Acta Chimica Sinica ›› 2021, Vol. 79 ›› Issue (7): 914-919.DOI: 10.6023/A21030121 Previous Articles Next Articles
Article
投稿日期:2021-03-30
发布日期:2021-06-16
通讯作者:
刘洪来
基金资助:
Wangping Ma, Yanyan He, Honglai Liu( )
)
Received:2021-03-30
Published:2021-06-16
Contact:
Honglai Liu
Supported by:Share
Wangping Ma, Yanyan He, Honglai Liu. Olefin-linked Conjugated Porous Networks and Their Visible-Light-Driven Hydrogen Evolution Performance[J]. Acta Chimica Sinica, 2021, 79(7): 914-919.

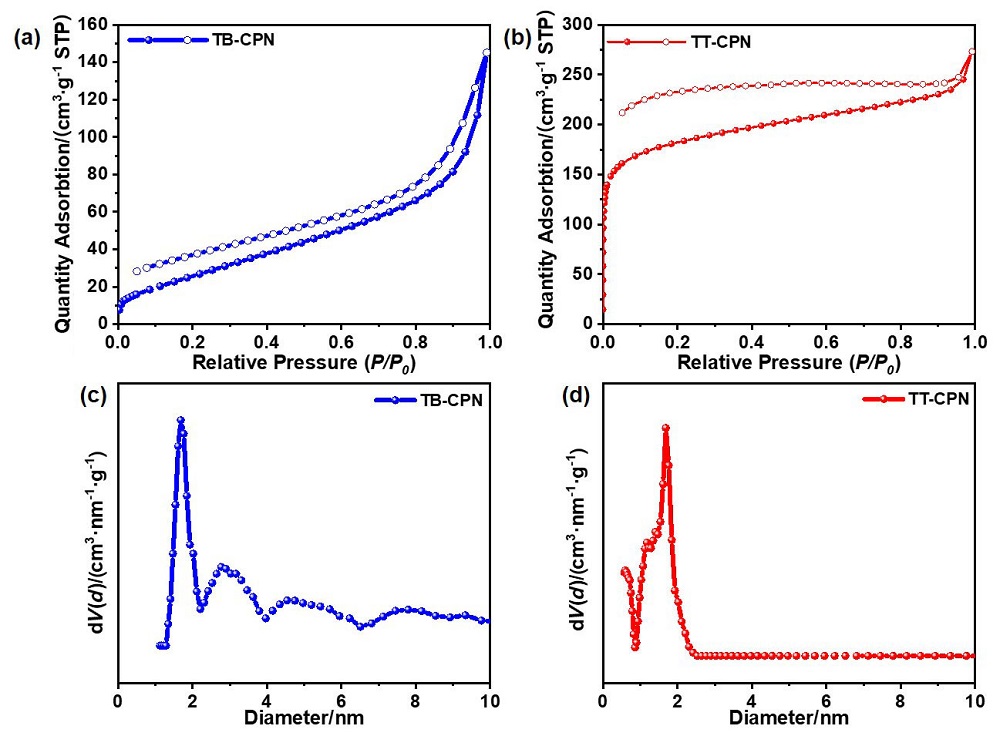
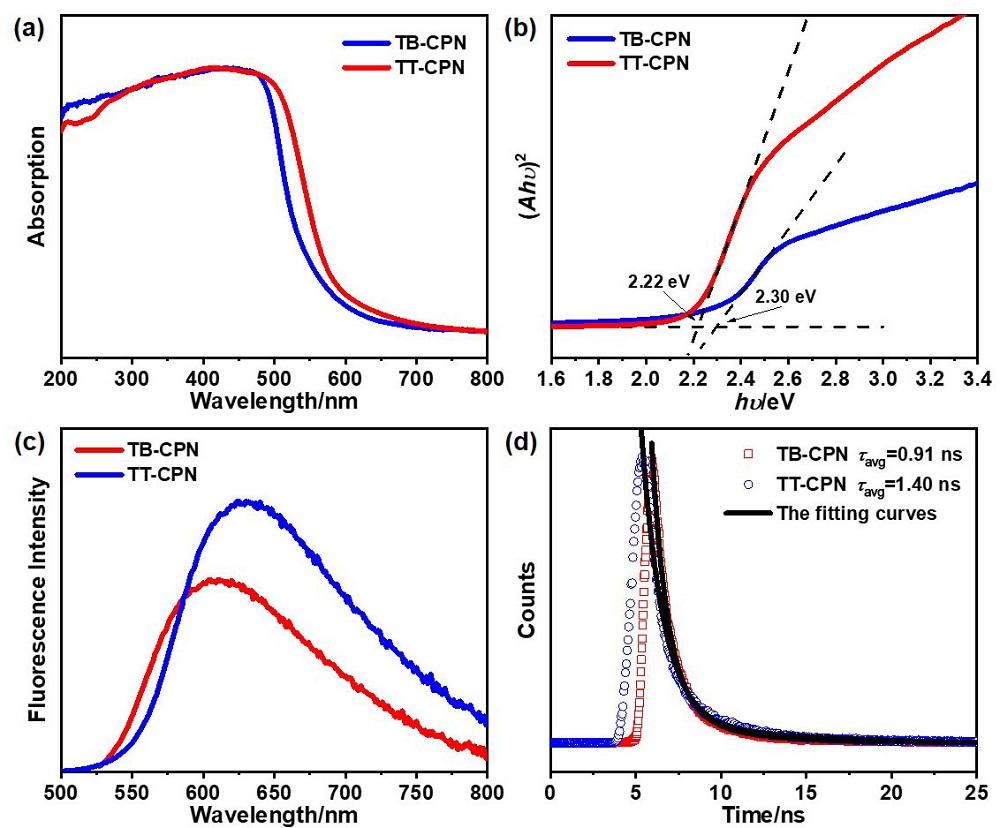
| Sample | SBETa/ (m2•g-1) | Eoptb/ eV | Evc/Ecd (V vs. SHE) | HERvise/ (μmol•h-1•g-1) |
|---|---|---|---|---|
| TB-CPN | 103 | 2.30 | 4.75/2.45 | 792.2 |
| TT-CPN | 620 | 2.22 | 5.12/2.90 | 913.3 |
| Sample | SBETa/ (m2•g-1) | Eoptb/ eV | Evc/Ecd (V vs. SHE) | HERvise/ (μmol•h-1•g-1) |
|---|---|---|---|---|
| TB-CPN | 103 | 2.30 | 4.75/2.45 | 792.2 |
| TT-CPN | 620 | 2.22 | 5.12/2.90 | 913.3 |

| [1] |
Zou, X.; Zhang, Y. Chem. Soc. Rev. 2015, 44, 5148.
doi: 10.1039/C4CS00448E |
| [2] |
Rahman, M. Z.; Kibria, M. G.; Mullins, C. B. Chem. Soc. Rev. 2020, 49, 1887.
doi: 10.1039/C9CS00313D |
| [3] |
Fujishima, A.; Honda, K. Nature 1972, 238, 37.
doi: 10.1038/238037a0 |
| [4] |
Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J. M.; Domen, K.; Antonietti, M. Nature Mater. 2009, 8, 76.
doi: 10.1038/nmat2317 |
| [5] |
