

Chinese Journal of Organic Chemistry >
Recent Advances in Visible-Light-Catalyzed C—C Bonds and C—Heteroatom Bonds Formation Using Sulfonium Salts
Received date: 2022-09-05
Revised date: 2022-10-06
Online published: 2022-11-01
Supported by
National Natural Science Foundation of China(22271170)
Sulfonium salts are one of the most important class of organosulfur (IV) compounds which have a positive charge on the sulfur center with three C—S bonds. Because of their bench-stable, easy synthesis, broad structural diversity, and rich reactivity, sulfonium salts are playing a significant role in synthetic chemistry. In recent years, visible-light promoted photoredox catalysis is rapidly developing into a powerful tool for organic synthesis. In this paper, the recent advances of different sulfonium salts in the radical type reactions induced by visible light are summarized. The formation reactions of C—C bond and C—X (X=B, N, O, S, Se, Te, F, Cl, I) bonds are introduced, and the applicable scope and mechanism of some reactions are also discussed.
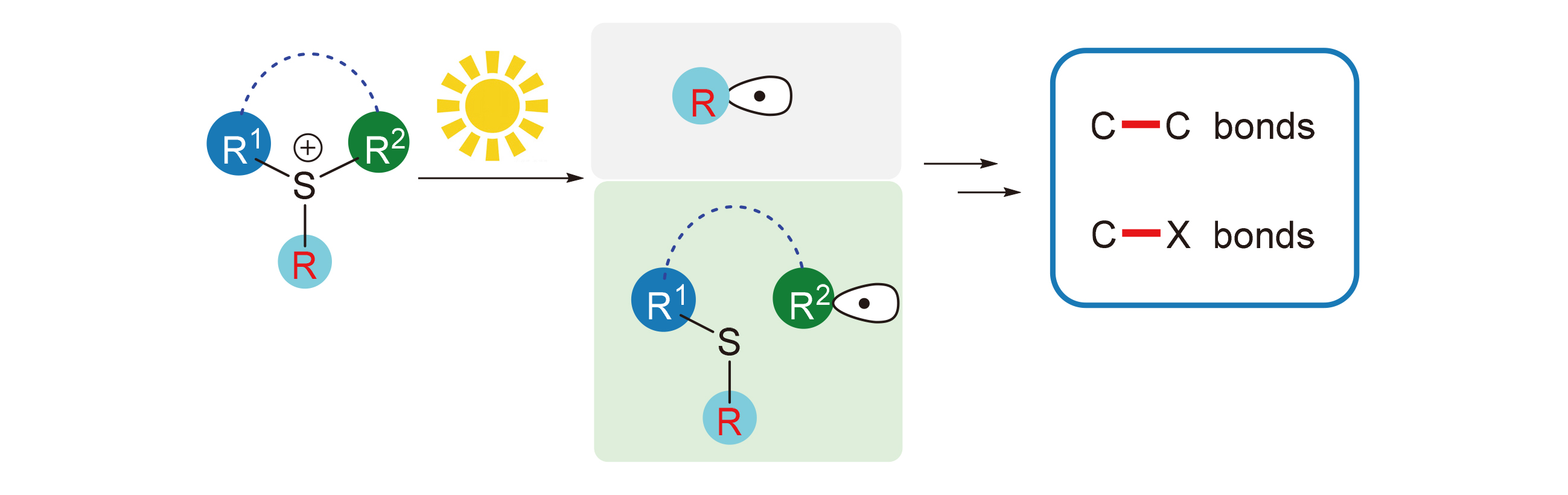
Key words: sulfur chemistry; sulfonium salt; visible light; free radical reaction
Hao Xu , Jie Zhang , Junze Zuo , Fengxiao Wang , Jian Lü , Xu Hun , Daoshan Yang . Recent Advances in Visible-Light-Catalyzed C—C Bonds and C—Heteroatom Bonds Formation Using Sulfonium Salts[J]. Chinese Journal of Organic Chemistry, 2022 , 42(12) : 4037 -4059 . DOI: 10.6023/cjoc202209004
| [1] | (a) Kozhushkov, S. I.; Alcarazo, M. Eur. J. Inorg. Chem. 2020, 2486. |
| [1] | (b) Wang, M.; Jiang, X. ACS Sustainable Chem. Eng. 2022, 10, 671. |
| [1] | (c) Gan, Z.; Zhu, X.; Yan, Q.; Song, X.; Yang, D. Chin. Chem. Lett. 2021, 32, 1705. |
| [2] | Ilardi, E. A.; Vitaku, E.; Njardarson, J. T. J. Med. Chem. 2014, 57, 2832. |
| [3] | (a) Mondal, M.; Chen, S.; Kerrigan, N. J. Molecules 2018, 23, 738. |
| [3] | (b) Nenaidenko, V. G.; Balenkova, E. S. Russ. J. Org. Chem. 2003, 39, 291. |
| [3] | (c) Chen, J.; Li, J.; Plutschack, M. B.; Berger, F.; Ritter, T. Angew. Chem., Int. Ed. 2020, 59, 5616 |
| [3] | (d) Chen, X.-Y.; Huang, Y.-H.; Zhou, J.; Wang, P. Chin. J. Chem. 2020, 38, 1269 |
| [3] | (e) Nie, X.-X.; Huang, Y.-H.; Wang, P. Org. Lett. 2020, 22, 7716. |
| [3] | (f) Kendall, A. J.; Mock, M. T. Eur. J. Inorg. Chem. 2020, 1347. |
| [3] | (g) Tian, Z.-Y.; Hu, Y.-T.; Teng, H.-B.; Zhang, C.-P. Tetrahedron Lett. 2018, 59, 299. |
