

Chinese Journal of Organic Chemistry >
Recent Advances in Photocatalytic Deracemization
Received date: 2022-07-31
Revised date: 2022-09-04
Online published: 2022-09-15
Supported by
Start-up Funding from Wuhan University(691000002); Start-up Funding from Wuhan University(600460026); National Natural Science Foundation of China(21820102003); National Natural Science Foundation of China(91956201)
Chirality is widely found in many natural products and drug molecules. It is of great significance to obtain optically pure chiral compounds because of the fact that different enantiomers have distinct or even opposite physiological activities. Deracemization is the most direct, efficient and atom-economic approach to obtain a single enantiomer, and the emerging photocatalytic deracemization attracts much attention because of its high efficiency. Herein, the recent advances in photocatalytic deracemization are summarized. The future research direction of this field is also prospected.
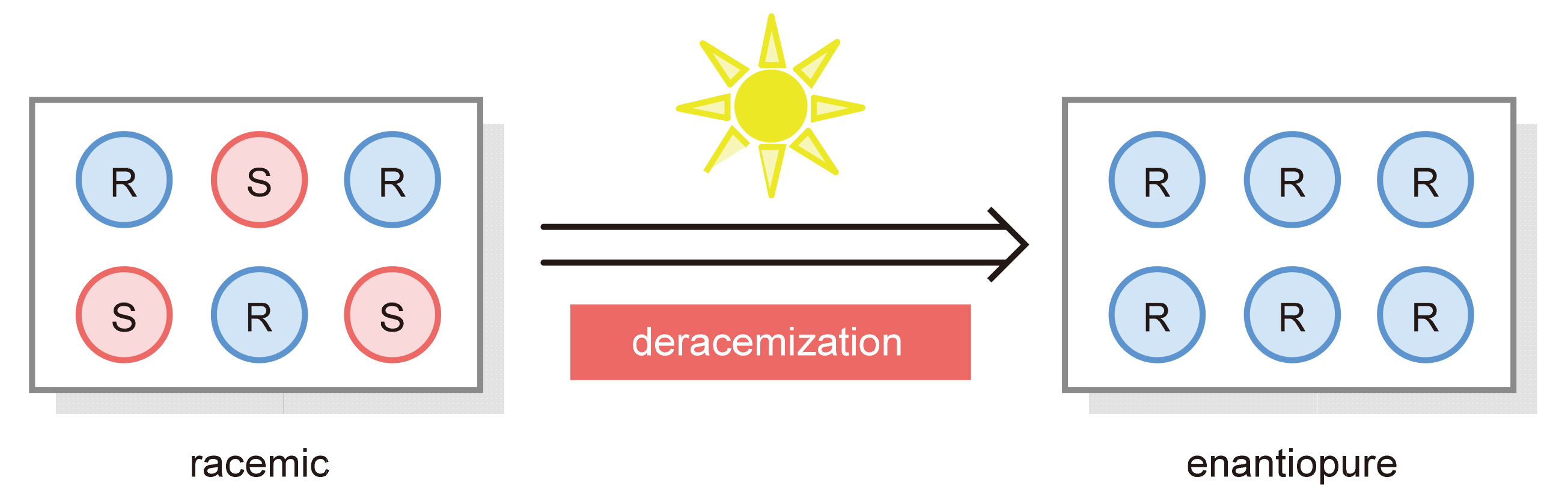
Key words: deracemization; chiral molecule; asymmetric synthesis; photocatalysis
Yiwen Su , Youquan Zou , Wenjing Xiao . Recent Advances in Photocatalytic Deracemization[J]. Chinese Journal of Organic Chemistry, 2022 , 42(10) : 3201 -3212 . DOI: 10.6023/cjoc202207046
| [1] | List, B.; Yang, J. W. Science 2006, 313, 1584. |
| [2] | Graaff, C.; Ruijter, E.; Orru, R. V. A. Chem. Soc. Rev. 2012, 41, 3969. |
| [3] | Han, B.; He, X. H.; Liu, Y. Q.; He, G.; Peng, C.; Li, J. L. Chem. Soc. Rev. 2021, 50, 1522. |
| [4] | Lin, G.-Q.; Wang, M.-X. Chiral Synthesis and Chiral Drugs, Chemical Industry Press, Beijing, 2008. (in Chinese). |
| [4] | (林国强, 王梅祥, 手性合成与手性药物, 化学工业出版社, 北京, 2008.) |
| [5] | Ding, K.-L.; Fan, Q.-H. New Concepts and Methods for Asymmetric Catalysis, Chemical Industry Press, Beijing, 2008. (in Chinese). |
| [5] | (丁奎岭, 范青华, 不对称催化新概念与新方法, 化学工业出版社, 北京, 2009.) |
| [6] | Zheng, C.; You, S.-L. ACS Cent. Sci. 2021, 7, 432. |
| [7] | Wang, D.-S.; Chen, Q.-A.; Lu, S.-M.; Zhou, Y.-G. Chem. Rev. 2012, 112, 2557. |
| [8] | Faber, K. Chem. Eur. J. 2001, 7, 5004. |
| [9] | Blackmond, D. G. Angew. Chem., Int. Ed. 2009, 48, 2648. |
| [10] | Nakamura, T.; Ban, K.; Yoshida, Y.; Mino, T.; Kasashima, Y.; Sakamoto, M. Chem. Eur. J. 2021, 27, 16338. |
