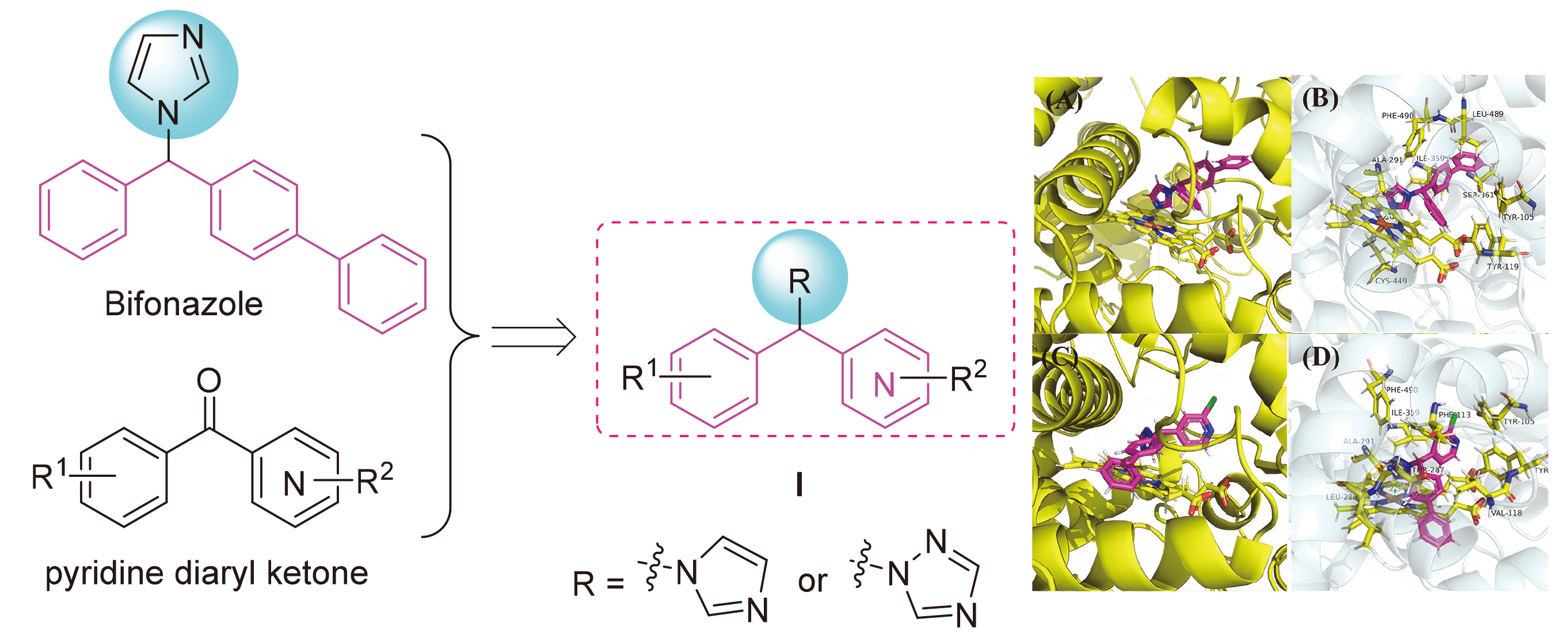
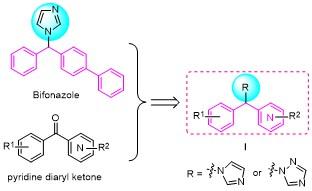
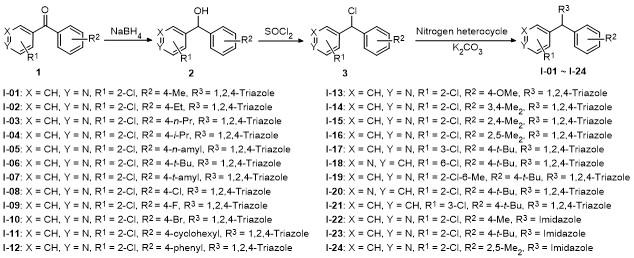
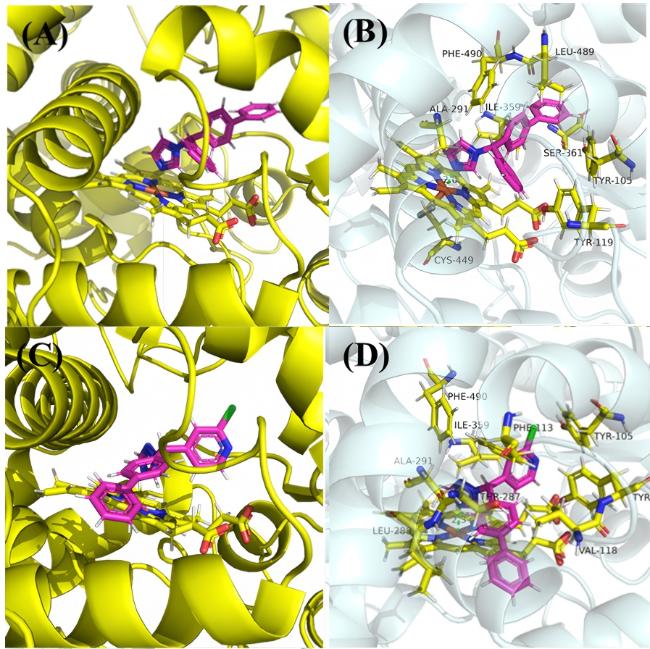
合成方法参考已报道文献[
34]. 将12.60 mmol硼氢化钠缓慢加入到12.60 mmol原料
1的50 mL甲醇溶液中, 室温下搅拌1 h. 向反应体系中加入1.5 mol/L盐酸50 mL), 用乙酸乙酯(50 mL×3)萃取, 合并有机相, 减压浓缩得到中间体
2.
