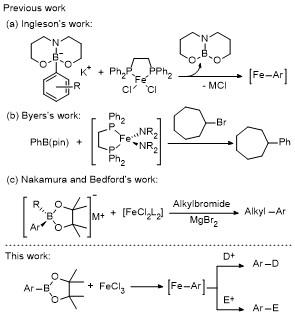
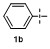
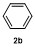
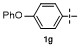
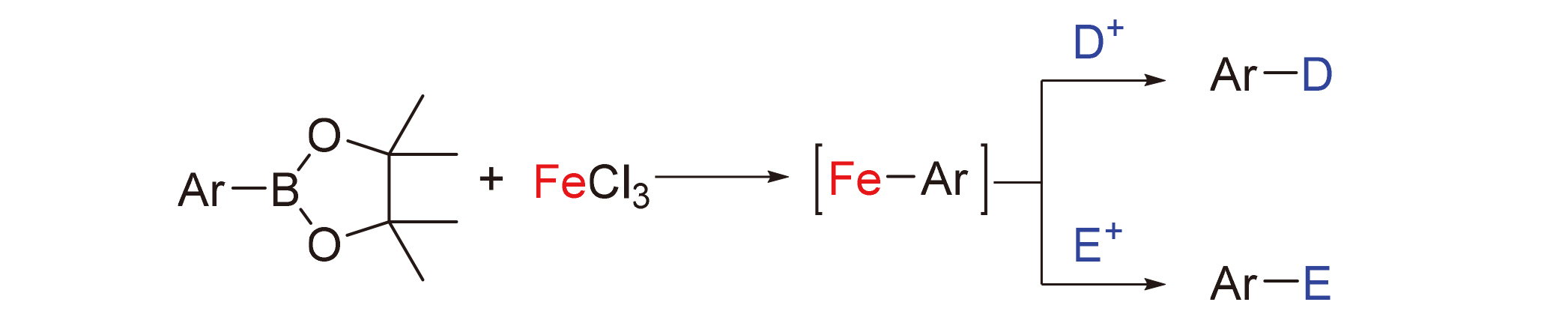1 Introduction
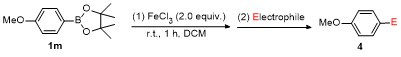
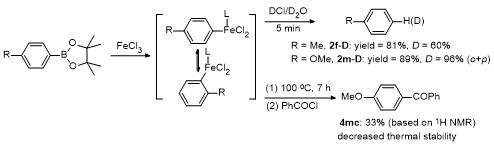
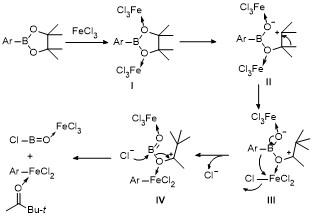
2 Results and discussion
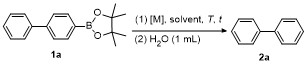
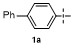
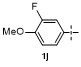
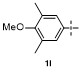
Table 1 Condition optimizations for C—B bond cleavage |
| Entry | [M] (equiv.) | Solvent | T/℃ | t/min | Yielda/% |
|---|---|---|---|---|---|
| 1 | FeCl3 (2.0) | CH2Cl2 | 100 | 5 | 84 |
| 2 | AlCl3 (2.0) | CH2Cl2 | 100 | 5 | 15 |
| 3 | CuCl2 (2.0) | CH2Cl2 | 100 | 30 | N.D.b |
| 4 | Fe2(SO4)3 (1.0) | CH2Cl2 | 100 | 180 | 7 |
| 5 | Fe(acac)3 (2.0) | CH2Cl2 | 100 | 60 | N.D. |
| 6 | Fe(OAc)2 (2.0) | CH2Cl2 | 100 | 60 | N.D. |
| 7 | FeCl2 (2.0) | CH2Cl2 | 100 | 60 | N.D. |
| 8 | Fe2O3 (2.0) | CH2Cl2 | 100 | 60 | N.D. |
| 9 | FeCl3 (2.0) | THF | 100 | 120 | N.D. |
| 10 | FeCl3 (2.0) | DMF | 100 | 120 | N.D. |
| 11 | FeCl3 (2.0) | CHCl3 | 100 | 90 | 61 |
| 12 | FeCl3 (1.0) | CH2Cl2 | 100 | 90 | 18 |
| 13 | FeCl3 (2.0) | CH2Cl2 | 25 | 30 | 87 |
| 14c | FeCl3 (2.0) | CH2Cl2 | 25 | 60 | 49 |
| 15d | FeCl3 (2.0) | CH2Cl2 | 25 | 60 | <5 |
a Isolated yield. b N.D.=not detected. c 4-Biphenylboronic acid was used as the substrates. d Diisopropyl[1'-biphenyl]-4-ylboronate was used as the subs- trates. |
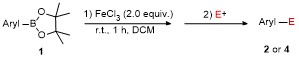
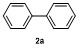
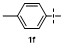
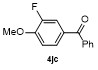
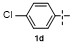
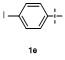
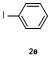
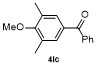
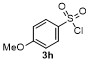
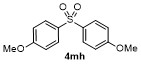
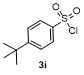
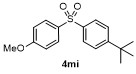
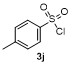
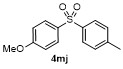
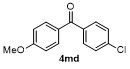
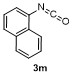
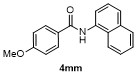