| [11] | Sanada, K.; Washio, A.; Ishikawa, H.; Yoshida, Y.; Mino, T.; Sakamoto, M. Angew. Chem., Int. Ed. 2022, 61, e202201268. |
| [12] | Sanada, K.; Washio, A.; Nishihata, K.; Yagishita, F.; Yoshida, Y.; Mino, T.; Suzuki, S.; Kasashima, Y.; Sakamoto, M. Cryst. Growth Des. 2021, 21, 6051. |
| [13] | Kolarovič, A.; Jakubec, P. Adv. Synth. Catal. 2021, 363, 4110. |
| [14] | Guillot, M.; Meester, J.; Huynen, S.; Collard, L.; Robeyns, K.; Riant, O.; Leyssens, T. Angew. Chem., Int. Ed. 2020, 59, 11303. |
| [15] | Kosmrlj, J.; Weigel, L. O.; Evans, D. A.; Downey, C. W.; Wu, J. J. Am. Chem. Soc. 2003, 125, 3208. |
| [16] | Aranda, C.; Oksdath-Mansilla, G.; Bisogno, F. R.; Gonzalo, G. Adv. Synth. Catal. 2020, 362, 1233. |
| [17] | Rachwalski, M.; Vermue, N.; Rutjes, F. P. J. T. Chem. Soc. Rev. 2013, 42, 9268. |
| [18] | Adair, G. R. A.; Williams, J. M. J. Chem. Commun. 2007, 25, 2608. |
| [19] | Shimada, Y.; Miyake, Y.; Matsuzawa, H.; Nishibayashi, Y. Chem. Asian J. 2007, 2, 393. |
| [20] | Lackner, A. D.; Samant, A. V.; Toste, F. D. J. Am. Chem. Soc. 2013, 135, 14090. |
| [21] | Ji, Y.; Shi, L.; Chen, M. W.; Feng, G. S.; Zhou, Y. G. J. Am. Chem. Soc. 2015, 137, 10496. |
| [22] | Wan, M.; Sun, S. T.; Li, Y. S.; Liu, L. Angew. Chem., Int. Ed. 2017, 56, 5116. |
| [23] | Qu, P. Y.; Kuepfert, M.; Jockusch, S.; Weck, M. ACS Catal. 2019, 9, 2701. |
| [24] | Mao, Y.; Wang, Z. H.; Wang, G.; Zhao, R.; Kan, L. L.; Pan, X. G.; Liu, L. ACS Catal. 2020, 10, 7785. |
| [25] | Zhang, Z. K.; Hu, X. L. Angew. Chem., Int. Ed. 2021, 60, 22833. |
| [26] | Zhao, Z. T.; Wang, C. Y.; Chen, Q. P.; Wang, Y.; Xiao, R.; Tan, C. X.; Liu, G. H. ChemCatChem 2021, 13, 4055. |
| [27] | Chen, X.-H.; Zhao, R.; Liu, Z.-Q.; Sun, S.-T.; Ma, Y.-G.; Liu, Q.-Y.; Sun, X.; Liu, L. Chin. Chem. Lett. 2021, 32, 2305. |
| [28] | Ma, Y.-G.; Liu, X.-G.; Mao, Y.; Huang, J.-C.; Ma, S.-T.; Liu, L. Org. Chem. Front. 2020, 7, 2526. |
| [29] | Zhang, L.; Zhu, R.-X.; Feng, A.-L.; Zhao, C.-Y.; Chen, L.; Feng, G.-D.; Liu, L. Chem. Sci. 2020, 11, 4444. |
| [30] | Meng, F.-J.; Shao, B.-R.; Velopolcek, M. K.; Guo, X.; Feng, G.-S.; Shi, L. Org. Biomol. Chem. 2021, 19, 10570. |
| [31] | Lu, R.; Li, Y.-S.; Zhao, J.-Q.; Li, J.; Wang, S.-G.; Liu, L. Chem. Commun. 2018, 54, 4445. |
