

Chinese Journal of Organic Chemistry >
Synthesis of Quinazolin-4(3H)-ones via Ammonium Iodide-Catalyzed Dual Amination of sp3 C—H Bonds
Received date: 2022-03-16
Revised date: 2022-04-25
Online published: 2022-05-27
Supported by
Natural Science Foundation of Anhui Province(2008085QB66); Anhui Science and Technology Talent Introduction Project(HCYJ201903); Innovation and Entrepreneurship Training Program for College Students in Anhui Province(S202010879140)
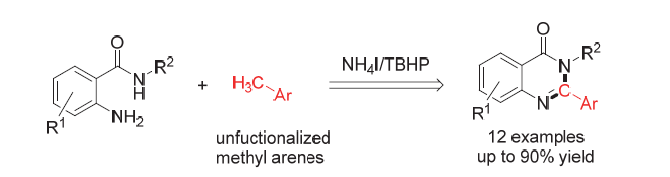
A novel method for synthesizing quinazoline-4(3H)-ones was developed by using inexpensive ammonium iodide as catalyst via dual amination of aromatic benzylic position sp3 C—H bonds. In this reaction, ammonium iodide was used as catalyst, tert-butanol hydrogen peroxide (TBHP) was used as oxidant and dimethyl sulfoxide (DMSO) was used as solvent. The reaction was carried out at 100 ℃ for 20 h to obtain quinazoline-4(3H)-ones compounds with moderate to excellent yields. Furthermore, the mechanism investigations were conducted, and the possible reaction mechanism was depicted. Due to the method does not require use metal catalyst, and has the advantages of simple conditions and wide substrate application range, the present protocol has the potential to be widely applied.
Xiaoxiao Yu , Wangheng Bai , Jianye Zhu , Yuting Zhang , Mengru Zhang , Jiwei Wu . Synthesis of Quinazolin-4(3H)-ones via Ammonium Iodide-Catalyzed Dual Amination of sp3 C—H Bonds[J]. Chinese Journal of Organic Chemistry, 2022 , 42(8) : 2449 -2455 . DOI: 10.6023/cjoc202203030
| [1] | (a) Witt, A.; Bergman, J. Curr. Org. Chem. 2003, 7, 659. |
| [1] | (b) Mhaske, S. B.; Argade, N. P. Tetrahedron 2006, 62, 9787. |
| [2] | (a) Cao, S. L.; Feng, Y. P.; Jiang, Y. Y.; Liu, S.; Ding, Y. G.; Li, R. T. Bioorg. Med. Chem. Lett. 2005, 15, 1915. |
| [2] | (b) He, L.; Li, H.; Chen, J.; Wu, X.-F. RSC Adv. 2014, 4, 12065. |
| [3] | (a) De Laszlo, S. E.; Quagliato, C. S.; Greenlee, W. J.; Patchett, A. A.; Lotti, V. J.; Chen, T. B.; Scheck, S. A.; Faust, A. J. Med. Chem. 1993, 36, 3207. |
| [3] | (b) Kung, P. P.; Casper, M. D.; Cook, K. L.; Wilson-Lingardo, L.; Risen, L. M.; Vickers, T.; Ranken, A. R.; Blyn, L. B.; Wyatt, J. R.; Cook, P. D. J. Med. Chem. 1999, 42, 4705. |
| [3] | (c) Connolly, D. J.; Cusack, D.; O’Sullivan, T. P.; Guiry, P. J. Tetrahedron 2005, 61, 10153. |
| [4] | (a) Horton, D. A.; Bourne, G. T.; Smythe, M. L. Chem. Rev. 2003, 103, 893. |
| [4] | (b) Dong, Y.; Zhang, J.; Yang, J.; Yan, C.; Wu, Y. New J. Chem. 2021, 45, 15344. |
| [4] | (c) Philips, A.; Raja, D.; Arumugam, A.; Lin, W.-Y.; Chandru Senadi, G. Asian J. Org. Chem. 2021, 10, 1795. |
| [4] | (d) Bao, Y.; Yan, Y.; Xu, K.; Su, J.; Zha, Z.; Wang, Z. J. Org. Chem. 2015, 80, 4736. |
| [5] | (a) Purandare, A. V.; Gao, A. M.; Wan, H. H.; Somerville, J.; Burke, C.; Seachord, C.; Vaccaro, W.; Wityak, J.; Poss, M. A. Bioorg. Med. Chem. Lett. 2005, 15, 2669. |