(2-氯吡啶-4-基)(对甲苯基)甲醇(2-1): 白色固体, m.p. 140~141 ℃, 收率68%. 1H NMR (500 MHz, DMSO-d6) δ: 8.30 (d, J=5.1 Hz, 1H), 7.47 (s, 1H), 7.36 (dd, J=5.2, 1.4 Hz, 1H), 7.30~7.11 (m, 4H), 6.20 (d, J=4.1 Hz, 1H), 5.71 (d, J=4.1 Hz, 1H), 2.25 (s, 3H).
(2-氯吡啶-4-基)(4-乙基苯基)甲醇(2-2): 白色固体, m.p. 102~103 ℃, 收率82%. 1H NMR (500 MHz, DMSO-d6) δ: 8.31 (d, J=5.1 Hz, 1H), 7.48 (s, 1H), 7.37 (dd, J=5.1, 1.4 Hz, 1H), 7.32~7.14 (m, 4H), 6.20 (s, 1H), 5.72 (s, 1H), 2.55 (q, J=7.6 Hz, 2H), 1.13 (t, J=7.6 Hz, 3H).
(2-氯吡啶-4-基)(4-丙基苯基)甲醇(2-3): 白色固体, m.p. 102~103 ℃, 收率83%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.50 (s, 1H), 7.38 (dd, J=5.2, 1.4 Hz, 1H), 7.33~7.13 (m, 4H), 6.21 (s, 1H), 5.73 (s, 1H), 2.50 (t, J=15.3 Hz, 2H), 1.60~1.49 (m, 2H), 0.86 (t, J=7.3 Hz, 3H).
(2-氯吡啶-4-基)(4-异丙基苯基)甲醇(2-4): 白色固体, m.p. 69~70 ℃, 收率86%. 1H NMR (500 MHz, DMSO-d6) δ: 8.31 (d, J=5.1 Hz, 1H), 7.50 (s, 1H), 7.38 (dd, J=5.2, 1.4 Hz, 1H), 7.33~7.16 (m, 4H), 6.19 (d, J=4.0 Hz, 1H), 5.72 (d, J=4.0 Hz, 1H), 2.88~2.79 (m, 1H), 1.16 (d, J=7.0 Hz, 6H).
(2-氯吡啶-4-基)(4-戊基苯基)甲醇(2-5): 白色固体, m.p. 73~74 ℃, 收率90%. 1H NMR (500 MHz, DMSO- d6) δ: 8.31 (d, J=5.1 Hz, 1H), 7.49 (s, 1H), 7.38 (dd, J=5.2, 1.4 Hz, 1H), 7.32~7.13 (m, 4H), 6.21 (d, J=4.0 Hz, 1H), 5.72 (d, J=4.1 Hz, 1H), 2.52 (t, J=7.8 Hz, 2H), 1.57~1.48 (m, 2H), 1.34~1.19 (m, 4H), 0.84 (t, J=7.0 Hz, 3H).
(2-氯吡啶-4-基)(4-叔丁基苯基)甲醇(2-6): 白色固体, m.p. 133~134 ℃, 收率84%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.51 (s, 1H), 7.39 (dd, J=5.2, 1.4 Hz, 1H), 7.37~7.30 (m, 4H), 6.20 (d, J=4.0 Hz, 1H), 5.73 (d, J=4.0 Hz, 1H), 1.24 (s, 9H).
(2-氯吡啶-4-基)(4-叔戊基苯基)甲醇(2-7): 白色固体, m.p. 103~104 ℃, 收率80%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.51 (s, 1H), 7.39 (dd, J=5.2, 1.4 Hz, 1H), 7.35~7.26 (m, 4H), 6.21 (d, J=4.0 Hz, 1H), 5.73 (d, J=4.0 Hz, 1H), 1.58 (q, J=7.4 Hz, 2H), 1.20 (s, 6H), 0.60 (t, J=7.4 Hz, 3H).