| [32] | Chen, X.-H.; Yan, L.; Zhang, L.; Zhao, C.-Y.; Feng, G.-D.; Chen, L.; Sun, S.-T.; Liu, Q.-Y.; Liu, L. Tetrahedron Lett. 2020, 61, 152107. |
| [33] | Yang, B.; Cui, P.; Chen, Y.-S.; Liu, Q.-X.; Zhou, H.-F. Tetrahedron Lett. 2020, 61, 152530. |
| [34] | Shi, Q. L.; Ye, J. T. Angew. Chem., Int. Ed. 2020, 59, 4998. |
| [35] | Hölzl-Hobmeier1, A.; Bauer, A.; Silva, A. V.; Huber, S. M.; Bannwarth, C.; Bach, T. Nature 2018, 564, 240. |
| [36] | Plaza, M.; Jandl, C.; Bach, T. Angew. Chem., Int. Ed. 2020, 59, 12785. |
| [37] | Plaza, M.; Großkopf, J.; Breitenlechner, S.; Bannwarth, C.; Bach, T. J. Am. Chem. Soc. 2021, 143, 11209. |
| [38] | Kratz, T.; Steinbach, P.; Breitenlechner, S.; Storch, G.; Bannwarth, C.; Bach, T. J. Am. Chem. Soc 2022, 144, 10133. |
| [39] | Tröster, A.; Bauer, A.; Jandl, C.; Bach, T. Angew. Chem., Int. Ed. 2019, 58, 3538. |
| [40] | Li, X. Y.; Kutta, R. J.; Jandl, C.; Bauer, A.; Nuernberger, P.; Bach, T. Angew. Chem., Int. Ed. 2020, 59, 21640. |
| [41] | Großkopf, J.; Plaza, M.; Seitz, A.; Breitenlechner, S.; Storch, G.; Bach, T. J. Am. Chem. Soc. 2021, 143, 21241. |
| [42] | Wimberger, L.; Kratz, T.; Bach, T. Synthesis 2019, 51, 4417. |
| [43] | Shin, N. Y.; Ryss, J. M.; Zhang, X.; Miller, S. J.; Knowles, R. R. Science 2019, 366, 364. |
| [44] | Zhang, C. H.; Gao, A. Z.; Nie, X; Ye, C. X.; Ivlev, S. I.; Chen, S. M.; Meggers, E. J. Am. Chem. Soc. 2021, 143, 13393. |
| [45] | Huang, M. X.; Zhang, L.; Pan, T. R.; Luo, S. Z. Science 2022, 375, 869. |
| [46] | Huang, X.; Ma, S. M. Acc. Chem. Res. 2019, 52, 1301. |
| [47] | Ouannes, C.; Beugelmans, R.; Roussi, G. J. Am. Chem. Soc. 1973, 95, 8472. |
| [48] | Mukherjee, S.; Yang, J. W.; Hoffmann, S.; List, B. Chem. Rev. 2007, 107, 5471. |
| [49] | Zou, Y.-Q.; Hörmann, F. M.; Bach, T. Chem. Soc. Rev. 2018, 47, 278. |
| [50] | Xuan, J.; Xiao, W.-J. Angew. Chem., Int. Ed. 2012, 51, 6828. |
| [51] | Chen, J.-R.; Hu, X.-Q.; Lu, L.Q.; Xiao, W.-J. Chem. Soc. Rev. 2016, 45, 2044. |
| [52] | Zhou, Q.-Q.; Zou, Y.-Q.; Lu, L.Q.; Xiao, W.-J. Angew. Chem., Int. Ed. 2019, 58, 1586. |
| [53] | Nevesely?, T.; Wienhold, M.; Molloy, J. J.; Gilmour, R. Chem. Rev. 2022, 122, 2650. |
| [54] | Fu, N. K.; Zhang, L.; Li, J. Y.; Luo, S. Z.; Cheng, J. P. Angew. Chem., Int. Ed. 2011, 50, 11451. |
| [55] | Balavoine, G.; Juge, S.; Kagan, H. B. Tetrahedron Lett. 1973, 14, 4159. |
/
| 〈 |
|
〉 |