

Chinese Journal of Organic Chemistry >
Recent Advances in the Synthesis of Fused Heterocyclic Compounds and Their Antitumor Activities
Received date: 2022-11-30
Revised date: 2023-02-15
Online published: 2023-03-01
Supported by
National Natural Science Foundation of China(22071063)
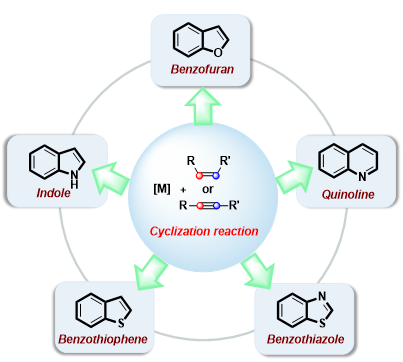
The unique physicochemical properties invest fused heterocyclic compounds with wide applications in the synthesis of natural products, drugs, superconducting materials, energy storage materials, polymer materials, organic dyes, etc. In recent years, the rapid development of transition metal-catalyzed reactions of unsaturated hydrocarbons has developed rapidly. Owing to the advantages of high step- and atom-economy, easy availability of raw starting materials, and efficient construction of carbon-carbon bonds or/and carbon-hetero bonds, it is a vital way to synthesize fused heterocyclic compounds. Herein, the recent progress on the reaction development of transition metal-catalyzed cyclizations involving unsaturated hydrocarbons for the synthesis of fused heterocyclic compounds including benzofurans, indoles, quinolines in the last five years has been summarized, as well as their applications in the field of medicinal chemistry with antitumor activities.
Kanghui Duan , Junlong Tang , Wanqing Wu . Recent Advances in the Synthesis of Fused Heterocyclic Compounds and Their Antitumor Activities[J]. Chinese Journal of Organic Chemistry, 2023 , 43(3) : 826 -854 . DOI: 10.6023/cjoc202211046
| [1] | Abd El Razik, H. A.; Wahab, A. E. Arch. Pharm. 2011, 344, 184. |
| [2] | Ahmed, O. M.; Mohamed, M. A.; Ahmed, R. R.; Ahmed, S. A. Eur. J. Med. Chem. 2009, 44, 3519. |
| [3] | An, Z.; Wu, M.; Kang, J.; Ni, J.; Qi, Z.; Yuan, B.; Yan, R. Eur. J. Org. Chem. 2018, 2018, 4812. |
| [4] | Blass, B. ACS Med. Chem. Lett. 2012, 3, 616. |
| [5] | Hassan, A. S.; Masoud, D. M.; Sroor, F. M.; Askar, A. A. Med. Chem. Res. 2017, 26, 2909. |
| [6] | Hassan, A. Y.; Mohamed, M. A.; Abdel-Aziem, A.; Hussain, A. O. Polycycl. Aromat. Compd. 2020, 40, 1280. |
| [7] | Komuraiah, B.; Ren, Y.; Xue, M.; Cheng, B.; Liu, J.; Liu, Y.; Chen, J. Chem. Biol. Drug Des. 2021, 97, 1109. |
| [8] | Liu, Y.; Zhang, X. H.; Ren, J.; Jin, G. Y. Synth. Commun. 2004, 34, 151. |
| [9] | Panda, S.; Roy, A.; Deka, S. J.; Trivedi, V.; Manna, D. ACS. Med. Chem. Lett. 2016, 7, 1167. |
| [10] | Tsyshevsky, R.; Smirnov, A. S.; Kuklja, M. M. J. Phys. Chem. C 2019, 123, 8688. |
| [11] | Zhang, H.; Wang, Q.; Huang, L.; Tian, Z.; Zhang, S.; Zhang, Y. Tetrahedron Lett. 2021, 72, 153070. |
| [12] | Fujihara, T. Asian J. Org. Chem. 2022, e202200535. |