| [5] | (b) Potewar, T. M.; Nadaf, R. N.; Daniel, T.; Lahoti, R. J.; Srinivasan, K. V. Synth. Commun. 2005, 35, 231. |
| [6] | (a) Hisano, T.; Ichikawa, M.; Nakagawa, A.; Tsuji, M. Chem. Pharm. Bull. 1975, 23, 1910. |
| [6] | (b) Mitobe, Y.; Ito, S.; Mizutani, T.; Nagase, T.; Sato, N.; Tokita, S. Bioorg. Med. Chem. Lett. 2009, 19, 4075. |
| [6] | (c) Balakumar, C.; Lamba, P.; Kishore, D. P.; Narayana, B. L.; Rao, K. V.; Rajwinder, K.; Rao, A. R.; Shireesha, B.; Narsaiah, B. Eur. J. Med. Chem. 2010, 45, 4904. |
| [7] | Hikawa, H.; Ino, Y.; Suzuki, H.; Yokoyama, Y. J. Org. Chem. 2012, 77, 7046. |
| [8] | Watson, A. J. A.; Maxwell, A. C.; Williams, J. M. J. Org. Biomol. Chem. 2012, 10, 240. |
| [9] | (a) Zhou, J.; Fang, J. J. Org. Chem. 2011, 76, 7730. |
| [9] | (b) Zhao, D.; Zhou, Y.-R.; Shen, Q.; Li, J.-X. RSC Adv. 2014, 4, 6486. |
| [10] | Sharif, M.; Opalach, J.; Langer, P.; Beller, M.; Wu, X.-F. RSC Adv. 2014, 4, 8. |
| [11] | Nguyen, T. B.; Ermolenko, L.; Al-Mourabit, A. Green Chem. 2013, 15, 2713. |
| [12] | Wei, H.; Li, T.; Zhou, Y.; Zhou, L.; Zeng, Q. Synthesis 2013, 3349. |
| [13] | (a) Wu, X.-F.; He, L.; Neumann, H.; Beller, M. Chem. Eur. J. 2013, 19, 12635. |
| [13] | (b) Li, H.; He, L.; Neumann, H.; Beller, M.; Wu, X. F. Green Chem. 2014, 16, 1336. |
| [13] | (c) Jiang, X.; Tang, T.; Wang, J.-M.; Chen, Z.; Zhu, Y.-M.; Ji, S.-J. J. Org. Chem. 2014, 79, 5082. |
| [13] | (d) You, S.; Huang, B.; Yan, T.; Cai, M. J. Organomet. Chem. 2018, 875, 35. |
| [13] | (e) Ram, S.; Sharma, A. K.; Das, P. Chem. Eur. J. 2019, 25, 14506. |
| [14] | (a) Zhao, Y.; Wang, H.; Hou, X.; Hu, Y.; Lei, A.; Zhang, H.; Zhu, L. J. Am. Chem. Soc. 2006, 128, 15048. |
| [14] | (b) Liu, C.; Zhang, H.; Shi, W.; Lei, A. Chem. Rev. 2011, 111, 1780. |
| [14] | (c) Tang, S.; Wang, P.; Li, H.; Lei, A. Nat. Commun. 2016, 7, 11676. |
| [14] | (d) Hu, X.; Zhang, G.; Bu, F.; Lei, A. Angew. Chem. Int. Ed. 2018, 57, 1286. |
| [14] | (e) Liu, Y.; Shi, B.; Liu, Z.; Gao, R.; Huang, C.; Alhumade, H.; Wang, S.; Qi, X.; Lei, A. J. Am. Chem. Soc. 2021, 143, 20863. |
| [15] | Zhao, D.; Wang, T.; Li, J. Chem. Commun. 2014, 50, 6471. |
| [16] | Li, Q.; Huang, Y.; Chen, T.; Zhou, Y.; Xu, Q.; Yin, S.; Han, L. Org. Lett. 2014, 16, 3672. |
| [17] | (a) Yang, L.; Shi, X.; Hu, B.; Wang, L. Asian J. Org. Chem. 2016, 5, 494. |
| [17] | (b) Liu, H.; Zhai, T.; Ding, S.; Hou, Y.; Zhang, X.; Feng, L.; Ma, C. Org. Chem. Front. 2016, 3, 1096. |
| [18] | Jang, Y.; Lee, S. B.; Hong, J.; Chun, S.; Lee, J.; Hong, S. Org. Biomol. Chem. 2020, 18, 5435. |
| [19] | (a) Tan, C.; Liu, Y.; Liu, X.; Jia, H.; Xu, K.; Huang, S.; Wang, J.; Tan, J. Org. Chem. Front. 2020, 7, 780. |
| [19] | (b) Tan, C.; Liu, X.; Jia, H.; Zhao, X.; Chen, J.; Wang, Z.; Tan, J. Chem. Eur. J. 2020, 26, 881. |
| [20] | (a) Zhao, D.; Shen, Q.; Lia, J.-X. Adv. Synth. Catal. 2015, 357, 339. |
| [20] | (b) Aruri, H.; Singh, U.; Kumar, S.; Kushwaha, M.; Gupta, A. P.; Wakarma, R. A. V.; Singh, P. P. Org. Lett. 2016, 18, 3638. |
/
| 〈 |
|
〉 |