化学学报 ›› 2021, Vol. 79 ›› Issue (11): 1331-1344.DOI: 10.6023/A21070338 上一篇 下一篇
综述
吴良a, 魏瀚林a, 申杰峰a,*( ), 陈建中b,*(
), 陈建中b,*( ), 张万斌a,b
), 张万斌a,b
投稿日期:2021-07-20
发布日期:2021-09-17
通讯作者:
申杰峰, 陈建中
作者简介: |
吴良, 2020年在上海交通大学获得博士学位(导师: 张万斌教授). 目前在上海交通大学张万斌教授课题组从事博士后研究工作, 主要研究方向为丰产金属催化的不对称反应. |
 |
申杰峰, 博士, 上海交通大学药学院副研究员, 硕士研究生导师. 2012博士毕业于上海交通大学化学化工学院(导师: 张万斌教授); 同年进入上海交通大学药学院从事博士后研究工作(导师: 刘燕刚教授). 2014年至今就职于上海交通大学药学院, 主要研究方向为不对称催化、化学工艺及新药开发研究. |
 |
陈建中, 上海交通大学化学化工学院副研究员. 本科毕业于浙江工业大学; 硕士毕业于华东理工大学(导师: 赵敏副教授); 博士毕业于上海交通大学(导师: 张万斌教授), 同年本校从事博士后研究(导师: 颜德岳院士). 自2015年起在上海交通大学主要从事不对称反应研究, 新型配体的设计、合成及应用, 药物和香料等合成工艺研究等. |
基金资助:
Liang Wua, Hanlin Weia, Jiefeng Shena( ), Jianzhong Chenb(
), Jianzhong Chenb( ), Wanbin Zhanga,b
), Wanbin Zhanga,b
Received:2021-07-20
Published:2021-09-17
Contact:
Jiefeng Shen, Jianzhong Chen
Supported by:文章分享
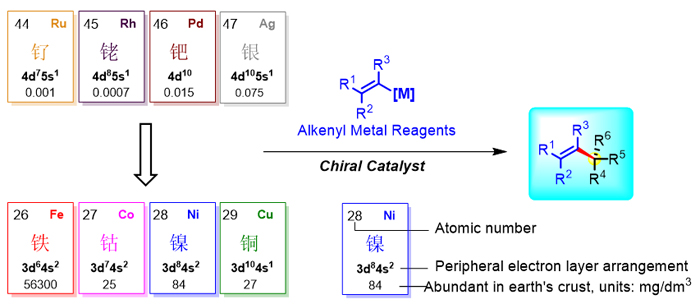
烯丙位手性中心不仅广泛存在于天然产物和药物活性分子中, 也是有机合成中的重要合成砌块. 过渡金属催化烯基金属试剂作为亲核试剂的不对称加成或偶联反应是构建这一结构非常有吸引力的策略之一. 在众多金属催化剂中, 铁钴镍铜等丰产金属由于其独特的催化活性以及低毒性、环境友好等优点而被用来代替铑钯等稀有金属应用于此类不对称烯基化反应中, 并取得了显著的成果. 基于此, 本文将综述丰产金属催化的烯基金属试剂参与的不对称烯基化反应研究进展. 主要包括: (1)钴催化的不对称烯基化反应, (2)镍催化的不对称烯基化反应, (3)铜催化的不对称烯基化反应以及(4)其他丰产金属催化的不对称烯基化反应等四部分.
吴良, 魏瀚林, 申杰峰, 陈建中, 张万斌. 丰产金属催化烯基金属试剂的不对称烯基化反应研究进展[J]. 化学学报, 2021, 79(11): 1331-1344.
Liang Wu, Hanlin Wei, Jiefeng Shen, Jianzhong Chen, Wanbin Zhang. Development of Earth-Abundant Metals-Catalyzed Enantioselective Alkenylations Using Alkenyl Metal Reagents[J]. Acta Chimica Sinica, 2021, 79(11): 1331-1344.
| [1] |
(a)Catalysis without Precious Metals, Ed.: Bullock, R. M., Wiley-VCH, Weinheim, 2010.
pmid: 29589919 |
|
(b) Morris R. H. Chem. Soc. Rev. 2009, 38, 2282.
doi: 10.1039/b806837m pmid: 29589919 |
|
|
(c) Zhang L.; Huang Z. Synlett 2013, 1745.
pmid: 29589919 |
|
|
(d) Zhu S.-F.; Zhou Q.-L. Natl. Sci. Rev. 2014, 1, 580.
doi: 10.1093/nsr/nwu019 pmid: 29589919 |
|
|
(e) Guo N.; Zhu S. Chin. J. Org. Chem. 2015, 35, 1383. (in Chinese)
pmid: 29589919 |
|
|
( 郭娜, 朱守非, 有机化学, 2015, 35, 1383.)
doi: 10.6023/cjoc201502032 pmid: 29589919 |
|
|
(f) Li J.; Liu K.; Duan X.; Liu J. Chin. J. Org. Chem. 2017, 37, 314. (in Chinese)
doi: 10.6023/cjoc201608009 pmid: 29589919 |
|
|
( 李娟华, 刘昆明, 段新方, 刘晋彪, 有机化学, 2017, 37, 314.)
doi: 10.6023/cjoc201608009 pmid: 29589919 |
|
|
(g) Wu Z.; Zhang W. Chin. J. Org. Chem. 2017, 37, 2250. (in Chinese)
doi: 10.6023/cjoc201704031 pmid: 29589919 |
|
|
( 吴正兴, 张万斌, 有机化学, 2017, 37, 2250.)
doi: 10.6023/cjoc201704031 pmid: 29589919 |
|
|
(h) Obligacion J. V.; Chirik P. J. Nat. Rev. Chem. 2018, 2, 15.
doi: 10.1038/s41570-018-0001-2 pmid: 29589919 |
|
|
(i) Chen J.; Lu Z. Org. Chem. Front. 2018, 5, 260.
