

Chinese Journal of Organic Chemistry >
Dirhodium/Xantphos-Catalyzed Tandem C—H Functionalization/Allylic Alkylation: Direct Access to 3-Acyl-3-allyl Oxindole Derivatives from N-Aryl-α-diazo-β-keto Amides
Received date: 2022-06-28
Revised date: 2022-08-26
Online published: 2022-09-09
Supported by
National Natural Science Foundation of China(21821002)
A novel [Rh2]/Xantphos-catalyzed one-pot sequence for the synthesis of diverse 3-acyl-3-allyl oxindole derivatives from easily available N-aryl-α-diazo-β-keto amides and allylic compounds has been developed. The notable features of this work include ease of operation, mild reaction conditions, good functional group compatibility and facile diversification of the products. Preliminary mechanistic studies indicate a tandem carbene-induced C—H functionalization and Rh(II)/Xantphos catalyzed allylic alkylation process. Moreover, the choice of Xantphos as the ligand is critical to enable the allylic alkylation process in this catalysis.
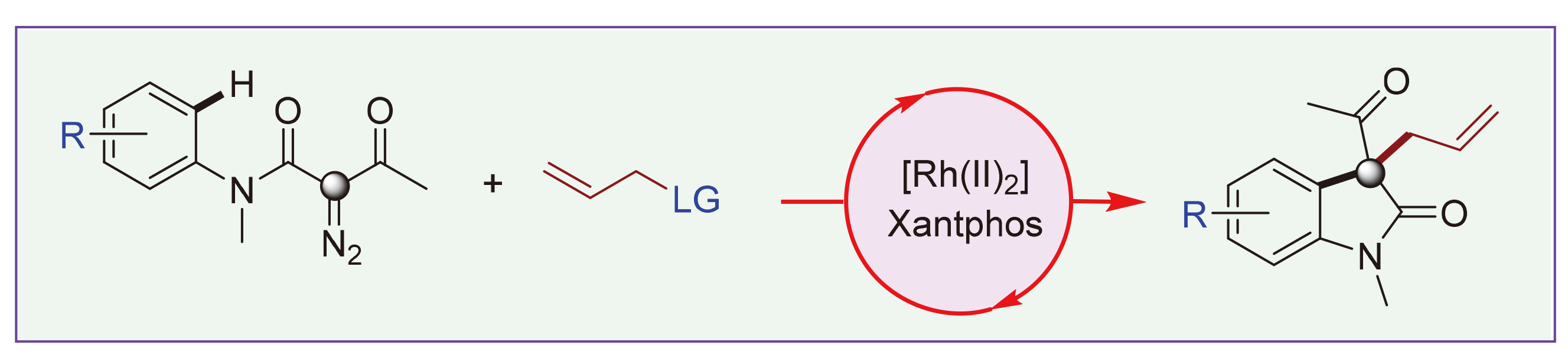
Key words: dirhodium(II); diazo compounds; oxindoles; quaternary centers; relay catalysis
Fangjie Li , Bin Lu , Yang Liu , Xiaoming Wang . Dirhodium/Xantphos-Catalyzed Tandem C—H Functionalization/Allylic Alkylation: Direct Access to 3-Acyl-3-allyl Oxindole Derivatives from N-Aryl-α-diazo-β-keto Amides[J]. Chinese Journal of Organic Chemistry, 2022 , 42(10) : 3390 -3397 . DOI: 10.6023/cjoc202206053
| [1] | (a) Galliford, C. V.; Scheidt, K. A. Angew. Chem., Int. Ed. 2007, 46, 8748. |
| [1] | (b) Yu, B.; Yu, D.-Q.; Liu, H.-M. Eur. J. Med. Chem. 2015, 97, 673. |
| [2] | (a) Zhou, F.; Liu, Y.-L.; Zhou, J. Adv. Synth. Catal. 2010, 352, 1381. |
| [2] | (b) Dalpozzo, R.; Bartoli, G.; Bencivenni, G. Chem. Soc. Rev. 2012, 41, 7247. |
| [2] | (c) Liu, Y.-L.; Wang, X.; Zhao, Y.-L.; Zhu, F.; Zeng, X.-P.; Chen, L.; Wang, C.-H.; Zhao, X.-L.; Zhou, J. Angew. Chem., Int. Ed. 2013, 52, 13735. |
| [2] | (d) Cao, Z.-Y.; Wang, X.; Tan, C.; Zhao, X.-L.; Zhou, J.; Ding, K. J. Am. Chem. Soc. 2013, 135, 8197. |
| [2] | (e) Zhou, F.; Tan, C.; Tang, J.; Zhang, Y.-Y.; Gao, W.-M.; Wu, H.-H.; Yu, Y.-H.; Zhou, J. J. Am. Chem. Soc. 2013, 135, 10994. |