(2-氯吡啶-4-基)(4-氯苯基)甲醇(2-8): 白色固体, m.p. 130~131 ℃, 收率54%. 1H NMR (500 MHz, DMSO-d6) δ: 8.33 (d, J=5.1 Hz, 1H), 7.51 (s, 1H), 7.45~7.42 (m, 2H), 7.41~7.37 (m, 3H), 6.37 (d, J=4.1 Hz, 1H), 5.78 (d, J=4.1 Hz, 1H).
(2-氯吡啶-4-基)(4-氟苯基)甲醇(2-9): 白色固体, m.p. 103~104 ℃, 收率79%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.51 (s, 1H), 7.48~7.41 (m, 2H), 7.38 (d, J=5.1, 1.4 Hz, 1H), 7.20~7.11 (m, 2H), 6.32 (d, J=4.1 Hz, 1H), 5.78 (d, J=4.1 Hz, 1H).
(2-氯吡啶-4-基)(4-溴苯基)甲醇(2-10): 白色固体, m.p. 131~132 ℃, 收率86%. 1H NMR (500 MHz, DMSO-d6) δ: 8.33 (d, J=5.1 Hz, 1H), 7.56~7.49 (m, 3H), 7.40~7.35 (m, 3H), 6.38 (d, J=4.1 Hz, 1H), 5.77 (d, J=4.1 Hz, 1H).
(2-氯吡啶-4-基)(4-环己基苯基)甲醇(2-11): 白色固体, m.p. 134~135 ℃, 收率87%. 1H NMR (500 MHz, DMSO-d6) δ: 8.31 (d, J=5.1 Hz, 1H), 7.50 (s, 1H), 7.38 (d, J=5.2, 1.4 Hz, 1H), 7.32~7.15 (m, 4H), 6.19 (d, J=4.0 Hz, 1H), 5.71 (d, J=3.9 Hz, 1H), 2.49~2.40 (m, 1H), 1.81~1.71 (m, 4H), 1.71~1.64 (m, 1H), 1.42~1.27 (m, 4H), 1.26~1.14 (m, 1H).
[1'-联苯]-4-基(2-氯吡啶-4-基)甲醇(2-12): 淡黄色固体, m.p. 168~169 ℃, 收率83%. 1H NMR (500 MHz, DMSO-d6) δ: 8.34 (d, J=5.1 Hz, 1H), 7.66~7.60 (m, 4H), 7.56 (s, 1H), 7.52~7.47 (m, 2H), 7.47~7.41 (m, 3H), 7.38~7.31 (m, 1H), 6.33 (d, J=4.1 Hz, 1H), 5.82 (d, J=3.7 Hz, 1H).
(2-氯吡啶-4-基)(4-甲氧基苯基)甲醇(2-13): 白色固体, m.p. 108~109 ℃, 收率76%. 1H NMR (500 MHz, DMSO-d6) δ: 8.31 (d, J=5.1 Hz, 1H), 7.47 (s, 1H), 7.36 (dd, J=5.2, 1.4 Hz, 1H), 7.33~7.27 (m, 2H), 6.92~6.86 (m, 2H), 6.17 (d, J=4.1 Hz, 1H), 5.71 (d, J=3.2 Hz, 1H), 3.72 (s, 3H).
(2-氯吡啶-4-基)(3,4-二甲基苯基)甲醇(2-14): 白色固体, m.p. 146~147 ℃, 收率84%. 1H NMR (500 MHz, DMSO-d6) δ: 8.30 (d, J=5.1 Hz, 1H), 7.47 (s, 1H), 7.36 (dd, J=5.1, 1.4 Hz, 1H), 7.15 (s, 1H), 7.12~7.05 (m, 2H), 6.16 (d, J=4.1 Hz, 1H), 5.67 (d, J=4.0 Hz, 1H), 2.18 (s, 3H), 2.16 (s, 3H).
(2-氯吡啶-4-基)(2,4-二甲基苯基)甲醇(2-15): 白色固体, m.p. 123~124 ℃, 收率82%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.37 (s, 1H), 7.26 (d, J=5.2, 1.4 Hz, 1H), 7.16 (d, J=7.7 Hz, 1H), 7.02~6.95 (m, 2H), 6.12 (d, J=4.5 Hz, 1H), 5.85 (d, J=4.5 Hz, 1H), 2.24 (s, 3H), 2.23 (s, 3H).
(2-氯吡啶-4-基)(2,5-二甲基苯基)甲醇(2-16): 白色固体, m.p. 104~105 ℃, 收率88%. 1H NMR (500 MHz, DMSO-d6) δ: 8.32 (d, J=5.1 Hz, 1H), 7.38 (s, 1H), 7.26 (dd, J=5.2, 1.4 Hz, 1H), 7.12 (s, 1H), 7.07~7.02 (m, 1H), 7.02~6.97 (m, 1H), 6.15 (s, 1H), 5.85 (s, 1H), 2.24 (s, 3H), 2.21 (s, 3H).