| [3] | (h) Chen, J.; Li, J.; Plutschack, M. B.; Berger, F.; Ritter, T. Angew. Chem., Int. Ed. 2020, 59, 5616. |
| [3] | (i) Kendall, A. J.; Mock, M. T. Eur. J. Inorg. Chem. 2020, 1347. |
| [3] | (j) Tian, Z.-Y.; Ma, Y.; Zhang, C.-P. Synthesis 2022, 54, 1478. |
| [3] | (k) Wang, C.; Liu, B.; Shao, Z.; Zhou, J.; Shao, A.; Zou, L.-H.; Wen, J. Org. Lett. 2022, 24, 5391. |
| [4] | (a) Kaiser, D.; Klose, I.; Oost, R.; Neuhaus, J.; Maulide, N. Chem. Rev. 2019, 119, 8701. |
| [4] | (b) Kozhushkov, S. I.; Alcarazo, M. Eur. J. Inorg. Chem. 2020, 2486. |
| [4] | (c) Péter, á.; Perry, G. J. P.; Procter, D. J. Adv. Synth. Catal. 2020, 362, 2135. |
| [4] | (d) Yorimitsu, H. Chem. Rec. 2021, 21, 3356. |
| [4] | (e) Tian, J.; Gao, W. C.; Jiang, X.-F. Chem. Reagents 2021, 43, 447. (in Chinese) |
| [4] | ( 田俊, 高文超, 姜雪峰, 化学试剂, 2021, 43, 447.) |
| [5] | Broderick, J. B.; Duffus, B. R.; Duschene, K. S.; Shepard, E. M. Chem. Rev. 2014, 114, 4229. |
| [6] | (a) Aggarwal, V. K.; Winn, C. L. Acc. Chem. Res. 2004, 37, 611. |
| [6] | (b) Lu, L.-Q.; Li, T.-R.; Wang, Q.; Xiao, W.-J. Chem. Soc. Rev. 2017, 46, 4135. |
| [6] | (c) Shi, J.; Li, Y.; Li, Y. Chem. Soc. Rev. 2017, 46, 1707. |
| [7] | Garst, M. E.; McBride, B. J.; Johnson, A. T. J. Org. Chem. 1983, 48, 8. |
| [8] | Wang, L.; He, W.; Yu, Z. Chem. Soc. Rev. 2013, 42, 599. |
| [9] | Otsuka, S.; Nogi, K.; Yorimitsu, H. Top. Curr. Chem. 2018, 376, 13. |
| [10] | (a) Beeson, T. D.; Mastracchio, A.; Hong, J.-B.; Ashton, K.; MacMillan, D. W. C. Science 2007, 316, 582. |
| [10] | (b) Du, J.; Yoon, T. P. J. Am. Chem. Soc. 2009, 131, 14604. |
| [10] | (c) Schultz, D. M.; Yoon, T. P. Science 2014, 343, 1239176. |
| [11] | (a) Prier, C. K.; Rankic, D. A.; MacMillan, D. W. C. Chem. Rev. 2013, 113, 5322. |
| [11] | (b) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Acc. Chem. Res. 2016, 49, 1911 |
| [11] | (c) Romero, N. A.; Nicewicz, D. A. Chem. Rev. 2016, 116, 10075. |
| [11] | (d) Xie, L.-Y.; Peng, S.; Yang, L.-H.; Peng, C.; Lin, Y.-W.; Yu, X.; Cao, Z.; Peng, Y.-Y.; He, W.-M. Green Chem. 2021, 23, 374. |
| [11] | (e) Liu, Y.; Chen, X.-L.; Li, X.-Y.; Zhu, S.-S.; Li, S.-J.; Song, Y.; Qu, L.-B.; Yu, B. J. Am. Chem. Soc. 2021, 143, 964. |
| [11] | (f) Li, G.-H.; Han, Q.-Q.; Sun, Y.-Y.; Chen, D.-M.; Wang, Z.-L.; Xu, X.-M.; Yu, X.-Y. Chin. Chem. Lett. 2020, 31, 3255. |
| [11] | (g) Wang, Z.; Liu, Q.; Ji, X.; Deng, G.-J.; Huang, H. ACS Catal. 2020, 10, 154. |