| [2] | (f) Zeng, X.-P.; Cao, Z-Y.; Wang, Y.-H.; Zhou, F.; Zhou, J. Chem. Rev. 2016, 116, 7330. |
| [2] | (g) Cao, Z.-Y.; Zhou, F.; Zhou, J. Acc. Chem. Res. 2018, 51, 1443. |
| [2] | (h) Xu, P.-W.; Yu, J.-S.; Chen, C.; Cao, Z.-Y.; Zhou, F.; Zhou, J. ACS Catal. 2019, 9, 1820. |
| [2] | (i) Marchese, A. D.; Larin, E. M.; Mirabi, B.; Lautens, M. Acc. Chem. Res. 2020, 53, 1605. |
| [2] | (j) Liu, T. T.; Zhang, X. Y.; Miao, Z. W. Chin. J. Org. Chem. 2021, 41, 3965. (in Chinese). |
| [2] | (刘奕彤, 张茜苑, 苗志伟, 有机化学, 2021, 41, 3965.) |
| [3] | (a) Trost, B. M.; Van Vranken, D. L. Chem. Rev. 1996, 96, 395. |
| [3] | (b) Cheng, Q.; Tu, H.-F.; Zheng, C.; Qu, J.-P.; Helmchen, G.; You, S.-L. Chem. Rev. 2019, 119, 1855. |
| [3] | (c) Butt, N. A.; Zhang, W. Chem. Soc. Rev. 2015, 44, 7929. |
| [3] | (d) Turnbull, B. W. H.; Evans, P. A. J. Org. Chem. 2018, 83, 11463. |
| [3] | (e) Zhang, H.; Gu, Q.; You, S.-L. Chin. J. Org. Chem. 2019, 39, 15. (in Chinese). |
| [3] | (张慧君, 顾庆, 游书力, 有机化学, 2019, 39, 15.) |
| [3] | (f) Huang, H.-M.; Bellotti, P.; Glorius, F. Chem. Soc. Rev. 2020, 49, 6186. |
| [4] | (a) Wei, F.; Song, C.-L.; Ma, Y.-D.; Zhou, L.; Tung, C.-H.; Xu, Z.-H. Sci. Bull. 2015, 60, 1479. |
| [4] | (b) Liu, |
| [5] | For selected examples, see: a , Qiu, H.; Li, M.; Jiang, L.-Q.; Lv, F.-P.; Zan, L.; Zhai, C.-W.; Doyle, M. P.; Hu, W.-H. Nat. Chem. 2012, 4, 733. |
| [5] | (b) Xi, Y.; Su, Y.; Yu, Z.; Dong, B.; McClain, E. J.; Lan, Y.; Shi, X. Angew. Chem., Int. Ed. 2014, 53, 9817. |
| [5] | (c) Yu, Z.; Ma, B.; Chen, M.; Wu, H.-H.; Liu, L.; Zhang, J. J. Am. Chem. Soc. 2014, 136, 6904. |
| [5] | (d) Chen, D.-F.; Zhao, F.; Hu, Y.; Gong, L.-Z. Angew. Chem., Int. Ed. 2014, 53, 10763. |
| [5] | (e) Chen, D.-F.; Zhang, C.-l.; Hu, Y.; Han, Z.-Y.; Gong, L.-Z. Org. Chem. Front. 2015, 2, 956. |
| [5] | (f) Jing C.; Xing, D.; Hu, W. Org. Lett. 2015, 17, 4336. |
| [5] | (g) Xu, B.; Li, ML.; Zuo, X.-D.; Zhu, S.-F.; Zhou, Q.-L. J. Am. Chem. Soc. 2015, 137, 8700. |
| [5] | (h) Yu, Z.; Li, Y.; Shi, J.; Ma, B.; Liu, L.; Zhang, J. Angew. Chem.,Int. Ed. 2016, 55, 14807. |
| [5] | (i) Fructos, M. R.; Díaz-Requejo M. M.; Pérez P. J. Chem. Commun. 2016, 52, 7326. |