doi: 10.1039/C7QO00613F pmid: 29589919 |
|
|
(j) Chen J.; Guo J.; Lu Z. Chin. J. Chem. 2018, 36, 1075.
doi: 10.1002/cjoc.201800314 pmid: 29589919 |
|
|
(k) Fu X.; Zhao W. Chin. J. Org. Chem. 2019, 39, 625. (in Chinese)
doi: 10.6023/cjoc201808031 pmid: 29589919 |
|
|
( 付晓飞, 赵文献, 有机化学, 2019, 39, 625.)
doi: 10.6023/cjoc201808031 pmid: 29589919 |
|
|
(l) Sun Y.; Guan R.; Liu Z.; Wang Y. Chin. J. Org. Chem. 2020, 40, 899. (in Chinese)
doi: 10.6023/cjoc201909035 pmid: 29589919 |
|
|
( 孙越, 关瑞, 刘兆洪, 王也铭, 有机化学, 2020, 40, 899.)
doi: 10.6023/cjoc201909035 pmid: 29589919 |
|
|
(m) Guo J.; Cheng Z.; Chen J.; Chen X.; Lu Z. Acc. Chem. Res. 2021, 54, 2701.
doi: 10.1021/acs.accounts.1c00212 pmid: 29589919 |
|
|
(n) Chen J.; Xi T.; Lu Z. Org. Lett. 2014, 16, 6452.
doi: 10.1021/ol503282r pmid: 29589919 |
|
|
(o) An L.; Tong F.; Zhang X. Acta Chim. Sinica 2018, 76, 977. (in Chinese)
doi: 10.6023/A18080314 pmid: 29589919 |
|
|
( 安伦, 童非非, 张新刚, 有机化学, 2018, 76, 977.)
pmid: 29589919 |
|
|
(p) Cheng B.; Liu W.; Lu Z. J. Am. Chem. Soc. 2018, 140, 5014.
doi: 10.1021/jacs.8b01638 pmid: 29589919 |
|
|
(q) Wang S.; Sun M.; Zhang H.; Zhang J.; He Y.; Feng Z. CCS Chem. 2020, 2, 2164.
pmid: 29589919 |
|
|
(r) Zou S.; Zhang T.; Wang S.; Huang H. Chin. J. Chem. 2020, 38, 389.
doi: 10.1002/cjoc.v38.4 pmid: 29589919 |
|
|
(s) Wang G.-X.; Yin J.; Li J.; Yin Z.-B.; Wu B.; Wei J.; Zhang W.-X.; Xi Z. CCS Chem. 2021, 3, 308.
pmid: 29589919 |
|
|
(t) Xu S.; Liu G.; Huang Z. Chin. J. Chem. 2021, 39, 585.
doi: 10.1002/cjoc.v39.3 pmid: 29589919 |
|
|
(u) Li W.-D.; Chen J.; Zhu D.-Y.; Xia J.-B. Chin. J. Chem. 2021, 39, 614.
doi: 10.1002/cjoc.v39.3 pmid: 29589919 |
|
| [2] |
(a) Pellissier H.; Clavier H. Chem. Rev. 2014, 114, 2775.
doi: 10.1021/cr4004055 pmid: 26854359 |
|
(b) Gandeepan P.; Cheng C.-H. Acc. Chem. Res. 2015, 48, 1194.
doi: 10.1021/ar500463r pmid: 26854359 |
|
|
(c) Gu Z.; Ji S. Acta Chim. Sinica 2018, 76, 347. (in Chinese)
doi: 10.6023/A18010023 pmid: 26854359 |
|
|
( 顾正洋, 纪顺俊, 化学学报, 2018, 76, 347.)
doi: 10.6023/A18010023 pmid: 26854359 |
|
|
(d) Sun Y.; Ding Q.; Yu Y.; He Y.; Huang F. Chin. J. Org. Chem. 2019, 39, 3363. (in Chinese)
doi: 10.6023/cjoc201906026 pmid: 26854359 |
|
|
( 孙义明, 丁奇峰, 于杨, 何益得, 黄菲, 有机化学, 2019, 39, 3363.)
doi: 10.6023/cjoc201906026 pmid: 26854359 |
|
|
(e) Dai Z.; Yu Z.; Bai Y.; Li J.; Peng J. Chin. J. Org. Chem. 2020, 40, 1177. (in Chinese)
doi: 10.6023/cjoc201910012 pmid: 26854359 |
|
|
( 代自男, 余泽浩, 白赢, 厉嘉云, 彭家建, 有机化学, 2020, 40, 1177.)
doi: 10.6023/cjoc201910012 pmid: 26854359 |
|
|
(f) Wen J.; Wang F.; Zhang X. Chem. Soc. Rev. 2021, 50, 3211.
doi: 10.1039/D0CS00082E pmid: 26854359 |
|
|
(g) Gao K.; Lee P.-S.; Fujita T.; Yoshikai N. J. Am. Chem. Soc. 2010, 132, 12249.
doi: 10.1021/ja106814p pmid: 26854359 |
|
|
(h) Friedfeld M. R.; Shevlin M.; Hoyt J. M.; Krska S. W.; Tudge M. T.; Chirik P. J. Science 2013, 342, 1076.
doi: 10.1126/science.1243550 pmid: 26854359 |
|
|
(i) Chen Q. A.; Kim D. K.; Dong V. M. J. Am. Chem. Soc. 2014, 136, 3772.
doi: 10.1021/ja500268w pmid: 26854359 |
|
|
(j) Yang J.; Yoshikai N. J. Am. Chem. Soc. 2014, 136, 16748.
doi: 10.1021/ja509919x pmid: 26854359 |
|
|
(k) Zhang L.; Zuo Z.; Wan X.; Huang Z. J. Am. Chem. Soc. 2014, 136, 15501.
doi: 10.1021/ja5093908 pmid: 26854359 |
|
|
(l) Santhoshkumar R.; Mannathan S.; Cheng C.-H. J. Am. Chem. Soc. 2015, 137, 16116.