| [13] | Gong, L.-Z.; Wang, P.-S.; Shen, M.-L. Synthesis 2018, 50, 956. |
| [14] | Li, B.-J.; Sun, X. Synthesis 2022, 54, 2103. |
| [15] | Shi, Z.; Li, N.; Lu, H.-K.; Chen, X.; Zheng, H.; Yuan, Y.; Ye, K.-Y. Curr. Opin. Electrochem. 2021, 28, 100713. |
| [16] | Shi, Y.; Xiao, T.; Xia, D.; Yang, W. Chin. J. Org. Chem. 2022, 42, 2715. (in Chinese) |
| [16] | (石云, 肖婷, 夏冬, 杨文超, 有机化学, 2022, 42, 2715.) |
| [17] | Dawood, K. M. Expert Opin. Ther. Pat. 2019, 29, 841. |
| [18] | Hofer, K. E.; Faber, K.; Muller, D. M.; Hauffe, T.; Wenger, U.; Kupferschmidt, H.; Rauber-Luthy, C. Ann. Emerg. Med. 2017, 69, 79. |
| [19] | Khanam, H.; Shamsuzzaman Eur. J. Med. Chem. 2015, 97, 483. |
| [20] | Radadiya, A.; Shah, A. Eur. J. Med. Chem. 2015, 97, 356. |
| [21] | Termentzi, A.; Khouri, I.; Gaslonde, T.; Prado, S.; Saint-Joanis, B.; Bardou, F.; Amanatiadou, E. P.; Vizirianakis, I. S.; Kordulakova, J.; Jackson, M.; Brosch, R.; Janin, Y. L.; Daffe, M.; Tillequin, F.; Michel, S. Eur. J. Med. Chem. 2010, 45, 5833. |
| [22] | Xu, Z.; Zhao, S.; Lv, Z.; Feng, L.; Wang, Y.; Zhang, F.; Bai, L.; Deng, J. Eur. J. Med. Chem. 2019, 162, 266. |
| [23] | Wu, W.; Yi, S.; Huang, W.; Luo, D.; Jiang, H. Org. Lett. 2017, 19, 2825. |
| [24] | Hu, W.; Li, M.; Jiang, G.; Wu, W.; Jiang, H. Org. Lett. 2018, 20, 3500. |
| [25] | Guo, S.; Li, P.; Guan, Z.; Cai, L.; Chen, S.; Lin, A.; Yao, H. Org. Lett. 2019, 21, 921. |
| [26] | Iqbal, N.; Iqbal, N.; Maiti, D.; Cho, E. J. Angew. Chem., Int. Ed. 2019, 58, 15808. |
| [27] | He, J.; Xue, Y.; Han, B.; Zhang, C.; Wang, Y.; Zhu, S. Angew. Chem., Int. Ed. 2020, 59, 2328. |
| [28] | Lin, Z.; Jin, Y.; Hu, W.; Wang, C. Chem. Sci. 2021, 12, 6712. |
| [29] | Zhou, F.; Li, C.; Li, M.; Jin, Y.; Jiang, H.; Zhang, Y.; Wu, W. Chem. Commun. 2021, 57, 4799. |
| [30] | Liu, Y.; Luo, W.; Xia, T.; Fang, Y.; Du, C.; Jin, X.; Li, Y.; Zhang, L.; Lei, W.; Wu, H. Org. Chem. Front. 2021, 8, 1732. |
| [31] | Chripkova, M.; Zigo, F.; Mojzis, J. Molecules 2016, 21, 1626. |
| [32] | Singh, T. P.; Singh, O. M. Mini-Rev. Med. Chem. 2018, 18, 9. |
| [33] | Warskulat, A. C.; Tatsis, E. C.; Dudek, B.; Kai, M.; Lorenz, S.; Schneider, B. ChemBioChem 2016, 17, 318. |
| [34] | Baeyer, A.; Knop, C. A. Ann. Pharm. 1866, 140, 1. |
| [35] | Kong, W.; Wang, Q.; Zhu, J. Angew. Chem., Int. Ed. 2017, 56, 3987. |
| [36] | Zhang, W.; Chen, P.; Liu, G. Angew. Chem., Int. Ed. 2017, 56, 5336. |
| [37] | Liu, Y.-Z.; Shang, S.-J.; Zhu, J.-Y.; Yang, W.-L.; Deng, W.-P. Adv. Synth. Catal. 2018, 360, 2191. |
| [38] | Zheng, X.; Yang, W.-L.; Liu, Y.-Z.; Wu, S.-X.; Deng, W.-P. Adv. Synth. Catal. 2018, 360, 2843. |
| [39] | Wang, K.; Ding, Z.; Zhou, Z.; Kong, W. J. Am. Chem. Soc. 2018, 140, 12364. |