| [5] | (j) Ma, B.; Chu, Z.; Huang, B.; Liu, Z.; Liu, L.; Zhang, J. Angew. Chem., Int. Ed. 2017, 56, 2749. |
| [5] | (k) Fructos, M. R.; Besora, M.; Braga, A. A. C.; Díaz-Requejo, M. M.; Maseras, F.; Perez, P. J. Organometallics 2017, 36, 172. |
| [5] | (l) Kang, Z.; Zhang, D.; Xu, X.; Hu, W. Org. Lett. 2019, 21, 9878. |
| [5] | (m) Yu, Z.; Li, Y.; Zhang, P.; Liu, L.; Zhang, J. Chem. Sci. 2019, 10, 6553. |
| [5] | (n) Jana, S.; Empel, C.; Pei, C.; Aseeva, P.; Nguyen, T. V.; Koenigs, R. M. ACS Catal. 2020, 10, 9925. |
| [5] | (o) Zhang, C.; Hong, K.; Pei, C.; Zhou, S.; Hu, W.; Hashmi, A. S. K.; Xu, X. Nat. Commun. 2021, 12, 1182. |
| [5] | (p) Zhu, D.-X.; Xia, H.; Liu, J.-G.; Chung, L.-W.; Xu, M.-H. J. Am. Chem. Soc. 2021, 143, 2608. |
| [5] | (q) Li, Z.; Chen, Y.; Wang, C.; Xu, G.; Shao, Y.; Zhang, X.; Sun, J. Angew. Chem., Int. Ed. 2021, 60, 25714. |
| [5] | (r) Pizarro, J. D.; Schmidtke, I. L.; Nova, A.; Fructos, M. R.; Pérez, P. J. ACS Catal. 2022, 12, 6851. |
| [5] | (s) Jia, S.; Xing, D.; Zhang, D.; Hu, W. Angew. Chem., Int. Ed. 2014, 53, 13098. |
| [6] | (a) Jing, C.; Xing, D.; Hu, W. Org. Lett. 2015, 17, 4336. |
| [6] | (b) Kang, Z.; Zhang, D.; Xu, X.; Hu, W. Org. Lett. 2019, 21, 9878. |
| [7] | (a) Yamamoto, K.; Qureshi, Z.; Tsoung, J.; Pisella, G.; Lautens, M. Org. Lett. 2016, 18, 4954. |
| [7] | (b) Chen, L. H.; Ma, Y. T.; Yang, F.; Huang, X. Y.; Chen, S. W.; Ji, K.; Chen, Z. S. Adv. Synth. Cat. 2019, 361, 1307. |
| [7] | (c) Lee, Y. L.; Lee, K. R.; Xuan, Z.; Lee, S.-G. Bull. Korean Chem. Soc. 2021, 42, 537. |
| [8] | (a) Tsuji, J.; Minami, I.; Shimizu, I. Tetrahedron Lett. 1984, 25, 5157. |
| [8] | (b) Turnbull, B. W. H.; Evans, P. A. J. Org. Chem. 2018, 83, 11463. |
| [8] | (c) Thoke, M. B.; Kang, Q. Synthesis 2019, 51, 2585. |
| [9] | (a) Cheng, M.; Huang, X.-Y.; Yang, F.; Zhao, D.-M.; Ji, K.; Chen, Z.-S. Org. Lett. 2022, 24, 1237. |
| [9] | For selected examples of RhII/Pd(0)dual catalyzed two-com- ponent reactions of diazo compounds with allylic compounds, see: (b) Chen, Z.-S.; Huang, X.-Y.; Gao, J.-M.; Ji, K. Org. Lett. 2016, 18, 5876. |
| [9] | (c) Chen, Z.-S.; Huang, X.-Y.; Chen, L.-H.; Gao, J.-M.; Ji, K. ACS Catal. 2017, 7, 7902. |