doi: 10.1021/jacs.5b10447 pmid: 26854359 |
|
|
(m) Friedfeld M. R.; Shevlin M.; Marguloeux G. W.; Campeau L.; Chirik P. J. J. Am. Chem. Soc. 2016, 138, 3314.
doi: 10.1021/jacs.5b10148 pmid: 26854359 |
|
|
(n) Wu L.; Shao Q.; Yang G.; Zhang W. Chem. Eur. J. 2018, 24, 1241.
doi: 10.1002/chem.v24.6 pmid: 26854359 |
|
|
(o) Hu Y.; Zhang Z.; Zhang J.; Liu Y.; Gridnev I. D.; Zhang W. Angew. Chem. Int. Ed. 2019, 58, 15767.
doi: 10.1002/anie.v58.44 pmid: 26854359 |
|
|
(p) Li X.; Wu X.; Tang L.; Xie F.; Zhang W. Chem. Asian J. 2019, 14, 3835.
doi: 10.1002/asia.v14.21 pmid: 26854359 |
|
|
(q) Cheng Z.; Xing S.; Guo J.; Cheng B.; Hu L.-F.; Zhang X.-H.; Lu Z. Chin. J. Chem. 2019, 37, 457.
doi: 10.1002/cjoc.v37.5 pmid: 26854359 |
|
|
(r) Wu L.; Shao Q.; Kong L.; Chen J.; Wei Q.; Zhang W. Org. Chem. Front. 2020, 7, 862.
doi: 10.1039/D0QO00072H pmid: 26854359 |
|
|
(s) Lin X.; Tan Z.; Yang W.; Yang W.; Liu X.; Feng X. CCS Chem. 2020, 2, 1423.
pmid: 26854359 |
|
|
(t) Hu Y.; Zhang Z.; Liu Y.; Zhang W. Angew. Chem. Int. Ed. 2021, 60, 16989.
doi: 10.1002/anie.v60.31 pmid: 26854359 |
|
| [3] |
(a) Tasker S. Z.; Standley E. A.; Jamison T. F. Nature 2014, 509, 299.
doi: 10.1038/nature13274 |
|
(b) Butt N. A.; Zhang W. Chem. Soc. Rev. 2015, 44, 7929.
doi: 10.1039/C5CS00144G |
|
|
(c) Choi J.; Fu G. C. Science 2017, 356, eaaf7230.
doi: 10.1126/science.aaf7230 |
|
|
(d) Fu G. C. ACS Cent. Sci. 2017, 3, 692.
doi: 10.1021/acscentsci.7b00212 |
|
|
(e) Zhang Z.; Butt N. A.; Zhou M.; Liu D.; Zhang W. Chin. J. Chem. 2018, 36, 443.
doi: 10.1002/cjoc.v36.5 |
|
|
(f) Quan M.; Wu L.; Yang G.; Zhang W. Chem. Commun. 2018, 54, 10394.
doi: 10.1039/C8CC04932G |
|
|
(g) Chen J.; Butt N. A.; Zhang W. Res. Chem. Intermed. 2019, 45, 5959.
doi: 10.1007/s11164-019-04013-w |
|
|
(h) Liu Y.; Dong X.-Q.; Zhang X. Chin. J. Org. Chem. 2020, 40, 1096. (in Chinese)
doi: 10.6023/cjoc201912025 |
|
|
( 刘元华, 董秀琴, 张绪穆, 有机化学, 2020, 40, 1096.)
doi: 10.6023/cjoc201912025 |
|
|
(i) Cheng L.; Zhou Q. Acta Chim. Sinica 2020, 78, 1017. (in Chinese)
doi: 10.6023/A20070335 |
|
|
( 程磊, 周其林, 化学学报, 2020, 78, 1017.)
doi: 10.6023/A20070335 |
|
|
(j) Dai H.; Wu F.; Bai D. Chin. J. Org. Chem. 2020, 40, 1423. (in Chinese)
doi: 10.6023/cjoc202002035 |
|
|
( 代洪雪, 吴芬, 白大昌, 有机化学, 2020, 40, 1423.)
doi: 10.6023/cjoc202002035 |
|
|
(k) Chen S.; Zhao Y. Chin. J. Org. Chem. 2020, 40, 3078. (in Chinese)
doi: 10.6023/cjoc202005072 |
|
|
( 陈思, 赵延川, 有机化学, 2020, 40, 3078.)
doi: 10.6023/cjoc202005072 |
|
|
(l) Quan M.; Tang L.; Shen J.; Yang G.; Zhang W. Chem. Commun. 2017, 53, 609.
doi: 10.1039/C6CC08759K |
|
|
(m) Zhang Y.; He J.; Song P.; Wang Y.; Zhu S. CCS Chem. 2020, 2, 2259.
|
|
|
(n) Li Z.; Wu D.; Ding C.; Yin G. CCS Chem. 2020, 2, 576.
|
|
|
(o) Li B.; Chen J.; Zhang Z.; Gridnev I. D.; Zhang W. Angew. Chem. Int. Ed. 2019, 58, 7329.
doi: 10.1002/anie.v58.22 |
|
|
(p) Liu D.; Li B.; Chen J.; Gridnev I. D.; Yan. D.; Zhang, W. Nat. Commun. 2020, 11, 5935.
doi: 10.1038/s41467-020-19807-5 |
|
|
(q) Hu Y.; Chen J.; Li B.; Zhang Z.; Gridnev I. D.; Zhang W. Angew. Chem. Int. Ed. 2020, 59, 5371.
doi: 10.1002/anie.v59.13 |
|
|
(r) Chen J.; Zhang W. Chin. J. Org. Chem. 2020, 40, 4372. (in Chinese)
doi: 10.6023/cjoc202000086 |
|
|
( 陈建中, 张万斌, 有机化学, 2020, 40, 4372. )
doi: 10.6023/cjoc202000086 |
|
|
(s) Li B.; Liu D.; Hu Y.; Chen J.; Zhang Z.; Zhang W. Eur. J. Org. Chem. 2021, 3421.