(3-氯吡啶-4-基)(4-叔丁基苯基)甲醇(2-17): 白色固体, m.p. 177~178 ℃, 收率77%. 1H NMR (500 MHz, DMSO-d6) δ: 8.56 (d, J=5.0 Hz, 1H), 8.53 (s, 1H), 7.75 (d, J=5.0 Hz, 1H), 7.37~7.24 (m, 4H), 5.90 (s, 1H), 1.24 (s, 9H).
(6-氯吡啶-3-基)(4-叔丁基苯基)甲醇(2-18): 淡黄色固体, m.p. 66~68 ℃, 收率86%. 1H NMR (500 MHz, DMSO-d6) δ: 8.44 (d, J=2.4 Hz, 1H), 7.78 (dd, J=8.3, 2.5 Hz, 1H), 7.43 (d, J=8.3 Hz, 1H), 7.35~7.28 (m, 4H), 5.78 (s, 1H), 1.23 (s, 9H).
(2-氯-6-甲基吡啶-4-基)甲醇(2-19): 白色固体, m.p. 98~99 ℃, 收率84%. 1H NMR (500 MHz, DMSO-d6) δ: 7.37~7.29 (m, 4H), 7.29 (s, 1H), 7.26 (s, 1H), 6.13 (s, 1H), 5.66 (s, 1H), 2.40 (s, 3H), 1.24 (s, 9H).
(2-氯吡啶-3-基) (4-叔丁基苯基)甲醇(2-20): 白色固体, m.p. 137~138 ℃, 收率85%. 1H NMR (500 MHz, DMSO-d6) δ: 8.31 (dd, J=4.7, 2.0 Hz, 1H), 8.12 (dd, J=7.7, 2.0 Hz, 1H), 7.49 (dd, J=7.7, 4.7 Hz, 1H), 7.36~7.23 (m, 4H), 6.15 (d, J=4.2 Hz, 1H), 5.89 (d, J=3.9 Hz, 1H), 1.24 (s, 9H).
(4-叔丁基苯基)(3-氯苯基)甲醇(2-21): 浅棕色固体, m.p. 72~73 ℃, 收率67%. 1H NMR (500 MHz, DMSO-d6) δ: 7.43 (s, 1H), 7.34~7.31 (m, 4H), 7.30~7.27 (m, 2H), 7.27~7.23 (m, 1H), 5.96 (d, J=4.0 Hz, 1H), 5.68 (d, J=3.9 Hz, 1H), 1.24 (s, 9H).
合成方法参考已报道文献[
36], 中间体
3用50 mL乙腈溶解, 搅拌下加入25.68 mmol 1,2,4-三氮唑或咪唑和25.68 mmol碳酸钾, 回流反应2 h. 冷却至室温, 加入100 mL水, 乙酸乙酯萃取(60 mL×3), 合并有机相, 减压浓缩, 残余物用
V(石油醚)∶
V(乙酸乙酯)=1∶2柱层析纯化, 得到目标化合物
I.
2-氯-4-((对甲苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-01): 淡黄色油状液体, 收率64%. 1H NMR (500 MHz, CDCl3) δ: 8.35 (d, J=5.2 Hz, 1H), 8.03 (s, 1H), 7.96 (s, 1H), 7.24~7.08 (m, 4H), 6.97 (s, 1H), 6.88 (dd, J=5.2, 1.6 Hz, 1H), 6.63 (s, 1H), 2.36 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.82, 152.42, 150.89, 150.20, 143.72, 139.96, 132.43, 130.23, 128.84, 122.91, 121.11, 66.15, 21.26; HRMS (ESI) calcd for C15H14N4Cl [M+H]+ 285.0902, found 285.0896.
2-氯-4-((4-乙基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-02): 白色固体, m.p. 64~66 ℃, 收率44%. 1H NMR (500 MHz, CDCl3) δ: 8.37 (d, J=5.2 Hz, 1H), 8.05 (s, 1H), 7.97 (s, 1H), 7.26~7.23 (m, 2H), 7.16~7.11 (m, 2H), 6.99 (s, 1H), 6.90 (dd, J=5.3, 1.6 Hz, 1H), 6.64 (s, 1H), 2.67 (q, J=7.6 Hz, 2H), 1.24 (t, J=7.6 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.86, 152.47, 150.91, 150.25, 146.25, 143.79, 132.63, 129.10, 128.96, 122.96, 121.15, 66.27, 28.63, 15.38; HRMS (ESI) calcd for C16H16N4Cl [M+H]+ 299.1058, found 299.1051.
