

Chinese Journal of Organic Chemistry >
Recent Progress in the Total Synthesis of abeo-Steroids
† The authors contributed equally to this work
Received date: 2025-06-17
Revised date: 2025-07-24
Online published: 2025-09-04
Supported by
National Natural Science Foundation of China(22271137)
Guangdong Basic and Applied Basic Research Foundation(2021A1515010164)
Research on steroids play a significant role in organic chemistry, medicinal chemistry of natural products, and drug discovery. To date, more than 100 steroidal drugs have been approved by U.S. Food and Drug Administration (FDA) for the treatment of cancer, inflammation, pain, and heart failure. Recently, more and more abeo-steroids have been isolated. Due to their novel structures and important biological activities, abeo-steroids have attracted considerable attention from the synthetic community. In general, abeo-steroids are complex and diverse, containing a medium ring (seven-membered ring) or bridged ring system and possessing multiple stereogenic centers. All these structural features present considerable synthetic challenges. The latest progress in the total synthesis of abeo-steroids in the past eight years is summarized, focusing on the innovative synthetic strategies and methods. Finally, a brief outlook for the future development of abeo-steroids and related field is discussed.
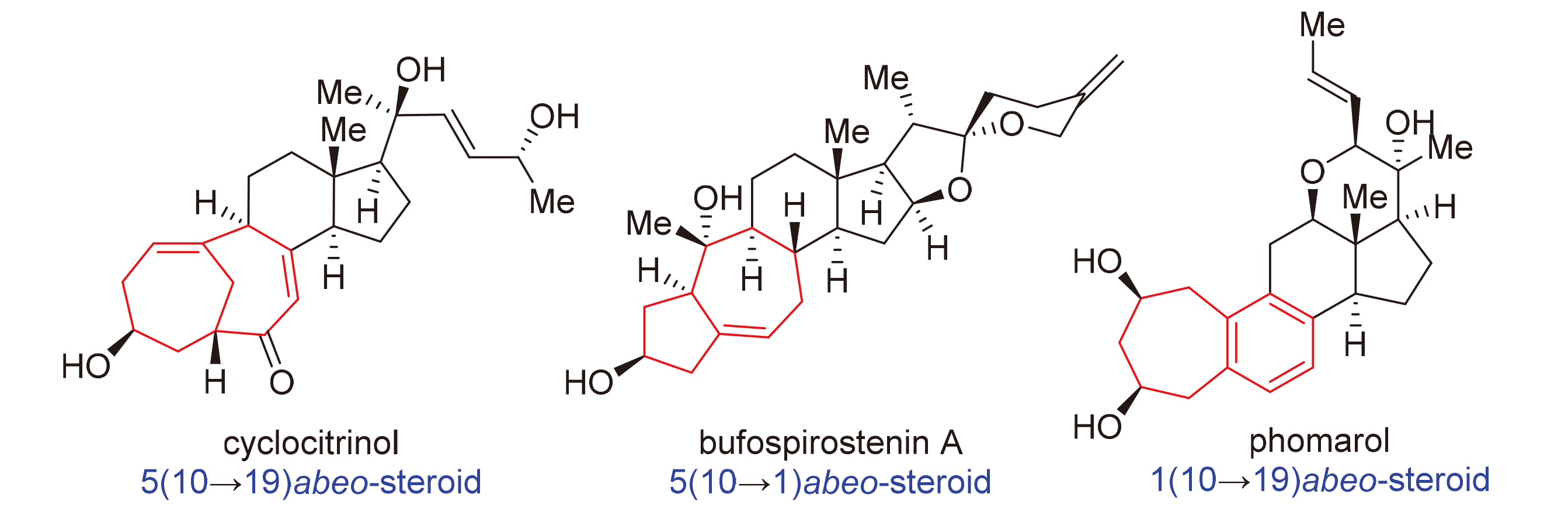
Key words: steroid; abeo-steroid; total synthesis; seven-membered ring; bridged ring system
Lilin Zhao , Zhaoqi Li , Liping Zhong , Long Min . Recent Progress in the Total Synthesis of abeo-Steroids[J]. Chinese Journal of Organic Chemistry, 2026 , 46(1) : 21 -38 . DOI: 10.6023/cjoc202506022
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
|
| [47] |
During our submission of this manuscript, an elegant review on synthesis of steroid natural products by Gui and co-workers was just published online, see: Wang, Y.; Gui, J. Chem. Soc. Rev. 2025, 54, 6807.
|
/
| 〈 |
|
〉 |