|
|
|
(t) Zhu C.; Yue H.; Nikolaienko P.; Rueping M. CCS Chem. 2020, 2, 179.
doi: 10.31635/ccschem.020.201900112 |
|
|
(u) Han X.-W.; Zhang T.; Yao W.-W.; Chen H.; Ye M. CCS Chem. 2020, 2, 955.
|
|
|
(v) Xiao C.; Xiao W. Chin. J. Org. Chem. 2020, 40, 3004. (in Chinese)
doi: 10.6023/cjoc202000059 |
|
|
( 肖聪, 肖文精, 有机化学, 2020, 40, 3004.)
doi: 10.6023/cjoc202000059 |
|
| [4] |
(a) Alexakis A.; Bäckvall J. E.; Krause N.; Pámies O.; Diéguez M. Chem. Rev. 2008, 108, 2796.
doi: 10.1021/cr0683515 pmid: 18671436 |
|
(b) Shi Z.; Zhang C.; Tang C.; Jiao N. Chem. Soc. Rev. 2012, 41, 3381.
doi: 10.1039/c2cs15224j pmid: 18671436 |
|
|
(c) Guo X.-X.; Gu D.-W.; Wu Z.; Zhang W. Chem. Rev. 2015, 115, 1622
doi: 10.1021/cr500410y pmid: 18671436 |
|
|
(d) Liu Y.; Zhang W. Chin. J. Org. Chem. 2016, 36, 2249. (in Chinese)
doi: 10.6023/cjoc201609005 pmid: 18671436 |
|
|
( 刘媛媛, 张万斌, 有机化学, 2016, 36, 2249.)
doi: 10.6023/cjoc201609005 pmid: 18671436 |
|
|
(e) Duan X.; Liu N.; Wang J.; Ma J. Chin. J. Org. Chem. 2019, 39, 661. (in Chinese)
pmid: 18671436 |
|
|
( 段希焱, 刘宁, 王佳, 马军营, 有机化学, 2019, 39, 661.)
doi: 10.6023/cjoc201808015 pmid: 18671436 |
|
|
(f) Xie J.; Wang X.; Wu F.; Zhang J. Chin. J. Org. Chem. 2019, 39, 3026. (in Chinese)
doi: 10.6023/cjoc201907051 pmid: 18671436 |
|
|
( 谢建伟, 汪小创, 吴丰田, 张洁, 有机化学, 2019, 39, 3026.)
doi: 10.6023/cjoc201907051 pmid: 18671436 |
|
|
(g) Lei L.; Li C.; Mo D. Chin. J. Org. Chem. 2019, 39, 2989. (in Chinese)
doi: 10.6023/cjoc201904037 pmid: 18671436 |
|
|
( 雷禄, 李承璟, 莫冬亮, 有机化学, 2019, 39, 2989.)
doi: 10.6023/cjoc201904037 pmid: 18671436 |
|
|
(h) Wang C.; Zhou F.; Zhou J. Chin. J. Org. Chem. 2020, 40, 3065. (in Chinese)
doi: 10.6023/cjoc202005020 pmid: 18671436 |
|
|
( 王才, 周锋, 周剑, 有机化学, 2020, 40, 3065.)
doi: 10.6023/cjoc202005020 pmid: 18671436 |
|
|
(i) Wang M.; Zhang Z.; Xie F.; Zhang W. Chem. Commun. 2014, 50, 3163.
doi: 10.1039/c3cc49107b pmid: 18671436 |
|
|
(j) Wu X.; Xie F.; Ling Z.; Tang L.; Zhang W. Adv. Synth. Catal. 2016, 358, 2510.
doi: 10.1002/adsc.201600375 pmid: 18671436 |
|
|
(k) Zhang D.-D.; Liu Y.-L.; Wang Y.; Wei H.; Shi M.; Wang F.-J. Chin. Chem. Lett. 2016, 27, 563.
doi: 10.1016/j.cclet.2016.01.047 pmid: 18671436 |
|
|
(l) Ling Z.; Singh S.; Xie F.; Wu L.; Zhang W. Chem. Commun. 2017, 53, 5364.
doi: 10.1039/C7CC02159C pmid: 18671436 |
|
|
(m) Shao Q.; Wu L.; Chen J.; Gridnev I. D.; Yang G.; Xie F.; Zhang W. Adv. Synth. Catal. 2018, 360, 4625.
doi: 10.1002/adsc.201800850 pmid: 18671436 |
|
|
(n) Ling Z.; Xie F.; Gridnev I. D., Terada M.; Zhang W. Chem. Commun. 2018, 54, 9446.
doi: 10.1039/C8CC05307C pmid: 18671436 |
|
|
(o) Wu X.; Xie F.; Gridnev I. D.; Zhang W. Org. Lett. 2018, 20, 1638.
doi: 10.1021/acs.orglett.8b00379 pmid: 18671436 |
|
|
(p) Cheng Z.; Chen P.; Liu G. Acta Chim. Sinica 2019, 77, 856. (in Chinese)
doi: 10.6023/A19070252 pmid: 18671436 |
|
|
( 成忠明, 陈品红, 刘国生, 化学学报, 2019, 77, 856.)
doi: 10.6023/A19070252 pmid: 18671436 |
|
|
(q) You Y.; Van Pham Q.; Ge S. CCS Chem. 2019, 1, 455.
doi: 10.31635/ccschem.019.20190053 pmid: 18671436 |
|
|
(r) Lin F.; Liang Y.; Li X.; Song S.; Jiao N. Acta Chim. Sinica 2019, 77, 906. (in Chinese)
doi: 10.6023/A19020070 pmid: 18671436 |
|
|
( 林凤闺蓉, 梁宇杰, 郦鑫耀, 宋颂, 焦宁, 化学学报, 2019, 77, 906.)