2-氯-4-((4-丙基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-03): 淡黄色油状液体, 收率49%. 1H NMR (500 MHz, CDCl3) δ: 8.34 (d, J=5.2 Hz, 1H), 8.03 (s, 1H), 7.96 (s, 1H), 7.24~7.10 (m, 4H), 6.98 (s, 1H), 6.89 (dd, J=5.2, 1.5 Hz, 1H), 6.64 (s, 1H), 2.59 (t, 2H), 1.69~1.57 (m, 2H), 0.92 (t, J=7.4 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.80, 152.39, 150.87, 150.19, 144.66, 143.73, 132.60, 129.60, 128.80, 122.91, 121.13, 66.18, 37.69, 24.35, 13.85; HRMS (ESI) calcd for C17H18N4Cl [M+H]+ 313.1215, found 313.1211.
2-氯-4-((4-异丙基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-04): 白色固体, m.p. 64~65 ℃, 收率47%. 1H NMR (500 MHz, CDCl3) δ: 8.36 (d, J=5.2 Hz, 1H), 8.05 (s, 1H), 7.96 (s, 1H), 7.30~7.25 (m, 2H), 7.17~7.11 (m, 2H), 6.99 (s, 1H), 6.90 (dd, J=5.3, 1.6 Hz, 1H), 6.64 (s, 1H), 2.97~2.88 (m, 1H), 1.25 (d, J=7.0 Hz, 6H); 13C NMR (126 MHz, CDCl3) δ: 152.88, 152.45, 150.91, 150.81, 150.24, 143.78, 132.71, 128.94, 127.68, 122.96, 121.16, 66.24, 33.95, 23.89; HRMS (ESI) calcd for C17H18N4Cl [M+H]+ 313.1215, found 313.1211.
2-氯-4-((4-戊基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-05): 淡黄色油状液体, 收率59%. 1H NMR (500 MHz, CDCl3) δ: 8.35 (d, J=5.2 Hz, 1H), 8.03 (s, 1H), 7.96 (s, 1H), 7.24~7.10 (m, 4H), 6.98 (s, 1H), 6.89 (dd, J=5.3, 1.6 Hz, 1H), 6.64 (s, 1H), 2.60 (t, J=8.8, 6.8 Hz, 2H), 1.63~1.55 (m, 2H), 1.35~1.24 (m, 4H), 0.87 (t, J=6.8 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.81, 152.41, 150.88, 150.20, 144.96, 143.74, 132.55, 129.56, 128.83, 122.93, 121.13, 66.20, 35.63, 31.51, 30.96, 22.54, 14.06; HRMS (ESI) calcd for C19H22N4Cl [M+H]+ 341.1528, found 341.1520.
2-氯-4-((4-叔丁基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-06): 白色固体, m.p. 72~73 ℃, 收率54%. 1H NMR (500 MHz, CDCl3) δ: 8.35 (d, J=5.2 Hz, 1H), 8.03 (s, 1H), 7.97 (s, 1H), 7.45~7.39 (m, 2H), 7.17~7.12 (m, 2H), 6.99 (s, 1H), 6.90 (dd, J=5.3, 1.7 Hz, 1H), 6.64 (s, 1H), 1.30 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 153.02, 152.78, 152.37, 150.85, 150.18, 143.75, 132.32, 128.61, 126.49, 122.92, 121.14, 66.09, 34.81, 31.24; HRMS (ESI) calcd for C18H20N4Cl [M+H]+ 327.1371, found 327.1367.
2-氯-4-((4-叔戊基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-07): 淡黄色油状液体, 收率52%. 1H NMR (500 MHz, CDCl3) δ: 8.30 (d, J=5.2 Hz, 1H), 7.98 (s, 1H), 7.89 (s, 1H), 7.34~7.26 (m, 2H), 7.11~7.05 (m, 2H), 6.94 (s, 1H), 6.84 (dd, J=5.4, 1.6 Hz, 1H), 6.59 (s, 1H), 1.57 (q, J=7.4 Hz, 2H), 1.21 (s, 6H), 0.60 (t, J=7.5 Hz, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.81, 152.40, 151.56, 150.85, 150.20, 143.76, 132.19, 128.54, 127.16, 122.94, 121.16, 66.13, 38.08, 36.79, 28.33, 9.18; HRMS (ESI) calcd for C19H22N4Cl [M+H]+ 341.1528, found 341.1523.
2-氯-4-((4-氯苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-08): 白色固体, m.p. 124~125 ℃, 收率53%. 1H NMR (500 MHz, CDCl3) δ: 8.39 (d, J=5.2 Hz, 1H), 8.07 (s, 1H), 8.06 (s, 1H), 7.44~7.38 (m, 2H), 7.21~7.15 (m, 2H), 7.01 (s, 1H), 6.92 (dd, J=5.2, 1.6 Hz, 1H), 6.65 (s, 1H); 13C NMR (126 MHz, CDCl3) δ: 153.04, 152.68, 150.47, 150.04, 143.74, 136.04, 134.12, 130.17, 129.85, 123.07, 121.18, 65.60; HRMS (ESI) calcd for C14H11N4Cl2 [M+H]+ 305.0355, found 305.0352.