| [11] | (h) Kim, J.; Kim, D.; Chang, S. J. Am. Chem. Soc. 2020, 142, 19052. |
| [11] | (i) Zhang, Q.-B.; Ban, Y.-L.; Zhou, D.-G.; Zhou, P.-P.; Wu, L.-Z.; Liu, Q. Org. Lett. 2016, 18, 5256. |
| [11] | (j) Wu, C.; Bian, Q.; Ding, T.; Tang, M.; Zhang, W.; Xu, Y.; Liu, B.; Xu, H.; Li, H.-B.; Fu, H. ACS Catal. 2021, 11, 9561. |
| [11] | (k) He, S.; Chen, X.; Zeng, F.; Lu, P.; Peng, Y.; Qu, L.; Yu, B. Chin. Chem. Lett. 2020, 31, 1863. |
| [11] | (l) Yang, D.; Yan, Q.; Zhu, E.; Lv, J.; He, W.-M. Chin. Chem. Lett. 2022, 33, 1798. |
| [11] | (m) Meng, N.; Lv, Y.; Liu, Q.; Liu, R.; Zhao, X.; Wei, W. Chin. Chem. Lett. 2021, 32, 258. |
| [11] | (n) Meng, Q.-Y.; Schirmer, T. E.; Berger, A. L.; Donabauer, K.; K?nig, B. J. Am. Chem. Soc. 2019, 141, 11393. |
| [11] | (o) Bagdi, A. K.; Rahman, M.; Bhattacherjee, D.; Zyryanov, G. V.; Ghosh, S.; Chupakhin, O. N.; Hajra, A. Green Chem. 2020, 22, 6632. |
| [11] | (p) Fu, Q.; Bo, Z.-Y.; Ye, J.-H.; Ju, T.; Huang, H.; Liao, L.-L.; Yu, D.-G. Nat. Commun. 2019, 10, 3592. |
| [11] | (q) Xie, J.; Jin, H.; Hashmi, A. S. K. Chem. Soc. Rev. 2017, 46, 5193. |
| [11] | (r) Chen, B.; Wu, L.-Z.; Tung, C.-H. Acc. Chem. Res. 2018, 51, 2512. |
| [12] | Speckmeier, E.; Fischer, T. G.; Zeitler, K. J. Am. Chem. Soc. 2018, 140, 15353. |
| [13] | Tam, W. J. Am. Chem. Soc. 2002, 124, 12630. |
| [14] | Hu, J.; Zhu, Z.; Xie, Z.; Le, Z. Chin. J. Org. Chem. 2022, 42, 978. (in Chinese) |
| [14] | ( 胡家榆, 祝志强, 谢宗波, 乐长高, 有机化学, 2022, 42, 978.) |
| [15] | Hedstrand, D. M.; Kruizinga, W. H.; Kellogg, R. M. Tetrahedron Lett. 1978, 19, 1255. |
| [16] | Sun, X.; Yu, S. Chem. Commun. 2016, 52, 10898. |
| [17] | Noto, N.; Koike, T.; Akita, M. Chem. Sci. 2017, 8, 6375. |
| [18] | Wang, H.; Xu, Q.; Yu, S. Org. Chem. Front. 2018, 5, 2224. |
| [19] | Varga, B.; Gonda, Z.; Tóth, B. L.; Kotschy, A.; Novák, Z. Eur. J. Inorg. Chem. 2020, 2020, 1466. |
| [20] | Chen, C.; Wang, Z.-J.; Lu, H.; Zhao, Y.; Shi, Z. Nat. Commun. 2021, 12, 4526. |
| [21] | Li, X.; Si, W.; Liu, Z.; Qian, H.; Wang, T.; Leng, S.; Sun, J.; Jiao, Y.; Zhang, X. Org. Lett. 2022, 24, 4070. |
| [22] | Yan, D.-M.; Xu, S.-H.; Qian, H.; Gao, P.-P.; Bi, M.-H.; Xiao, W.-J.; Chen, J.-R. ACS Catal. 2022, 12, 3279. |
| [23] | Donck, S.; Baroudi, A.; Fensterbank, L.; Goddard, J.-P.; Ollivier, C. Adv. Synth. Catal. 2013, 355, 1477. |
| [24] | (a) Cheng, Y.; Yuan, X.; Jiang, H.; Wang, R.; Ma, J.; Zhang, Y.; Yu, S. Adv. Synth. Catal. 2014, 356, 2859. |
