

Chinese Journal of Organic Chemistry >
Recent Advances of Minisci Reactions in Aqueous Solution
Received date: 2024-01-15
Revised date: 2024-03-05
Online published: 2024-03-28
Supported by
National Natural Science Foundation of China(31972850)
Minisci reaction is a convenient method for the functionalization of C—H in electron-defficient N-heterocycles. Different substituents could be introduced onto heterocycles through Minisci reaction, such as alkyl, acyl, silyl and alkylthio, etc. Water is eco-friendly and an ideal medium in organic synthesis, which has been applied in Minisci reaction. Recent progress in Minisci reaction in aqueous solution is reviewed herein. Minisci reaction is a convenient method for the functionalization of electron-defficient N-heterocycles. Different substituents could be introduced onto heterocycles through Minisci reaction, such as alkyl, acyl, silyl and alkylthio, boryl, etc. Thus, Minisci reaction is a powerful toolbox for the synthesis of substituted N-heterocylces. Water is eco-friendly and an ideal medium in organic synthesis, which has been applied in Minisci reaction. Although water was used from the discovery of Minisci reaction, Minisci reaction in aqueous solution is still underdeveloped. The recent progress in Minisci reaction in aqueous solution, including alkylation, arylation and acylation, is reviewed according to different radical precursors.
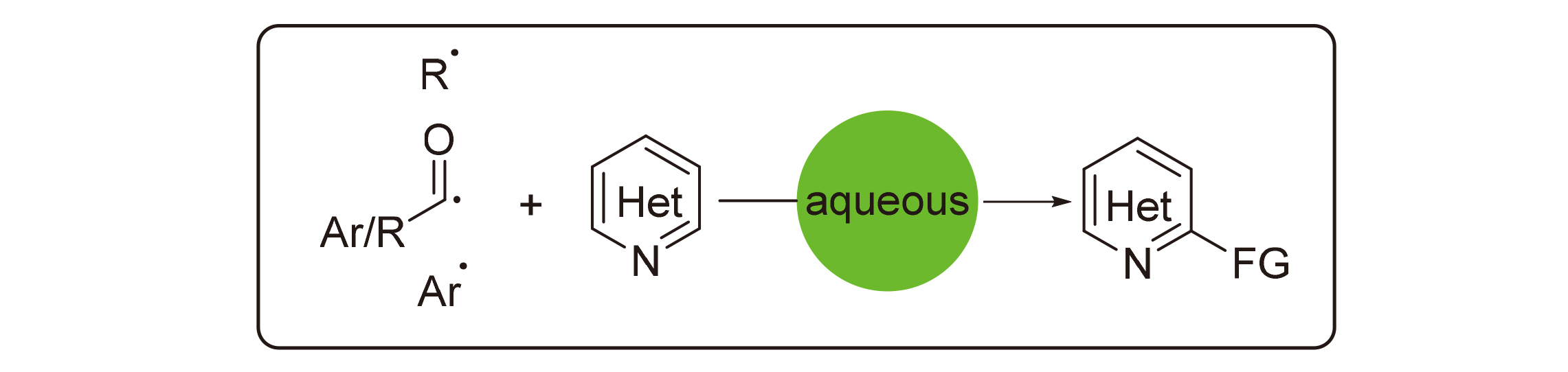
Key words: Minisci reaction; radical; aqueous solution; heterocycle
Wengui Wang , Shoufeng Wang . Recent Advances of Minisci Reactions in Aqueous Solution[J]. Chinese Journal of Organic Chemistry, 2024 , 44(7) : 2136 -2146 . DOI: 10.6023/cjoc202401015
| [1] | For selected reviews, see: (a) Liu, C.-X.; Yin, S.-Y.; Zhao, F.; Yang, H.; Feng, Z.; Gu, Q.; You, S.-L. Chem. Rev. 2023, 123, 10079. |
| [1] | (b) Zhang, J.; Rueping, M. Chem. Soc. Rev. 2023, 52, 4099. |
| [1] | (c) Josephitis, C. M.; Nguyen, H. M. H.; McNally, A. Chem. Rev. 2023, 123, 7655. |
| [1] | (d) Roy, S.; Panja, S.; Sahoo, S. R.; Chatterjee, S.; Maiti, D. Chem. Soc. Rev. 2023, 52, 2391. |
| [1] | (e) Bellotti, P.; Huang, H.-M.; Faber, T.; Glorius, F. Chem. Rev. 2023, 123, 4237. |
| [2] | Lu, M.-Z.; Goh, J.; Maraswami, M.; Jia, Z.; Tian, J.-S.; Loh, T.-P. Chem. Rev. 2022, 122, 17479. |
| [3] | For selected reviews and articles, see: (a) Baco?, P. D.; Lahdenper?, A. S. K.; Phipps, R. J. Acc. Chem. Res. 2023, 56, 2037. |
| [3] | (b) Dong, J.; Liu, Y.; Wang, Q. Chin. J. Org. Chem. 2021, 41, 3771. (in Chinese) |
| [3] | (董建洋, 刘玉秀, 汪清民, 有机化学, 2021, 41, 3771.) |
| [3] | (c) Meng, W.; Xu, K.; Guo, B.; Zeng, C. Chin. J. Org. Chem. 2021, 41, 2621. (in Chinese) |
| [3] | (孟薇, 徐坤, 郭兵兵, 曾程初, 有机化学, 2021, 41, 2621.) |
| [3] | (d) Wang, W.; Wang, S. Curr. Org. Chem. 2021, 25, 894. |