2-氯-4-((4-氟苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-09): 淡黄色油状液体, 收率44%. 1H NMR (500 MHz, CDCl3) δ: 8.34 (d, J=5.2 Hz, 1H), 8.02 (d, J=4.4 Hz, 2H), 7.26~7.18 (m, 2H), 7.13~7.05 (m, 2H), 6.98 (s, 1H), 6.89 (dd, J=5.3, 1.6 Hz, 1H), 6.66 (s, 1H); 13C NMR (126 MHz, CDCl3) δ: 163.17 (d, J=250.7 Hz), 152.95, 152.48, 150.36, 150.31, 143.69, 131.48 (d, J=3.4 Hz), 130.75 (d, J=8.5 Hz), 122.91, 121.08, 116.59 (d, J=21.8 Hz), 65.42; HRMS (ESI) calcd for C14H11N4ClF [M+H]+289.0651, found 289.0647.
2-氯-4-((4-溴苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-10): 白色固体, m.p. 131~132 ℃, 收率46%. 1H NMR (500 MHz, CDCl3) δ: 8.38 (d, J=5.2 Hz, 1H), 8.04 (d, J=10.7 Hz, 2H), 7.59~7.53 (m, 2H), 7.14~7.08 (m, 2H), 7.03~6.99 (m, 1H), 6.91 (dd, J=5.2, 0.9 Hz, 1H), 6.62 (s, 1H); 13C NMR (126 MHz, CDCl3) δ: 153.11, 152.66, 150.46, 149.96, 143.74, 134.66, 132.79, 130.40, 124.18, 123.07, 121.19, 65.62; HRMS (ESI) calcd for C14H11N4ClBr [M+H]+ 348.9850, found 348.9848.
2-氯-4-((4-环己基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-11): 白色固体, m.p. 59~60 ℃, 收率50%. 1H NMR (500 MHz, CDCl3) δ: 8.40 (d, J=5.2 Hz, 1H), 8.08 (s, 1H), 8.00 (s, 1H), 7.31~7.26 (m, 2H), 7.19~7.13 (m, 2H), 7.02 (s, 1H), 6.93 (dd, J=5.3, 1.6 Hz, 1H), 6.66 (s, 1H), 2.59~2.50 (m, 1H), 1.92~1.85 (m, 4H), 1.81~1.73 (m, 1H), 1.48~1.36 (m, 4H), 1.34~1.22 (m, 1H); 13C NMR (126 MHz, CDCl3) δ: 152.80, 152.49, 150.90, 150.27, 150.07, 143.79, 132.65, 128.93, 128.08, 122.98, 121.18, 66.34, 44.37, 34.38, 26.86, 26.14; HRMS (ESI) calcd for C20H22N4Cl [M+H]+ 353.1528, found 353.1522.
4-([1'-联苯]-4-基(1H-1,2,4-三唑-1-基)甲基)-2-氯吡啶(I-12): 淡黄色油状液体, 收率59%. 1H NMR (500 MHz, CDCl3) δ: 8.38 (d, J=5.1 Hz, 1H), 8.07 (d, J=6.2 Hz, 2H), 7.67~7.61 (m, 2H), 7.60~7.54 (m, 2H), 7.48~7.41 (m, 2H), 7.41~7.34 (m, 1H), 7.33~7.27 (m, 2H), 7.05 (s, 1H), 6.96 (dd, J=5.3, 1.6 Hz, 1H), 6.72 (s, 1H); 13C NMR (126 MHz, CDCl3) δ: 152.91, 152.48, 150.54, 150.30, 143.78, 142.69, 139.76, 134.30, 129.27, 129.02, 128.16, 128.06, 127.19, 123.02, 121.21, 66.02; HRMS (ESI) calcd for C20H16N4Cl [M+H]+ 347.1058, found 347.1054.
2-氯-4-((4-甲氧基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-13): 白色固体, m.p. 65~66 ℃, 收率61%. 1H NMR (500 MHz, CDCl3) δ: 8.32 (d, J=5.2 Hz, 1H), 8.01 (s, 1H), 7.94 (s, 1H), 7.17~7.10 (m, 2H), 6.94 (s, 1H), 6.93~6.88 (m, 2H), 6.85 (dd, J=5.2, 1.6 Hz, 1H), 6.61 (s, 1H), 3.78 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 160.47, 152.79, 152.32, 151.07, 150.12, 143.65, 130.29, 127.26, 122.74, 120.97, 114.82, 65.77, 55.42; HRMS (ESI) calcd for C15H14N4ClO [M+H]+ 301.0851, found 301.0844.