Table 2 Scope of arylboronatesa |
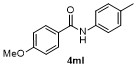
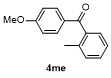
| Entry | Aryl | Electrophile | Product | Yieldb/% of 2 | Entry | Aryl | Electrophile | Product | Yieldb/% of 4 |
|---|---|---|---|---|---|---|---|---|---|
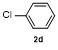
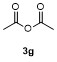
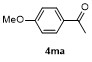
| 1 | | H2O | | 87 | 6 | | | | 51 |
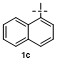
| 2c | | H2O | | 85d | 7 | | | | 68 |
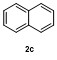
| 3 | | H2O | | 64e | 8 | | | | 63 |
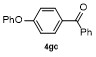
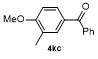
| 4f | | H2O | | 58d | 9 | | | | 66 |
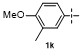
| 5g | | H2O | | 52d | 10 | | | | 86 |
a Condition: 0.2 mmol of 1, at 25 ℃ for 1 h in 2 mL of CH2Cl2, followed 2.0 equiv. of electrophile (except H2O), stirring for 4 h at 25 ℃. b Isolated yields. c At 60 ℃ for 1 h. d GC yield using tetradecane as internal standard. e NMR yield using mesitylene as internal standard. f 3 h at 100 ℃. g 4 h, at 100 ℃. |
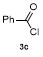
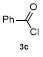
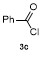
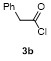
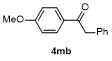
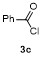
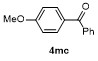
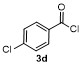
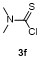
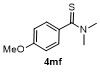
Table 3 Scope of electrophilesa |
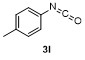
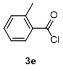
| Entry | E+ | Product | Yieldb/% | Entry | E+ | Product | Yieldb/% |
|---|---|---|---|---|---|---|---|
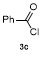
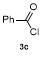
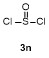
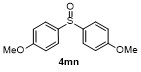
| 1 | | | 68 | 8 | | | 60 |
| 2 | | | 65 | 9 | | | 23 |
| 3 | | | 81 | 10 | | | 21 |
| 4 | | | 81 | 11 | | | 51 |
| 5 | | | 79 | 12 | | | 30 |
| 6 | | | 15 | 13 | | | 62 |
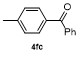
| 7 | | | 85 | 14 | | | 55 |
a Condition: 0.2 mmol of 1m, 2.0 equiv. of FeCl3, 2.0 equiv. of electrophiles. b Isolated yields. |