| [24] | (b) Wang, H.; Cheng, Y.; Yu, S. Sci. China: Chem. 2016, 59, 195. |
| [24] | (c) Yu, S.; Zhang, Y.; Wang, R.; Jiang, H.; Cheng, Y.; Kadi, A.; Fun, H.-K. Synthesis 2014, 46, 2711. |
| [25] | Otsuka, S.; Nogi, K.; Rovis, T.; Yorimitsu, H. Chem. Asian. J. 2019, 14, 532. |
| [26] | Zhu, X.; Jiang, M.; Li, X.; Zhu, E.; Deng, Q.; Song, X.; Lv, J.; Yang, D. Org. Chem. Front. 2022, 9, 347. |
| [27] | (a) Berger, F.; Plutschack, M. B.; Riegger, J.; Yu, W.; Speicher, S.; Ho, M.; Frank, N.; Ritter, T. Nature 2019, 567, 223. |
| [27] | (b) Ritter, T.; Berger, F. Synlett 2021, 33, 339. |
| [27] | (c) Sang, R.; Korkis, S. E.; Su, W.; Ye, F.; Engl, P. S.; Berger, F.; Ritter, T. Angew. Chem., Int. Ed. 2019, 58, 16161. |
| [28] | Liang, L.; Niu, H. Y.; Li, R. L.; Wang, Y. F.; Yan, J. K.; Li, C. G.; Guo, H. M. Org. Lett. 2020, 22, 6842. |
| [29] | Lu, Y.; Liu, Q.; Wang, Z.-X.; Chen, X.-Y. Angew. Chem., Int. Ed. 2022, 61, e202116071. |
| [30] | Li, X.; Jiang, M.; Zhu, X.; Song, X.; Deng, Q.; Lv, J.; Yang, D. Org. Chem. Front. 2022, 9, 386. |
| [31] | Aukland, M. H.; ?iau?iulis, M.; West, A.; Perry, G. J. P.; Procter, D. J. Nat. Catal. 2020, 3, 163. |
| [32] | Mkrtchyan, S.; Iaroshenko, V. O. J. Org. Chem. 2021, 86, 4896. |
| [33] | Sun, K.; Shi, A.; Liu, Y.; Chen, X.; Xiang, P.; Wang, X.; Qu, L.; Yu, B. Chem. Sci. 2022, 13, 5659. |
| [34] | Wang, X.; Xun, X.; Song, H.; Liu, Y.; Wang, Q. Org. Lett. 2022, 24, 4580. |
| [35] | Engl, P. S.; Haring, A. P.; Berger, F.; Berger, G.; Perez-Bitrian, A.; Ritter, T. J. Am. Chem. Soc. 2019, 141, 13346. |
| [36] | Ye, F.; Berger, F.; Jia, H.; Ford, J.; Wortman, A.; Borgel, J.; Genicot, C.; Ritter, T. Angew. Chem., Int. Ed. 2019, 58, 14615. |
| [37] | Li, J.; Chen, J.; Sang, R.; Ham, W. S.; Plutschack, M. B.; Berger, F.; Chabbra, S.; Schnegg, A.; Genicot, C.; Ritter, T. Nat. Chem. 2020, 12, 56. |
| [38] | Huang, C.; Feng, J.; Ma, R.; Fang, S.; Lu, T.; Tang, W.; Du, D.; Gao, J. Org. Lett. 2019, 21, 9688. |
| [39] | Chen, C.; Wang, Z. J.; Lu, H.; Zhao, Y.; Shi, Z. Nat. Commun. 2021, 12, 4526. |
| [40] | Tian, Z.-Y.; Zhang, C.-P. Org. Chem. Front. 2022, 9, 2220. |
| [41] | Li, Q.; Huang, J.; Cao, Z.; Zhang, J.; Wu, J. Org. Chem. Front. 2022, 9, 3781. |
| [42] | He, F.-S.; Bao, P.; Tang, Z.; Yu, F.; Deng, W.-P.; Wu, J. Org. Lett. 2022, 24, 2955. |
| [43] | Cui, W.; Li, X.; Guo, G.; Song, X.; Lv, J.; Yang, D. Org. Lett. 2022, 24, 5391. |
/
| 〈 |
|
〉 |