| [3] | (e) Zheng, H.; Lu, H.; Su, C.; Yang, R.; Zhao, L.; Liu, X.; Cao, H. Chin. J. Chem. 2023, 41, 193. |
| [4] | Minisci, F.; Vismara, E.; Fontana, F.; Morini, G.; Serravalle, M. J. Org. Chem. 1987, 52, 730 |
| [5] | Minisci, F. Tetrahedron 1971, 27, 3575. |
| [6] | Minisci, F.; Vismara, E. Tetrahedron Lett. 1986, 26, 4803. |
| [7] | Shore, D. G. M.; Wasik, K. A.; Lyssikatos, J. P.; Estrada, A. A. Tetrahedron Lett. 2015, 56, 4063. |
| [8] | Tung, T. T.; Christensen, S. B.; Nielsen, J. Chem.-Eur. J. 2017, 23, 18125. |
| [9] | Xie, X.; Zhang, Y.; Hao, J.; Wan, W. Org. Biomol. Chem. 2020, 18, 400. |
| [10] | Mai, D. N.; Baxter, R. D. Org. Lett. 2016, 18, 3738. |
| [11] | Galloway, J. D.; Mai, D. N.; Baxter, R. D. Org. Lett. 2017, 19, 5772. |
| [12] | Wang, W.; Song, Y.; Xing, S.; Li, J.; Feng, W.; Qu, X.; Wang, S. ChemistrySelect 2023, 8, e202300958. |
| [13] | Sutherland, D. R.; Veguillas, M.; Oates, C. L.; Lee, A.-L. Org. Lett. 2018, 20, 6863. |
| [14] | Jafarpour, F.; Darvishmolla, M.; Azaddoost, N.; Mohaghegh, F. New J. Chem. 2019, 43, 9328. |
| [15] | Dong, J.; Wang, Z.; Wang, X.; Song, H.; Liu, Y.; Wang, Q. J. Org. Chem. 2019, 84, 7532. |
| [16] | Shao, M.; Liang, H.; Liu, Y.-L.; Qin, W.; Li, Z. Asian J. Org. Chem. 2020, 9, 782. |
| [17] | Jin, J.; MacMillan, D. W. C. Angew. Chem., Int. Ed. 2015, 54, 1565. |
| [18] | Devariab, S.; Shah, B. A. Chem. Commun. 2016, 52, 1490. |
| [19] | McCallum, T.; McCallum, L.-A.; McCallum, A.; Barriault, L. Synlett 2016, 27, 1282. |
| [20] | Bohman, B.; Berntsson, B.; Dixon, R. C. M.; Stewart, C. D.; Barrow, R. A. Org. Lett. 2014, 16, 2787. |
| [21] | Xu, D.; Huang, F.; Tang, L.; Zhang, X.; Zhang, W. Chin. J. Org. Chem. 2022, 42, 1493. (in Chinese) |
| [21] | (徐东平, 黄飞, 汤琳, 张新明, 张武, 有机化学, 2022, 42, 1493.) |
| [22] | Lu, S.-C.; Li, H.-S.; Xu, S.; Duan, G.-Y. Org. Biomol. Chem. 2017, 15, 324. |
| [23] | Wu, X.; Wang, M.; Huan, L.; Wang, D.; Wang, J.; Zhu, C. Angew. Chem., Int. Ed. 2018, 57, 1640. |
| [24] | (a) Wang, Z.; Ji, X.; Zhao, J.; Huang, H. Green Chem. 2019, 21, 5512. |
| [24] | (b) Ji, X.; Liu, Q.; Wang, Z.; Wang, P.; Deng, G.-J.; Huang, H. Green Chem. 2020, 22, 8233. |
| [25] | Bosset, C.; Beucher, H.; Bretel, G.; Pasquier, E.; Queguiner, L.; Henry, C.; Vos, A.; Edwards, J. P.; Meerpoel, L.; Berthelot, D. Org. Lett. 2018, 20, 6003. |
| [26] | Santos, M. S.; Cybularczyk-Cecotka, M.; K?nig, B.; Giedyk, M. Chem. Eur. J. 2020, 26, 15323. |
| [27] | Molander, G. A.; Colombel, V.; Braz, V. A. Org. Lett. 2011, 13, 1852. |
| [28] | Presset, M.; Fleury-Brégeot, N.; Oehlrich, D.; Rombouts, F.; Molander, G. A. J. Org. Chem. 2013, 78, 4615. |
| [29] | Matsui, J. K.; Molander, G. A. Org. Lett. 2017, 19, 950. |
| [30] | Matsui, J. K.; Primer, D. N.; Molander, G. A. Chem. Sci. 2017, 8, 3512. |
