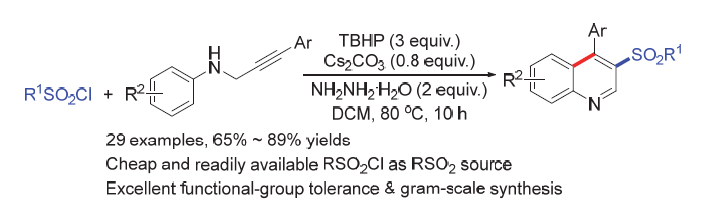
3-Sulfonylquinolines broadly exist in plenty of bioactive molecules and synthetic pharmaceuticals. Herein, an efficient and facile method for direct synthesizing various 3-sulfonylquinolines through one-pot sequential hydrazination/sulfonylation/cyclization reaction from N-propargylanilines and sulfonyl chlorides under mild reaction conditions is reported. This procedure not only can be performed on a gram scale but also be applicable to prepare marketed pharmaceutical derivative.
Keli Wang , Jing Huang , Wei Liu , Zhilin Wu , Xianyong Yu , Jun Jiang , Weimin He . Direct Synthesis of 3-Sulfonylquinolines from N-Propargylanilines with Sulfonyl Chlorides[J]. Chinese Journal of Organic Chemistry, 2022 , 42(8) : 2527 -2534 . DOI: 10.6023/cjoc202203055
| [1] | (a) Cao, M.; Fang, Y.-L.; Wang, Y.-C.; Xu, X.-J.; Xi, Z.-W.; Tang, S. ACS Comb. Sci. 2020, 22, 268. |
| [1] | (b) Wu, Y.; Chen, J.-Y.; Ning, J.; Jiang, X.; Deng, J.; Deng, Y.; Xu, R.; He, W.-M. Green Chem. 2021, 23, 3950. |
| [1] | (c) Yang, W.-C.; Chen, C.-Y.; Li, J.-F.; Wang, Z.-L. Chin. J. Catal. 2021, 42, 1865. |
| [1] | (d) Chen, S.-F.; Liu, X.-C.; Xu, J.-K.; Li, L.; Lang, J.-J.; Wen, G.-B.; Lin, Y.-W. Inorg. Chem. 2021, 60, 2839. |
| [1] | (e) Tong, S.; Li, K.; Ouyang, X.; Song, R.; Li, J. Green Synth. Catal. 2021, 2, 145. |
| [2] | (a) Ivachtchenko, A. V.; Golovina, E. S.; Kadieva, M. G.; Mitkin, O. D.; Okun, I. M. Pharm. Chem. J. 2015, 48, 646. |
| [2] | (b) Galambos, J.; Domány, G.; Nógrádi, K.; Wágner, G.; Keserű, G. M.; Bobok, A.; Kolok, S.; Mikó-Bakk, M. L.; Vastag, M.; Sághy, K.; Kóti, J.; Szakács, Z.; Béni, Z.; Gál, K.; Szombathelyi, Z.; Greiner, I. Bioorg. Med. Chem. Lett. 2016, 26, 1249. |
| [3] | (a) Deng, Q.; Xu, Y.; Liu, P.; Tan, L.; Sun, P. Org. Chem. Front. 2018, 5, 19. |
| [3] | (b) Li, X.-F.; Zhang, X.-G.; Hu, B.-L.; Zhang, X.-H. Org. Biomol. Chem. 2018, 16, 1736. |
| [3] | (c) Chen, W.; Zhang, Y.; Li, P.; Wang, L. Org. Chem. Front. 2018, 5, 855. |
| [3] | (d) Yang, M.; Hu, X.; Ouyang, B.; Xie, W.; Liu, J.-B. Tetrahedron 2019, 75, 3516. |
| [3] | (e) Zhou, N.; Xia, Z.; Wu, S.; Kuang, K.; Xu, Q.; Zhang, M. J. Org. Chem. 2021, 86, 15253. |
| [4] | (a) Feng, Y.; Zhang, Z.; Fu, Q.; Yao, Q.; Huang, H.; Shen, J.; Cui, X. Chin. Chem. Lett. 2020, 31, 58. |
| [4] | (b) Tang, L.; Du, K.; Yu, B.; He, L. Chin. Chem. Lett. 2020, 31, 2991. |
| [4] | (c) Lv, Y.; Cui, H.; Meng, N.; Yue, H.; Wei, W. Chin. Chem. Lett. 2022, 33, 97. |