Xing, J.; Chen, J. F.; Li, Y. H.; Yuan, W. T.; Zhou, Y.; Zheng, L. R.; Wang, H. F.; Hu, P.; Wang, Y.; Zhao, H. J.; Wang, Y.; Yang, H. G. Chemistry 2014, 20, 2138.
|
| [6] |
Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S. T.; Zhong, J.; Kang, Z. Science 2015, 347, 970.
doi: 10.1126/science.aaa3145 |
| [7] |
Li, C.; Chen, F.; Ye, L.; Li, W.; Yu, H.; Zhao, T. Acta Chim. Sinica 2020, 78, 1448.
doi: 10.6023/A20070322 |
|
(李宸, 陈凤华, 叶丽, 李伟, 于晗, 赵彤, 化学学报, 2020, 78, 1448).
|
|
| [8] |
Guo, Y.; Li, Y.; Wang, C.; Long, R.; Xiong, Y. Acta Chim. Sinica 2019, 77, 520.
doi: 10.6023/A19040108 |
|
(郭宇, 李燕瑞, 王成名, 龙冉, 熊宇杰, 化学学报, 2019, 77, 520).
|
|
| [9] |
Xu, Z.-Y.; Luo, Y.; Wang, H.; Zhang, D.-W.; Li, Z.-T. Chin. J. Org. Chem. 2020, 40, 3777.
doi: 10.6023/cjoc202003070 |
|
(徐子悦, 罗驿, 王辉, 张丹维, 黎占亭, 有机化学, 2020, 40, 3777).
|
|
| [10] |
Zhang, G.; Lan, Z. A.; Wang, X. Angew. Chem. Int. Ed. 2016, 55, 15712.
doi: 10.1002/anie.201607375 |
| [11] |
Dai, C.; Liu, B. Energy Environ. Sci. 2020, 13, 24.
doi: 10.1039/C9EE01935A |
| [12] |
Ma, X.; Wang, H.; Cheng, J.; Cheng, H.; Wang, L.; Wu, X.; Xu, H. Chin. J. Chem. 2021, 39, 1079.
doi: 10.1002/cjoc.v39.5 |
| [13] |
Banerjee, T.; Gottschling, K.; Savasci, G.; Ochsenfeld, C.; Lotsch, B. V. ACS Energy Lett. 2018, 3, 400.
doi: 10.1021/acsenergylett.7b01123 pmid: 29457140 |
| [14] |
Wang, X.; Chen, L.; Chong, S. Y.; Little, M. A.; Wu, Y.; Zhu, W. H.; Clowes, R.; Yan, Y.; Zwijnenburg, M. A.; Sprick, R. S.; Cooper, A. I. Nature Chem. 2018, 10, 1180.