doi: 10.6023/A19020070 pmid: 18671436 |
|
|
(s) Gan X.-C.; Yin L. CCS Chem. 2020, 2, 203.
doi: 10.31635/ccschem.020.201900102 pmid: 18671436 |
|
|
(t) Gao X.; Xiao Y.-L.; Zhang S.; Wu J.; Zhang X. CCS Chem. 2020, 2, 1463.
pmid: 18671436 |
|
|
(u) Zhao Q.; Isenegger P. G.; Wilson T. C.; Sap J. B. I.; Guibbal F.; Lu L.; Gouverneur V.; Shen Q. CCS Chem. 2020, 2, 1921.
pmid: 18671436 |
|
|
(v) Huang H.; Lin H.; Wang M.; Liao J. Acta Chim. Sinica 2020, 78, 1229. (in Chinese)
doi: 10.6023/A20090424 pmid: 18671436 |
|
|
( 黄浩, 林华鑫, 王敏, 廖建, 化学学报, 2020, 78, 1229.)
doi: 10.6023/A20090424 pmid: 18671436 |
|
|
(w) Zhang R.; Xu B.; Zhang Z.; Zhang J. Acta Chim. Sinica 2020, 78, 245. (in Chinese)
doi: 10.6023/A20010019 pmid: 18671436 |
|
|
( 张荣华, 许冰, 张展鸣, 张俊良, 化学学报, 2020, 78, 245.)
doi: 10.6023/A20010019 pmid: 18671436 |
|
|
(x) Zhang G.; Liang Y.; Qin T.; Xiong T.; Liu S.; Guan W.; Zhang Q. CCS Chem. 2020, 2, 1737.
pmid: 18671436 |
|
|
(y) Wu F.-P.; Holz J.; Yuan Y.; Wu X.-F. CCS Chem. 2020, 2, 2643.
pmid: 18671436 |
|
|
(z) Jiang C.; Chen P.; Liu G. CCS Chem. 2020, 2, 1884.
pmid: 18671436 |
|
| [5] |
(a) Hoveyda A. H.; Evans D. A.; Fu G. C. Chem. Rev. 1993, 93, 1307.
doi: 10.1021/cr00020a002 pmid: 28125210 |
|
(b) Corey E. J.; Lalic G. Org. Lett. 2007, 9, 4921.
doi: 10.1021/ol702323s pmid: 28125210 |
|
|
(c) König C. M.; Gebhardt B.; Schleth C.; Dauber M.; Koert U. Org. Lett. 2009, 11, 2728.
doi: 10.1021/ol900757k pmid: 28125210 |
|
|
(d) Hickmann V.; Alcarazo M.; Fürstner A. J. Am. Chem. Soc. 2010, 132, 11042.
doi: 10.1021/ja104796a pmid: 28125210 |
|
|
(e) May T. L.; Dabrowski J. A.; Hoveyda A. H. J. Am. Chem. Soc. 2011, 133, 736.
doi: 10.1021/ja110054q pmid: 28125210 |
|
|
(f) McDonald R. I.; Liu G.; Stahl S. S. Chem. Rev. 2011, 111, 2981.
doi: 10.1021/cr100371y pmid: 28125210 |
|
|
(g) Dong Z.; Ren Z.; Thompson S. J.; Xu Y.; Dong G. Chem. Rev. 2017, 117, 9333.
doi: 10.1021/acs.chemrev.6b00574 pmid: 28125210 |
|
|
(h) Yan T.; Guironnet D. Sci. Chin. Chem. 2020, 63, 755.
doi: 10.1007/s11426-020-9724-9 pmid: 28125210 |
|
| [6] |
(a) Denmark S. E.; Almstead N. G. Modern Carbonyl Chemistry, Ed.: Otera, J., Wiley-VCH, Weinheim, 2000, Chapter 10, p. 299.
|
|
(b) Yamamoto Y.; Asao N. Chem. Rev. 1993, 93. 2207.
doi: 10.1021/cr00022a010 |
|
|
(c) Kennedy J. W. J.; Hall D. G. Angew. Chem. Int. Ed. 2003, 42, 4732.
doi: 10.1002/(ISSN)1521-3773 |
|
|
(d) Chrétien J.-M.; Zammattio F.; Gauthier D.; Grognec E. L.; Paris M.; Quintard J.-P. Chem. Eur. J. 2006, 12, 6816.
doi: 10.1002/(ISSN)1521-3765 |
|
|
(e) Yamamoto H.; Wadamoto M. Chem. Asian J. 2007, 2, 692.
doi: 10.1002/(ISSN)1861-471X |
|
|
(f) Yus M.; González-Gómez J. C.; Foubelo F. Chem. Rev. 2011, 111, 7774.
doi: 10.1021/cr1004474 |
|
| [7] |
(a) Hokanson G. C.; French J. C. J. Org. Chem. 1985, 50, 462.
doi: 10.1021/jo00204a007 |
|
(b) Shevchenko V. P.; Myagkova G. I.; Lazurkina T. Y.; Dyomin P. M.; Shrarn S. I.; Zabolotsky D. A.; Nagayev I. Y.; Belosludtsev Y. Y.; Evstigneeva R. P.; Myasoyedov N. F. J. Label. Compd. Radiopharm. 1989, 27, 1177.
doi: 10.1002/jlcr.v27:10 |
|
|
(c) Yoshida W. Y.; Bryan P. J.; Baker B. J.; McClintock J. B. J. Org. Chem. 1995, 60, 780.
doi: 10.1021/jo00108a057 |
|
|
(d) Inoue M.; Nakada M. J. Am. Chem. Soc. 2007, 129, 4164.
doi: 10.1021/ja070812w |
|
|
(e) Lachance H.; Hall D. G. Organic Reactions, Vol. 73, Eds.: Denmark, S. E. et al. John Wiley & Sons, Inc., 2008, Chapter 1, p. 1.
|
|
| [8] |
Huang Y.; Huang R.-Z.; Zhao Y. J. Am. Chem. Soc. 2016, 138, 6571.