| [4] | (d) Wang, Z.; Liu, Q.; Liu, R.; Ji, Z.; Li, Y.; Zhao, X.; Wei, W. Chin. Chem. Lett. 2022, 33, 1479. |
| [5] | (a) Tang, S.; Deng, Y.-L.; Li, J.; Wang, W.-X.; Wang, Y.-C.; Li, Z.-Z.; Yuan, L.; Chen, S.-L.; Sheng, R.-L. Chem. Commun. 2016, 52, 4470. |
| [5] | (b) Yuan, L.; Jiang, S.-M.; Li, Z.-Z.; Zhu, Y.; Yu, J.; Li, L.; Li, M.-Z.; Tang, S.; Sheng, R.-R. Org. Biomol. Chem. 2018, 16, 2406. |
| [5] | (c) Yu, J.; Sheng, H.-X.; Wang, S.-W.; Xu, Z.-H.; Tang, S.; Chen, S.-L. Chem. Commun. 2019, 55, 4578. |
| [5] | (d) Gui, Q.-W.; Teng, F.; Yang, H.; Xun, C.; Huang, W.-J.; Lu, Z.-Q.; Zhu, M.-X.; Ouyang, W.-T.; He, W.-M. Chem. Asian J. 2022, 17, e202101139. |
| [6] | (a) Li, L.; Zhang, X.-G.; Hu, B.-L.; Zhang, X.-H. Chem. Asian J. 2019, 14, 4358. |
| [6] | (b) Yuan, J.-M.; Li, J.; Zhou, H.; Xu, J.; Zhu, F.; Liang, Q.; Liu, Z.; Huang, G.; Huang, J. New J. Chem. 2020, 44, 3189. |
| [7] | (a) Zhang, Y.; Chen, W.; Jia, X.; Wang, L.; Li, P. Chem. Commun. 2019, 55, 2785. |
| [7] | (b) Liu, J.; Wang, M.; Li, L.; Wang, L. Green Chem. 2021, 23, 4733. |
| [8] | Zhang, L.; Chen, S.; Gao, Y.; Zhang, P.; Wu, Y.; Tang, G.; Zhao, Y. Org. Lett. 2016, 18, 1286. |
| [9] | Sun, D.; Yin, K.; Zhang, R. Chem. Commun. 2018, 54, 1335. |
| [10] | Liu, Q.; Mei, Y.; Wang, L.; Ma, Y.; Li, P. Adv. Synth. Catal. 2020, 362, 5669. |
| [11] | (a) Zhu, C.; Han, M.-Y.; Liang, X.-X.; Guan, B.; Li, P.; Wang, L. Org. Lett. 2021, 23, 54. |
| [11] | (b) Zhou, D.; Li, Z.-H.; Li, J.; Li, S.-H.; Wang, M.-W.; Luo, X.-L.; Ding, G.-L.; Sheng, R.-L.; Fu, M.-J.; Tang, S. Eur. J. Org. Chem. 2015, 2015, 1606. |
| [11] | (c) Tang, S.; Li, Z.-H.; Wang, M.-W.; Li, Z.-P.; Sheng, R.-L. Org. Biomol. Chem. 2015, 13, 5285. |
| [11] | (d) Ma, C.-H.; Ji, Y.; Zhao, J.; He, X.; Zhang, S.-T.; Jiang, Y.-Q.; Yu, B. Chin. J. Catal. 2022, 43, 571. |
| [12] | (a) Chen, J.-Y.; Wu, H.-Y.; Gui, Q.-W.; Yan, S.-S.; Deng, J.; Lin, Y.-W.; Cao, Z.; He, W.-M. Chin. J. Catal. 2021, 42, 1445. |
| [12] | (b) Yin, L. L.; Yuan, H.; Liu, C.; He, B.; Gao, S.-Q.; Wen, G.-B.; Tan, X.; Lin, Y.-W. ACS Catal. 2018, 8, 9619. |
| [12] | (c) Chen, X.; Xiao, F.; He, W.-M. Org. Chem. Front. 2021, 8, 5206. |
| [12] | (d) Wu, Y.; Chen, J.-Y.; Liao, H.-R.; Shu, X.-R.; Duan, L.-L.; Yang, X.-F.; He, W.-M. Green Synth. Catal. 2021, 2, 233. |
| [12] | (e) Sun, K.; Xiao, F.; Yu, B.; He, W.-M. Chin. J. Catal. 2021, 42, 1921. |
| [12] | (f) Wu, Z.-L.; Chen, J.-Y.; Tian, X.-Z.; Ouyang, W.-T.; Zhang, Z.-T.; He, W.-M. Chin. Chem. Lett. 2022, 33, 1501. |
/
| 〈 |
|
〉 |