| [40] | Loup, J.; Larin, E. M.; Lautens, M. Angew. Chem., Int. Ed. 2021, 60, 22345. |
| [41] | Chen, J.; Han, X.; Lu, X. Angew. Chem., Int. Ed. 2017, 56, 14698. |
| [42] | Kolli, M. K.; Shaik, N. M.; Chandrasekar, G.; Chidara, S.; Korupolu, R. B. New J. Chem. 2017, 41, 8187. |
| [43] | Li, J.; Li, C.; Ouyang, L.; Li, C.; Wu, W.; Jiang, H. Org. Biomol. Chem. 2017, 15, 7898. |
| [44] | Luo, Y. G.; Basha, R. S.; Reddy, D. M.; Xue, Y. J.; Chen, T. H.; Lee, C. F. Org. Lett. 2018, 20, 6872. |
| [45] | He, Y. P.; Wu, H.; Wang, Q.; Zhu, J. Angew. Chem., Int. Ed. 2020, 59, 2105. |
| [46] | Cheng, C.; Zuo, X.; Tu, D.; Wan, B.; Zhang, Y. Org. Lett. 2020, 22, 4985. |
| [47] | Fan, L.; Hao, J.; Yu, J.; Ma, X.; Liu, J.; Luan, X. J. Am. Chem. Soc. 2020, 142, 6698. |
| [48] | Wu, J.; Li, L.; Liu, M.; Bai, L.; Luan, X. Angew. Chem., Int. Ed. 2022, 61, e202113820. |
| [49] | Hu, X.-D.; Chen, Z.-H.; Zhao, J.; Sun, R.-Z.; Zhang, H.; Qi, X.; Liu, W.-B. J. Am. Chem. Soc. 2021, 143, 3734. |
| [50] | Arora, R.; Rodriguez, J. F.; Whyte, A.; Lautens, M. Angew. Chem., Int. Ed. 2022, 61, e202112288. |
| [51] | Baruah, S.; Saikia, P.; Duarah, G.; Gogoi, S. Org. Lett. 2018, 20, 3753. |
| [52] | Shang, Y.; Jonnada, K.; Yedage, S. L.; Tu, H.; Zhang, X.; Lou, X.; Huang, S.; Su, W. Chem. Commun. 2019, 55, 9547. |
| [53] | Xu, Y.; Shen, M.; Zhang, X.; Fan, X. Org. Lett. 2020, 22, 4697. |
| [54] | Voth, C. N.; Dake, G. R. Eur. J. Org. Chem. 2020, 2020, 744. |
| [55] | Wang, H. R.; Huang, E. H.; Luo, C.; Luo, W. F.; Xu, Y.; Qian, P. C.; Zhou, J. M.; Ye, L. W. Chem. Commun. 2020, 56, 4832. |
| [56] | Clarke, A. K.; Rossi-Ashton, J. A.; Taylor, R. J. K.; Unsworth, W. P. Tetrahedron 2020, 76, 131392. |
| [57] | Yuan, K.; Liu, L.; Chen, J.; Guo, S.; Yao, H.; Lin, A. Org. Lett. 2018, 20, 3477. |
| [58] | Rodriguez, J. F.; Marchese, A. D.; Lautens, M. Org. Lett. 2018, 20, 4367. |
| [59] | Irfan, A.; Batool, F.; Zahra Naqvi, S. A.; Islam, A.; Osman, S. M.; Nocentini, A.; Alissa, S. A.; Supuran, C. T. J. Enzyme Inhib. Med. Chem. 2020, 35, 265. |
| [60] | Pathak, N.; Rathi, E.; Kumar, N.; Kini, S. G.; Rao, C. M. Mini-Rev. Med. Chem. 2020, 20, 12. |
| [61] | Williams, N. S.; Gonzales, S.; Naidoo, J.; Rivera-Cancel, G.; Voruganti, S.; Mallipeddi, P.; Theodoropoulos, P. C.; Geboers, S.; Chen, H.; Ortiz, F.; Posner, B.; Nijhawan, D.; Ready, J. M. J. Med. Chem. 2020, 63, 9773. |
| [62] | Shen, G.; Yang, B.; Huang, X.; Hou, Y.; Gao, H.; Cui, J.; Cui, C.; Zhang, T. J. Org. Chem. 2017, 82, 3798. |
| [63] | Huang, Y.; Yan, D.; Wang, X.; Zhou, P.; Wu, W.; Jiang, H. Chem. Commun. 2018, 54, 1742. |
| [64] | Zhou, P.; Huang, Y.; Wu, W.; Yu, W.; Li, J.; Zhu, Z.; Jiang, H. Org. Biomol. Chem. 2019, 17, 3424. |