2-氯-4-((3,4-二甲基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-14): 白色固体, m.p. 86~87 ℃, 收率49%. 1H NMR (500 MHz, CDCl3) δ: 8.36 (d, J=5.2 Hz, 1H), 8.04 (s, 1H), 7.95 (s, 1H), 7.17 (d, J=7.8 Hz, 1H), 7.00~6.96 (m, 2H), 6.93 (dd, J=7.8, 2.1 Hz, 1H), 6.88 (dd, J=5.3, 1.6 Hz, 1H), 6.60 (s, 1H), 2.27 (s, 3H), 2.24 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.85, 152.42, 151.03, 150.19, 143.77, 138.67, 138.14, 132.76, 130.73, 130.08, 126.36, 122.92, 121.12, 66.29, 19.95, 19.64; HRMS (ESI) calcd for C16H16N4Cl [M+H]+ 299.1058, found 299.1053.
2-氯-4-((2,4-二甲基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-15): 白色固体, m.p. 106~107 ℃, 收率45%. 1H NMR (500 MHz, CDCl3) δ: 8.37 (d, J=5.2 Hz, 1H), 8.06 (s, 1H), 7.84 (s, 1H), 7.11 (s, 1H), 7.02 (d, J=7.9 Hz, 1H), 6.90 (s, 1H), 6.87 (s, 1H), 6.81 (dd, J=5.4, 1.6 Hz, 1H), 6.72 (d, J=7.9 Hz, 1H), 2.34 (s, 3H), 2.23 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.82, 152.56, 151.07, 150.27, 143.73, 140.02, 136.94, 132.41, 130.83, 128.85, 127.75, 122.94, 121.16, 63.16, 21.18, 19.16; HRMS (ESI) calcd for C16H16N4Cl [M+H]+ 299.1058, found 299.1053.
2-氯-4-((2,5-二甲基苯基)(1H-1,2,4-三唑-1-基)甲基)吡啶(I-16): 白色固体, m.p. 106~107 ℃, 收率31%. 1H NMR (500 MHz, CDCl3) δ: 8.38 (d, J=5.2 Hz, 1H), 8.06 (s, 1H), 7.84 (s, 1H), 7.19~7.11 (m, 2H), 6.91 (s, 1H), 6.87 (s, 1H), 6.83 (dd, J=5.2, 1.6 Hz, 1H), 6.61 (s, 1H), 2.25 (s, 3H), 2.20 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.82, 152.56, 150.82, 150.28, 143.79, 136.83, 133.86, 133.54, 131.51, 130.62, 129.23, 123.04, 121.28, 63.33, 21.17, 18.78; HRMS (ESI) calcd for C16H16N4Cl [M+H]+299.1058, found 299.1052.
4-((4-叔丁基苯基)(1H-1,2,4-三唑-1-基)甲基)-3-氯吡啶(I-17): 白色固体, m.p. 94~95 ℃, 收率55%. 1H NMR (500 MHz, CDCl3) δ: 8.61 (s, 1H), 8.49 (d, J=5.1 Hz, 1H), 8.04 (s, 1H), 7.93 (s, 1H), 7.45~7.39 (m, 2H), 7.14~7.08 (m, 2H), 6.98 (s, 1H), 6.81 (d, J=5.1 Hz, 1H), 1.31 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 152.92, 152.88, 150.11, 148.43, 144.96, 144.25, 131.19, 130.98, 128.53, 126.45, 123.02, 63.85, 34.86, 31.32; HRMS (ESI) calcd for C18H20N4Cl [M+H]+ 327.1371, found 327.1364.
5-((4-叔丁基苯基)(1H-1,2,4-三唑-1-基)甲基)-2-氯吡啶(I-18): 淡黄色油状液体, 收率38%. 1H NMR (500 MHz, CDCl3) δ: 8.18 (d, J=2.6 Hz, 1H), 8.01 (d, J=6.7 Hz, 2H), 7.43 (dd, J=8.4, 2.6 Hz, 1H), 7.41~7.36 (m, 2H), 7.31 (d, J=8.3 Hz, 1H), 7.11~7.04 (m, 2H), 6.70 (s, 1H), 1.28 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 152.67, 152.54, 151.59, 149.14, 143.62, 138.35, 133.44, 133.39, 127.91, 126.32, 124.43, 64.61, 34.73, 31.24; HRMS (ESI) calcd for C18H20N4Cl [M+H]+ 327.1371, found 327.1368.
4-((4-叔丁基苯基)(1H-1,2,4-三唑-1-基)甲基)-2-氯-6-甲基吡啶(I-19): 白色固体, m.p. 96~98 ℃, 收率46%. 1H NMR (500 MHz, CDCl3) δ: 8.02 (s, 1H), 7.94 (s, 1H), 7.44~7.38 (m, 2H), 7.15~7.10 (m, 2H), 6.80 (s, 1H), 6.72 (s, 1H), 6.59 (s, 1H), 2.46 (s, 3H), 1.29 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 160.28, 153.04, 152.78, 151.47, 150.91, 143.75, 132.45, 128.66, 126.53, 120.55, 119.90, 66.28, 34.88, 31.31, 24.40; HRMS (ESI) calcd for C18H20N4Cl [M+H]+ 341.1528, found 341.1518.