| [31] | Yan, H.; Hou, Z.-W.; Xu, H.-C. Angew. Chem., Int. Ed. 2019, 58, 4592. |
| [32] | Gutie?rrez-Bonet, á.; Remeur, C.; Matsui, J. K.; Molander, G. A. J. Am. Chem. Soc. 2017, 139, 12251. |
| [33] | Ji, Y.; Brueckl, T.; Baxter, R. D.; Fujiwara, Y.; Seiple, I. B.; Su, S.; Blackmond, D. G.; Baran, P. S. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 14411. |
| [34] | (a) Fujiwara, Y.; Dixon, J. A.; Rodriguez, R. A.; Baxter, R. D.; Dixon, D. D.; Collins, M. R.; Blackmond, D. G.; Baran, P. S. J. Am. Chem. Soc. 2012, 134, 1494. |
| [34] | (b) O’Hara, F.; Baxter, R. D.; O’Brien, A. G.; Collins, M. R.; Dixon, J. A.; Fujiwara, Y.; Ishihara, Y.; Baran, P. S. Nat. Protoc. 2013, 8, 1042. |
| [35] | Lytkinaa, M. A.; Eliseenkovb, E. V.; Boyarskiib, V. P.; Petrovb, A. A. Russ. J. Org. Chem. 2017, 53, 533. |
| [36] | Zhou, Q.; Ruffoni, A.; Gianatassio, R.; Fujiwara, Y.; Sella, E.; Shabat, D.; Baran, P. S. Angew. Chem., nt. Ed. 2013, 52, 3949. |
| [37] | Gianatassio, R.; Kawamura, S.; Eprile, C. L.; Foo, K.; Ge, J.; Burns, A. C.; Collins, M. R.; Baran, P. S. Angew. Chem., Int. Ed. 2014, 53, 9851. |
| [38] | Jia, X.-M.; Wei, L.; Chen, F.; Tang, R.-Y. RSC Adv. 2015, 5, 29766. |
| [39] | Seiple, I. B.; Su, S.; Rodriguez, R. A.; Gianatassio, R.; Fujiwara, Y.; Sobel, A. L.; Baran, P. S. J. Am. Chem. Soc. 2010, 132, 13194. |
| [40] | Patel, N. R.; Flowers, R. A. J. Am. Chem. Soc. 2013, 135, 4672. |
| [41] | Baxter, R. D.; Liang, Y.; Hong, X.; Brown, T. A.; Zare, R. N.; Houk, K. N.; Baran, P. S.; Blackmond, D. G. ACS Cent. Sci. 2015, 1, 456. |
| [42] | Biaco, J. L.; Jones, S. L.; Barker, T. J. Heterocycles 2016, 92, 1687. |
| [43] | Xue, D.; Jia, Z.-H.; Zhao, C.-J.; Zhang, Y.-Y.; Wang, C.; Xiao, J. Chem.-Eur. J. 2014, 20, 2960. |
| [44] | Wang, R.; Falck, J. R. Org. Chem. Front. 2014, 1, 1029. |
| [45] | Fontana, F.; Minisci, F.; Barbosa, M. C. N.; Vismara, E. J. Org. Chem. 1991, 56, 2866. |
| [46] | Zeng, X.; Liu, C.; Wang, X.; Zhang, J.; Wang, X.; Hu, Y. Org. Biomol. Chem. 2017, 15, 8929. |
| [47] | Regan, C. F.; Pierre, F.; Schwaebe, M. K.; Haddach, M.; Jung, M. E.; Ryckman, D. M. Synlett 2012, 23, 443. |
| [48] | Wang, X.-Z.; Zeng, C.-C. Tetrahedron 2019, 75, 1425. |
| [49] | Manna, S.; Prabhu, K. R. J. Org. Chem. 2019, 84, 5067. |
| [50] | Caronna, T.; Gardini, G. P.; Minisci, F. J. Chem. Soc. D 1969, 201. |
| [51] | (a) Caronna, T.; Fronza, G.; Minisci, F.; Porta, O. J. Chem. Soc., Perkin Trans. 2 1972, 2035. |
| [51] | (b) Minisci, F.; Citterio, A.; Vismara, E.; Giordano, C. Tetrahedron 1986, 41, 4157. |
| [52] | Song, Y.; Yu, Z.; Wang, W.; Wang, S. Tetrahedron 2023, 141, 133518. |
| [53] | Sultan, S.; Ahmad Rizvi, M. A.; Kumar, J.; Shah, B. A. Chem. Eur. J. 2018, 24, 10617. |
| [54] | Sharma, S.; Kumar, M.; Vishwakarma, R. A.; Verma, M. K.; Singh, P. P. J. Org. Chem. 2018, 83, 12420. |
/
| 〈 |
|
〉 |