| [65] | Moon, S.; Kato, M.; Nishii, Y.; Miura, M. Adv. Synth. Catal. 2020, 362, 1669. |
| [66] | Assis, L. C.; de Castro, A. A.; de Jesus, J. P. A.; Nepovimova, E.; Kuca, K.; Ramalho, T. C.; La Porta, F. A. Sci. Rep. 2021, 11, 6397. |
| [67] | Narwal, S.; Kumar, S.; Verma, P. K. Res. Chem. Intermed. 2016, 43, 2765. |
| [68] | Horák, R.; Ko?istek, K.; ?am?ulová, V.; Slaninová, L.; Grepl, M.; Kvapil, L.; Funk, P.; Hradil, P.; Soural, M. J. Heterocycl. Chem. 2020, 57, 1605. |
| [69] | Kaishap, P. P.; Duarah, G.; Chetia, D.; Gogoi, S. Org. Biomol. Chem. 2017, 15, 3491. |
| [70] | Zhang, X.; Han, X.; Chen, J.; Lu, X. Tetrahedron 2017, 73, 1541. |
| [71] | Cen, J.; Li, J.; Zhang, Y.; Zhu, Z.; Yang, S.; Jiang, H. Org. Lett. 2018, 20, 4434. |
| [72] | Ranjith Kumar, G.; Kumar, R.; Rajesh, M.; Sridhar Reddy, M. Chem. Commun. 2018, 54, 759. |
| [73] | Wang, X.; He, D.; Huang, Y.; Fan, Q.; Wu, W.; Jiang, H. J. Org. Chem. 2018, 83, 5458. |
| [74] | Mule, R. D.; Shaikh, A. C.; Gade, A. B.; Patil, N. T. Chem. Commun. 2018, 54, 11909. |
| [75] | Shaikh, A. C.; Banerjee, S.; Mule, R. D.; Bera, S.; Patil, N. T. J. Org. Chem. 2019, 84, 4120. |
| [76] | Chintawar, C. C.; Mane, M. V.; Tathe, A. G.; Biswas, S.; Patil, N. T. Org. Lett. 2019, 21, 7109. |
| [77] | De Abreu, M.; Tang, Y.; Brachet, E.; Selkti, M.; Michelet, V.; Belmont, P. Org. Biomol. Chem. 2021, 19, 1037. |
| [78] | Liao, J.; Fan, L.; Guo, W.; Zhang, Z.; Li, J.; Zhu, C.; Ren, Y.; Wu, W.; Jiang, H. Org. Lett. 2017, 19, 1008. |
| [79] | Cai, S.; Lin, S.; Yi, X.; Xi, C. J. Org. Chem. 2017, 82, 512. |
| [80] | Zhang, Z. M.; Xu, B.; Wu, L.; Wu, Y.; Qian, Y.; Zhou, L.; Liu, Y.; Zhang, J. Angew. Chem., Int. Ed. 2019, 58, 14653. |
| [81] | Tyagi, A.; Reshi, N. U. D.; Daw, P.; Bera, J. K. Dalton. Trans. 2020, 49, 15238. |
| [82] | Li, J.; Tang, H.; Lin, Z.; Yang, S.; Wu, W.; Jiang, H. Org. Biomol. Chem. 2020, 18, 4071. |
| [83] | Zhang, P.; Wang, C.; Cui, M.; Du, M.; Li, W.; Jia, Z.; Zhao, Q. Org. Lett. 2020, 22, 1149. |
| [84] | Pan, Q.; Ping, Y.; Wang, Y.; Guo, Y.; Kong, W. J. Am. Chem. Soc. 2021, 143, 10282. |
| [85] | Bajohr, J.; Diallo, A. G.; Whyte, A.; Gaillard, S.; Renaud, J. L.; Lautens, M. Org. Lett. 2021, 23, 2797. |
| [86] | Dong, J.; Bao, L.; Hu, Z.; Ma, S.; Zhou, X.; Hao, M.; Li, N.; Xu, X. Org. Lett. 2018, 20, 1244. |
| [87] | Guo, S.; Pan, R.; Guan, Z.; Li, P.; Cai, L.; Chen, S.; Lin, A.; Yao, H. Org. Lett. 2019, 21, 6320. |
| [88] | Saha, R.; Arunprasath, D.; Sekar, G. J. Catal. 2019, 377, 673. |
| [89] | Hong, F. L.; Wang, Z. S.; Wei, D. D.; Zhai, T. Y.; Deng, G. C.; Lu, X.; Liu, R. S.; Ye, L. W. J. Am. Chem. Soc. 2019, 141, 16961. |