doi: 10.1021/jacs.6b02372 pmid: 27139596 |
| [9] |
Zhang H.; Huang W.; Wang T.; Meng F. Angew. Chem. Int. Ed. 2019, 58, 11049.
doi: 10.1002/anie.v58.32 |
| [10] |
Zhou Y.; Wang L.; Yuan G.; Liu S.; Sun X.; Yuan C.; Yang Y.; Bian Q.; Wang M.; Zhong J. Org. Lett. 2020, 22, 4532.
doi: 10.1021/acs.orglett.0c01557 pmid: 32432878 |
| [11] |
(a) Brak K.; Ellman J. A. Org. Lett. 2010, 12, 2004.
doi: 10.1021/ol100470g pmid: 10821714 |
|
(b) Charest M. G.; Siegel D. R.; Myers A. G. J. Am. Chem. Soc. 2005, 127, 8292.
pmid: 10821714 |
|
|
(c) Corbett J. W.; Ko S. S.; Rodgers J. D.; Gearhart L. A.; Magnus N. A.; Bacheler L. T.; Diamond S.; Jeffrey S.; Klabe R. M.; Cordova B. C.; Garber S.; Logue K.; Trainor G. L.; Anderson P. S.; Erickson-Viitanen S. K. J. Med. Chem. 2000, 43, 2019.
pmid: 10821714 |
|
| [12] |
Wang X.; Quan M.; Xie F.; Yang G.; Zhang W. Tetrahedron Lett. 2018, 59, 1573.
doi: 10.1016/j.tetlet.2018.03.026 |
| [13] |
Quan M.; Wang X.; Wu L.; Gridnev I. D.; Yang G.; Zhang W. Nat. Commun. 2018, 9, 2258.
doi: 10.1038/s41467-018-04645-3 pmid: 29884893 |
| [14] |
Lv X.-Y.; Fan C.; Xiao L.-J.; Xie J.-H.; Zhou Q.-L. CCS Chem. 2019, 1, 328.
doi: 10.31635/ccschem.019.20190026 |
| [15] |
Chen Y.-G.; Shuai B.; Xu X.-T.; Li Y.-Q.; Yang Q.-L.; Qiu H.; Zhang K.; Fang P.; Mei T.-S. J. Am. Chem. Soc. 2019, 141, 3395.
doi: 10.1021/jacs.8b13524 |
| [16] |
Wang Z.-C.; Gao J.; Cai Y.; Ye X.; Shi S.-L. CCS Chem. 2021, 3, 1445.
doi: 10.31635/ccschem.020.202000356 |
| [17] |
Dai X.; Strotman N. A.; Fu G. C. J. Am. Chem. Soc. 2008, 130, 3302.
doi: 10.1021/ja8009428 |
| [18] |
Lou S.; Fu G. C. J. Am. Chem. Soc. 2010, 132, 5010.
doi: 10.1021/ja1017046 |
| [19] |
Choi J.; Fu G. C. J. Am. Chem. Soc. 2012, 134, 9102.
doi: 10.1021/ja303442q |
| [20] |
(a)Organosulfur Chemistry in Asymmetric Synthesis, Eds.: Toru, T.; Bolm, C., Wiley-VCH, Weinheim, 2008.
pmid: 22664250 |
|
(b) Wilden J. D. J. Chem. Res. 2010, 34, 541.
pmid: 22664250 |
|
|
(c) Chen X.; Hussain S.; Parveen S.; Zhang S.; Yang Y.; Zhu C. Curr. Med. Chem. 2012, 19, 3578.
pmid: 22664250 |
|
|
(d) Shah S. S. A.; Rivera G.; Ashfaq M. Mini-Rev. Med. Chem. 2013, 13, 70.
pmid: 22664250 |
|
|
(e) Scozzafava A.; Carta F.; Supuran C. T. Expert Opin. Ther. Patents 2013, 23, 203.
doi: 10.1517/13543776.2013.742065 pmid: 22664250 |
|
| [21] |
Choi J.; Martín-Gago P.; Fu G. C. J. Am. Chem. Soc. 2014, 136, 12161.
doi: 10.1021/ja506885s |
| [22] |
Wang Z.; Yin H.; Fu G. C. Nature 2018, 563, 379.
doi: 10.1038/s41586-018-0669-y |
| [23] |
Wu L.; Yang G.; Zhang W. CCS Chem. 2019, 1, 623.
|
| [24] |
Wang Z.; Yang Z.-P.; Fu G. C. Nat. Chem. 2021, 13, 236.
doi: 10.1038/s41557-020-00609-7 |
| [25] |
Tomita D.; Wada R.; Kanai M.; Shibasaki M. J. Am. Chem. Soc. 2005, 127, 4138.
doi: 10.1021/ja0507362 |
| [26] |
Tomita D.; Kanai M.; Shibasaki M. Chem. Asian. J. 2006, 1-2, 161.
|
| [27] |
Lipshutz B. H.; Pegram J. J.; Morey M. C. Tetrahedron Lett. 1981, 22, 4603.
doi: 10.1016/S0040-4039(01)82992-0 |
| [28] |
(a) Lin G.-Q.; You Q.-D.; Cheng J.-F. Chiral Drugs: Chemistry and Biological Action, Wiley, Hoboken, NJ, 2011.
pmid: 17901324 |
|
(b) Chambers R. D. Fluorine in Organic Chemistry, Blackwell, Oxford, 2004.
pmid: 17901324 |
|
|
(c) Shah P.; Westwell A. D. J. Enzyme Inhib. Med. Chem. 2007, 22, 527.
doi: 10.1080/14756360701425014 pmid: 17901324 |
|
|
(d) Müller K.; Faeh C.; Diederich F. Science 2007, 317, 1881.
pmid: 17901324 |
|
|
(e) Lv C.-P.; Shen Q.-L.; Liu D. Chin. J. Org. Chem. 2012, 32, 1380. (in Chinese)
doi: 10.6023/cjoc1110151 pmid: 17901324 |
|
|
( 吕翠萍, 沈其龙, 刘丹, 有机化学, 2012, 32, 1380.)