| [9] | (d) Huang, L.-Z.; Xuan, Z.; Jeon, H. J.; Du, Z.-T.; Kim, J. H.; Lee, S.-G. ACS Catal. 2018, 8, 7340. |
| [9] | (e) Wang, X. X.; Huang, X. Y.; Lei, S. H.; Yang, F.; Gao, J. M.; Ji, K.; Chen, Z. S. Chem. Commun. 2020, 56, 782. |
| [9] | For an elegant RhII/Pd(0)/chiral phosphoric acid catalyzed three- component allylation of α-diazo carbonyl compounds with alcohols and allyl carbonates, see: (f) Kang, Z.; Chang, W.; Tian, X.; Fu, X.; Zhao, W.; Xu, X.; Liang, Y.; Hu, W. J. Am. Chem. Soc. 2021, 143, 20818. |
| [10] | (a) Lu, B.; Liang, X.; Zhang, J.; Wang, Z.; Peng, Q.; Wang, X. J. Am. Chem. Soc. 2021, 143, 11799. |
| [10] | (b) Yang, Y.; Lu, B.; Xu, G.; Wang, X. ACS Cent. Sci. 2022. 8, 581 |
| [11] | For excellent reviews about the axial modification of dirhodium in catalysis, see: (a) Trindade, A. F.; Coelho, J. A. S.; Afonso, C. A. M.; Veiros, L. F.; Gois, P. M. P. ACS Catal. 2012, 2, 370. |
| [11] | (b) Hong, B.; Shi, L.; Li, L.; Zhan, S.; Gu, Z. Green Synth. Catal. 2022. doi.org/10.1016/j.gresc.2022.03.001. |
| [12] | For selected examples, see: (a) Wang, D.; Zhao, Y.; Yuan, C.; Wen, J.; Zhao, Y.; Shi, Z. Angew. Chem., Int. Ed. 2019, 58, 12529. |
| [12] | (b) Fu, L.; Li, S.; Cai, Z.; Ding, Y.; Guo, X.-Q.; Zhou, L.-P.; Yuan, D.; Sun, Q.-F.; Li, G. Nat. Catal. 2018, 1, 469. |
| [12] | (c) Vora, H. U.; Silvestri, A. P.; Engelin, C. J.; Yu, J. Q. Angew. Chem., Int. Ed. 2014, 53, 2683. |
| [12] | (d) Kwak, J.; Kim, M.; Chang, S. J. Am. Chem. Soc. 2011, 133, 3780. |
| [12] | (e) Trindade, A. F.; Gois, P. M. P.; Veiros, L. F.; André, V.; Duarte, M. T.; Afonso, C. A. M.; Caddick, S.; Cloke, F. G. N. J. Org. Chem. 2008, 73, 4076. |
| [12] | (f) Gois, P. M. P.; Trindade, A. F.; Veiros, L. F.; André, V.; Duarte, M. T.; Afonso, C. A. M.; Caddick, S.; Cloke, F. G. N. Angew. Chem., Int. Ed. 2007, 46, 5750. |
| [13] | Moreno-Cabrerizo, C.; Ortega-Martínez, A.; Esteruelas, M. A.; López, A. M.; Nájera, C.; Sansano, J. M. Eur. J. Org. Chem. 2020, 2020, 3101 |
| [14] | Mandal, T.; Chakraborti, G.; Karmakar, S.; Dash, J. Org. Lett. 2018, 20, 4759. |
| [15] | (a) Guo, X.; Hu, W. Acc. Chem. Res. 2013, 46, 2427. |
| [15] | (b) Zhang, D.; Hu, W. Chem. Rec. 2017, 17, 739. |
/
| 〈 |
|
〉 |