3-((4-叔丁基苯基)(1H-1,2,4-三唑-1-基)甲基)-2-氯吡啶(I-20): 淡黄色油状液体, 收率46%. 1H NMR (500 MHz, CDCl3) δ: 8.37 (dd, J=4.7, 2.0 Hz, 1H), 8.00 (d, J=12.7 Hz, 2H), 7.42~7.36 (m, 2H), 7.34~7.29 (m, 1H), 7.25~7.21 (m, 1H), 7.09~7.03 (m, 2H), 7.02 (s, 1H), 1.29 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 152.70, 152.46, 150.46, 149.52, 144.16, 138.26, 133.22, 132.32, 128.07, 126.26, 122.86, 63.95, 34.75, 31.28; HRMS (ESI) calcd for C18H20N4Cl [M+H]+ 327.1371, found 327.1366.
1-((4-叔丁基苯基)(3-氯苯基)甲基)-1H-1,2,4-三唑(I-21): 白色固体, m.p. 76~77 ℃, 收率52%. 1H NMR (500 MHz, CDCl3) δ: 8.03 (s, 1H), 7.95 (s, 1H), 7.43~7.38 (m, 2H), 7.35~7.28 (m, 2H), 7.13~7.06 (m, 3H), 7.02~6.96 (m, 1H), 6.69 (s, 1H), 1.31 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 152.46, 152.25, 143.65, 140.45, 135.02, 134.07, 130.24, 128.78, 128.20, 128.12, 126.21, 126.10, 67.17, 34.78, 31.34; HRMS (ESI) calcd for C19H21N3Cl [M+H]+ 326.1419, found 326.1415.
4-((1H-咪唑-1-基)(对甲苯基)甲基)-2-氯吡啶(I-22): 白色固体, m.p. 67~68 ℃, 收率53%. 1H NMR (500 MHz, CDCl3) δ: 8.37 (d, J=5.2 Hz, 1H), 7.46 (s, 1H), 7.21 (d, J=7.9 Hz, 2H), 7.14 (s, 1H), 7.05~7.00 (m, 2H), 6.97 (s, 1H), 6.88 (dd, J=5.1, 1.6 Hz, 1H), 6.83 (s, 1H), 6.44 (s, 1H), 2.37 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.65, 152.02, 150.37, 139.67, 137.22, 133.53, 130.19, 130.04, 128.64, 122.93, 121.13, 119.07, 63.59, 21.28; HRMS (ESI) calcd for C16H15N3Cl [M+H]+ 284.0949, found 284.0943.
4-((4-叔丁基苯基)(1H-咪唑-1-基)甲基)-2-氯吡啶(I-23): 白色固体, m.p. 84~85 ℃, 收率67%. 1H NMR (500 MHz, CDCl3) δ: 8.37 (d, J=5.1 Hz, 1H), 7.46 (s, 1H), 7.45~7.38 (m, 2H), 7.14 (s, 1H), 7.10~7.03 (m, 2H), 7.02~6.97 (m, 1H), 6.92~6.87 (m, 1H), 6.84 (t, J=1.4 Hz, 1H), 6.45 (s, 1H), 1.32 (s, 9H); 13C NMR (126 MHz, CDCl3) δ: 152.81, 152.63, 152.00, 150.37, 137.25, 133.40, 129.99, 128.44, 126.45, 122.95, 121.16, 119.12, 63.55, 34.86, 31.33; HRMS (ESI) calcd for C19H21N3Cl [M+H]+ 326.1419, found 326.1418.
2-氯-4-((2,5-二甲基苯基)(1H-咪唑-1-基)甲基)吡啶(I-24): 白色固体, m.p. 149~150 ℃, 收率36%. 1H NMR (500 MHz, CDCl3) δ: 8.38 (d, J=5.2 Hz, 1H), 7.37 (s, 1H), 7.16~7.09 (m, 3H), 6.93 (s, 1H), 6.84 (dd, J=5.2, 1.6 Hz, 1H), 6.78 (s, 1H), 6.61 (s, 1H), 6.50 (s, 1H), 2.23 (s, 3H), 2.18 (s, 3H); 13C NMR (126 MHz, CDCl3) δ: 152.67, 152.06, 150.36, 137.35, 136.63, 134.69, 133.82, 131.38, 130.22, 130.04, 123.01, 121.29, 119.28, 60.61, 21.18, 18.71; HRMS (ESI) calcd for C17H17N3Cl [M+H]+298.1106, found 298.1103.