doi: 10.6023/cjoc1110151 pmid: 17901324 |
|
| [29] |
Motoki R.; Tomita D.; Kanai M.; Shibasaki M. Tetrahedron Lett. 2006, 47, 8083.
doi: 10.1016/j.tetlet.2006.09.048 |
| [30] |
Tomita D.; Yamatsugu K.; Kanai M.; Shibasaki M. J. Am. Chem. Soc. 2009, 131, 6946.
doi: 10.1021/ja901995a |
| [31] |
Müller D.; Hawner C.; Tissot M.; Palais L.; Alexakis A. Synlett 2010, 11, 1694.
|
| [32] |
(a) Müller D.; Tissot M.; Alexakis A. Org. Lett. 2011, 13, 3040.
doi: 10.1021/ol200898c |
|
(b) Müller D.; Alexakis A. Chem. Eur. J. 2013, 19, 15226.
doi: 10.1002/chem.201302856 |
|
| [33] |
Müller D.; Alexakis A. Org. Lett. 2012, 14, 1842.
doi: 10.1021/ol3004436 |
| [34] |
Cottet P.; Müller D.; Alexakis A. Org. Lett. 2013, 15, 828.
doi: 10.1021/ol303505k |
| [35] |
May T. L.; Dabrowski J. A.; Hoveyda A. H. J. Am. Chem. Soc. 2011, 133, 736.
doi: 10.1021/ja110054q |
| [36] |
Müller D. S.; Werner V.; Akyol S.; Schmalz H.-G.; Marek I. Org. Lett. 2017, 19, 3970.
doi: 10.1021/acs.orglett.7b01661 |
| [37] |
Chong Q.; Yue Z.; Zhang S.; Ji C.; Cheng F.; Zhang H.; Hong X.; Meng F. ACS Catal. 2017, 7, 5693.
doi: 10.1021/acscatal.7b02132 |
| [38] |
Mcgrath K. P.; Hubbell A. K.; Zhou Y.; Santos D. P.; Torker S.; Romiti F.; Hoveyda A. H. Adv. Synth. Catal. 2020, 362, 370.
doi: 10.1002/adsc.v362.2 |
| [39] |
(a) Trost B. M.; Van Vranken D. L. Chem. Rev. 1996, 96, 395.
doi: 10.1021/cr9409804 |
|
(b) Trost B. M.; Crawley M. L. Chem. Rev. 2003, 103, 2921.
doi: 10.1021/cr020027w |
|
|
(c) Lu Z.; Ma S. Angew. Chem. Int. Ed. 2008, 47, 258.
|
|
|
(d) Diéguez M.; Pàmies O. Acc. Chem. Res. 2010, 43, 312.
doi: 10.1021/ar9002152 |
|
|
(e) Weaver J. D.; Recio A.; Grenning A. J.; Tunge J. A. Chem. Rev. 2011, 111, 1846.
doi: 10.1021/cr1002744 |
|
|
(f) Huo X.; Yang G.; Liu D.; Liu Y.; Gridnev I. D.; Zhang W. Angew. Chem. Int. Ed. 2014, 53, 6776.
doi: 10.1002/anie.201403410 |
|
|
(g) Huo X.; He R.; Zhang X.; Zhang W. J. Am. Chem. Soc. 2016, 138, 11093.
doi: 10.1021/jacs.6b06156 |
|
|
(h) Tang H.; Huo X.; Meng Q.; Zhang W. Acta Chim. Sinica 2016, 74, 219. (in Chinese)
doi: 10.6023/A16020078 |
|
|
( 汤溟淏, 霍小红, 孟庆华, 张万斌, 化学学报, 2016, 74, 219.)
|
|
|
(i) Huo X.; He R.; Fu J.; Zhang J.; Yang G.; Zhang W. J. Am. Chem. Soc. 2017, 139, 9819.
doi: 10.1021/jacs.7b05460 |
|
|
(j) Huo X.; Zhang J.; Fu J.; Zhang W. J. Am. Chem. Soc. 2018, 140, 2080.
doi: 10.1021/jacs.8b00187 |
|
|
(k) Zhang M.-M.; Luo Y.-Y.; Lu L.-Q.; Xiao W.-J. Acta Chim. Sinica 2018, 76, 838. (in Chinese)
doi: 10.6023/A18060237 |
|
|
( 张毛毛, 骆元元, 陆良秋, 肖文精, 化学学报, 2018, 76, 838.)
doi: 10.6023/A18060237 |
|
|
(l) Li Z.; Zheng J.; Li C.; Wu W.; Jiang H. Chin. J. Chem. 2019, 37, 140.
doi: 10.1002/cjoc.v37.2 |
|
|
(m) Wang R.; Luan Y.; Ye M. Chin. J. Chem. 2019, 37, 720.
doi: 10.1002/cjoc.v37.7 |
|
|
(n) Yao K.; Liu H.; Yuan Q.; Liu Y.; Liu D.; Zhang W. Acta Chim. Sinica 2019, 77, 993. (in Chinese)
doi: 10.6023/A19060210 |
|
|
( 姚坤, 刘浩, 袁乾家, 刘燕刚, 刘德龙, 张万斌, 化学学报, 2019, 77, 993.)
doi: 10.6023/A19060210 |
|
|
(o) Zhang H.-H.; Yu S. Acta Chim. Sinica 2019, 77, 832. (in Chinese)
doi: 10.6023/A19050177 |
|
|
( 张洪浩, 俞寿云, 化学学报, 2019, 77, 832.)
doi: 10.6023/A19050177 |
|
|
(p) Zhang H.; Gu Q.; You S. Chin. J. Org. Chem. 2019, 39, 15. (in Chinese)
|
|
|
( 张慧君, 顾庆, 游书力, 有机化学, 2019, 39, 15.)