| [90] | Liu, X.; Wang, Z. S.; Zhai, T. Y.; Luo, C.; Zhang, Y. P.; Chen, Y. B.; Deng, C.; Liu, R. S.; Ye, L. W. Angew. Chem., Int. Ed. 2020, 59, 17984. |
| [91] | Whyte, A.; Bajohr, J.; Arora, R.; Torelli, A.; Lautens, M. Angew. Chem., Int. Ed. 2021, 60, 20231. |
| [92] | Wu, Y.; Xu, B.; Zhao, G.; Pan, Z.; Zhang, Z. M.; Zhang, J. Chin. J. Chem. 2021, 39, 3255. |
| [93] | Li, J. F.; Xu, W. W.; Wang, R. H.; Li, Y.; Yin, G.; Ye, M. Nat. Commun. 2021, 12, 3070. |
| [94] | Jin, L. P.; Xie, Q.; Huang, E. F.; Wang, L.; Zhang, B. Q.; Hu, J. S.; Wan, D. C.; Jin, Z.; Hu, C. Bioorg. Chem. 2020, 95, 103566. |
| [95] | Li, Q.; Jian, X. E.; Chen, Z. R.; Chen, L.; Huo, X. S.; Li, Z. H.; You, W. W.; Rao, J. J.; Zhao, P. L. Bioorg. Chem. 2020, 102, 104076. |
| [96] | Romagnoli, R.; Baraldi, P. G.; Sarkar, T.; Carrion, M. D.; Cruz-Lopez, O.; Lopez Cara, C.; Tolomeo, M.; Grimaudo, S.; Di Cristina, A.; Pipitone, M. R.; Balzarini, J.; Gambari, R.; Ilaria, L.; Saletti, R.; Brancale, A.; Hamel, E. Bioorg. Med. Chem. 2008, 16, 8419. |
| [97] | Hu, H.; Li, Q.; Li, Z.; Li, W.; Zhang, F.; Chen, H. Chem. Res. Appl. 2019, 31, 511. (in Chinese) |
| [97] | (胡鸿雨, 李全涛, 李正辉, 李威威, 张发饶, 陈红征, 化学研究与应用, 2019, 31, 511.) |
| [98] | Iacopetta, D.; Catalano, A.; Ceramella, J.; Barbarossa, A.; Carocci, A.; Fazio, A.; La Torre, C.; Caruso, A.; Ponassi, M.; Rosano, C.; Franchini, C.; Sinicropi, M. S. Bioorg. Chem. 2020, 105, 104440. |
| [99] | Fu, D. J.; Cui, X. X.; Zhu, T.; Zhang, Y. B.; Hu, Y. Y.; Zhang, L. R.; Wang, S. H.; Zhang, S. Y. Bioorg. Chem. 2021, 107, 104634. |
| [100] | Jia, H. W.; Yang, H. L.; Xiong, Z. L.; Deng, M. H.; Wang, T.; Liu, Y.; Cheng, M. Bioorg. Chem. 2022, 129, 106213. |
| [101] | Abdelgawad, M. A.; Belal, A.; Ahmed, O. M. J. Chem. Pharm. Res. 2013, 5, 318. |
| [102] | Seenaiah, D.; Reddy, P. R.; Reddy, G. M.; Padmavathi, V.; Krishna, N. S. Eur. J. Med. Chem. 2014, 77, 1. |
| [103] | Gabr, M. T.; El-Gohary, N. S.; El-Bendary, E. R.; El-Kerdawy, M. M.; Ni, N. Chin. Chem. Lett. 2016, 27, 380. |
| [104] | Diao, P. C.; Lin, W. Y.; Jian, X. E.; Li, Y. H.; You, W. W.; Zhao, P. L. Eur. J. Med. Chem. 2019, 179, 196. |
| [105] | Abd El-Meguid, E. A.; Mohi El-Deen, E. M.; Moustafa, G. O.; Awad, H. M.; Nossier, E. S. Bioorg. Chem. 2022, 119, 105504. |
| [106] | Vennila, K. N.; Sunny, D.; Madhuri, S.; Ciattini, S.; Chelazzi, L.; Elango, K. P. Bioorg. Chem. 2018, 81, 184. |
| [107] | Abdelsalam, E. A.; Zaghary, W. A.; Amin, K. M.; Abou Taleb, N. A.; Mekawey, A. A. I.; Eldehna, W. M.; Abdel-Aziz, H. A.; Hammad, S. F. Bioorg. Chem. 2019, 89, 102985. |
| [108] | Akkachairin, B.; Rodphon, W.; Reamtong, O.; Mungthin, M.; Tummatorn, J.; Thongsornkleeb, C.; Ruchirawat, S. Bioorg. Chem. 2020, 98, 103732. |
/
| 〈 |
|
〉 |