doi: 10.6023/cjoc201809037 |
|
|
(q) Huang L.; Cai Y.; Zhang H.-J.; Zheng C.; Dai L.-X.; You S.-L. CCS Chem. 2019, 1, 106.
|
|
|
(r) Wang R.-Q.; Shen C.; Cheng X.; Wang Z.-F.; Tao H.-Y.; Dong X.-Q.; Wang C.-J. Chin. J. Chem. 2020, 38, 807.
doi: 10.1002/cjoc.v38.8 |
|
|
(s) He R.; Huo X.; Zhao L.; Wang F.; Jiang L.; Liao J.; Zhang W. J. Am. Chem. Soc. 2020, 142, 8097.
doi: 10.1021/jacs.0c02150 |
|
|
(t) Wang Y.; Luo S. Chin. J. Org. Chem. 2020, 40, 2161. (in Chinese)
doi: 10.6023/cjoc202000038 |
|
|
( 王娅宁, 罗三中, 有机化学, 2020, 40, 2161.)
doi: 10.6023/cjoc202000038 |
|
|
(u) Ju C.; Wu Z.; Li Y.; Zhang W. Chin. J. Org. Chem. 2020, 40, 3925. (in Chinese)
doi: 10.6023/cjoc202004025 |
|
|
( 居辰阳, 吴正兴, 李云艺, 张万斌, 有机化学, 2020, 40, 3925.)
doi: 10.6023/cjoc202004025 |
|
|
(v) Li G.; Huo X.; Jiang X.; Zhang W. Chem. Soc. Rev. 2020, 49, 2060.
doi: 10.1039/C9CS00400A |
|
|
(w) Ma X.; Yu J.; Wang Z.; Zhang Y.; Zhou Q. Chin. J. Org. Chem. 2020, 40, 2669. (in Chinese)
doi: 10.6023/cjoc202005013 |
|
|
( 马献涛, 于静, 王子龙, 张赟, 周秋菊, 有机化学, 2020, 40, 2669.)
doi: 10.6023/cjoc202005013 |
|
|
(x) Tian F.; Zhang J.; Yang W.; Deng W. Chin. J. Org. Chem. 2020, 40, 3262. (in Chinese)
doi: 10.6023/cjoc202005008 |
|
|
( 田飞, 张键, 杨武林, 邓卫平, 有机化学, 2020, 40, 3262.)
doi: 10.6023/cjoc202005008 |
|
|
(y) Huo X.; Zhao L.; Luo Y.; Wu Y.; Sun Y.; Li G.; Gridneva T.; Zhang J.; Ye Y.; Zhang W. CCS Chem. 2021, 3, 1933.
|
|
|
(z) Xiao J.; Xu H.; Huo X.; Zhang W. Ma S. Chin. J. Chem. 2021, 39, 1958.
doi: 10.1002/cjoc.v39.7 |
|
| [40] |
Lee Y.; Akiyama K.; Gillingham D. G.; Brown M. K.; Hoveyda A. H. J. Am. Chem. Soc. 2008, 130, 446.
doi: 10.1021/ja0782192 |
| [41] |
Gao F.; Mcgrath K. P.; Lee Y.; Hoveyda A. H. J. Am. Chem. Soc. 2010, 132, 14315.
doi: 10.1021/ja106829k |
| [42] |
Akiyama K.; Gao F.; Hoveyda A. H. Angew. Chem. Int. Ed. 2010, 49, 419.
doi: 10.1002/anie.v49:2 |
| [43] |
Gao F.; Carr J. L.; Hoveyda A. H. Angew. Chem. Int. Ed. 2012, 51, 6613.
doi: 10.1002/anie.201202856 |
| [44] |
Gao F.; Carr J. L.; Hoveyda A. H. J. Am. Chem. Soc. 2014, 136, 2149.
doi: 10.1021/ja4126565 |
| [45] |
Trost B. M.; Hung C.-I.; Koester D. C.; Miller Y. Org. Lett. 2015, 17, 3778.
doi: 10.1021/acs.orglett.5b01755 |
| [46] |
Jin M.; Adak L.; Nakamura M. J. Am. Chem. Soc. 2015, 137, 7128.
doi: 10.1021/jacs.5b02277 |
| [47] |
(a) Hatakeyama T.; Hashimoto T.; Kondo Y.; Fujiwara Y.; Seike H.; Takaya H.; Tamada Y.; Ono T.; Nakamura M. J. Am. Chem. Soc. 2010, 132, 10674.
doi: 10.1021/ja103973a pmid: 20681696 |
|
(b) Jin M.; Nakamura M. Chem. Lett. 2011, 40, 1012.
doi: 10.1246/cl.2011.1012 pmid: 20681696 |
|
|
(c) Hatakeyama T.; Fujiwara Y.; Okada Y.; Itoh T.; Hashimoto T.; Kawamura S.; Ogata K.; Takaya H.; Nakamura M. Chem. Lett. 2011, 40, 1030.
doi: 10.1246/cl.2011.1030 pmid: 20681696 |
|
| [48] |
(a) Adams C. J.; Bedford R. B.; Carter E.; Gower N. J.; Haddow M. F.; Harvey J. N.; Huwe M.; Cartes M. Á.; Mansell S. M.; Mendoza C.; Murphy D. M.; Neeve E. C.; Nunn J. J. Am. Chem. Soc. 2012, 134, 10333.
doi: 10.1021/ja303250t |
|
(b) Bedford R. B.; Brenner P. B.; Carter E.; Clifton J.; Cogswell P. M.; Gower N. J.; Haddow M. F.; Harvey J. N.; Kehl J. A.; Murphy D. M.; Neeve E. C.; Neidig M. L.; Nunn J.; Snyder B. E. R.; Taylor J. Organometallics 2014, 33, 5767.
doi: 10.1021/om500518r |
|
| [49] |
Evans D. A.; Aye Y. J. Am. Chem. Soc. 2006, 128, 11034.
doi: 10.1021/ja063878k |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||