doi: 10.1038/s41557-018-0141-5 |
| [15] |
Yu, K.; Bi, S.; Ming, W.; Wei, W.; Zhang, Y.; Xu, J.; Qiang, P.; Qiu, F.; Wu, D.; Zhang, F. Polym. Chem. 2019, 10, 3758.
doi: 10.1039/C9PY00512A |
| [16] |
Kong, D.; Han, X.; Xie, J.; Ruan, Q.; Windle, C. D.; Gadipelli, S.; Shen, K.; Bai, Z.; Guo, Z.; Tang, J. ACS Catal. 2019, 9, 7697.
doi: 10.1021/acscatal.9b02195 pmid: 32064148 |
| [17] |
Huang, W.; He, Q.; Hu, Y.; Li, Y. Angew. Chem. Int. Ed. 2019, 58, 8676.
doi: 10.1002/anie.v58.26 |
| [18] |
Horner, L.; Hoffmann, H.; Wipp, H. G. Chem. Ber. 1958, 91, 61.
doi: 10.1002/(ISSN)1099-0682 |
| [19] |
Wadsworth., W. S.; Emmons, W. D. J. Am. Chem. Soc. 1961, 83, 1733.
doi: 10.1021/ja01468a042 |
| [20] |
Ando, K. J. Org. Chem. 1998, 63, 8411.
doi: 10.1021/jo981337a |
| [21] |
Shi, M.; Yamamoto, K.; Okamoto, Y.; Takamuku, S. Phosphorus Sulfur 1991, 60, 1.
doi: 10.1080/10426509108233919 |
| [22] |
Xu, J.; He, Y.; Bi, S.; Wang, M.; Yang, P.; Wu, D.; Wang, J.; Zhang, F. Angew. Chem. Int. Ed. 2019, 58, 12065.
doi: 10.1002/anie.v58.35 |
| [23] |
Bi, S.; Thiruvengadam, P.; Wei, S.; Zhang, W.; Zhang, F.; Gao, L.; Xu, J.; Wu, D.; Chen, J. S.; Zhang, F. J. Am. Chem. Soc. 2020, 142, 11893.
doi: 10.1021/jacs.0c04594 |
| [24] |
Zhuang, X.; Zhao, W.; Zhang, F.; Cao, Y.; Liu, F.; Bi, S.; Feng, X. Polym. Chem. 2016, 7, 4176.
doi: 10.1039/C6PY00561F |
| [25] |
Zhu, X.; Tian, C.; Mahurin, S. M.; Chai, S. H.; Wang, C.; Brown, S.; Veith, G. M.; Luo, H.; Liu, H.; Dai, S. J. Am. Chem. Soc. 2012, 134, 10478.
doi: 10.1021/ja304879c |
| [26] |
Jiang, J.-X.; Trewin, A.; Adams, D. J.; Cooper, A. I. Chem. Sci. 2011, 2, 1777.
doi: 10.1039/c1sc00329a |
| [27] |
Aitchison, C. M.; Sachs, M.; Little, M. A.; Wilbraham, L.; Brownbill, N. J.; Kane, C. M.; Blanc, F.; Zwijnenburg, M. A.; Durrant, J. R.; Sprick, R. S.; Cooper, A. I. Chem. Sci. 2020, 11, 8744.
doi: 10.1039/D0SC02675A |
| [28] |
Vyas, V. S.; Haase, F.; Stegbauer, L.; Savasci, G.; Podjaski, F.; Ochsenfeld, C.; Lotsch, B. V. Nat. Commun. 2015, 6, 8508.
doi: 10.1038/ncomms9508 |
| [29] |
Xu, Y.; Mao, N.; Zhang, C.; Wang, X.; Zeng, J.; Chen, Y.; Wang, F.; Jiang, J. Appl. Catal. B 2018, 228, 1.
|
| [30] |
Mothika, V. S.; Sutar, P.; Verma, P.; Das, S.; Pati, S. K.; Maji, T. K. Chemistry 2019, 25, 3867.
|
| [31] |
Sachs, M.; Cha, H.; Kosco, J.; Aitchison, C. M.; Francàs, L.; Corby, S.; Chiang, C.-L.; Wilson, A. A.; Godin, R.; Fahey-Williams, A.; Cooper, A. I.; Sprick, R. S.; McCulloch, I.; Durrant, J. R. J. Am. Chem. Soc. 2020, 142, 14574.
doi: 10.1021/jacs.0c06104 |
| [32] |
Wu, Q.; Zhang, C.; Sun, K.; Jiang, H.-L. Acta Chim. Sinica 2020, 78, 688.
doi: 10.6023/A20050141 |
|
(吴浅耶, 张晨曦, 孙康, 江海龙, 化学学报, 2020, 78, 688).
|
|
| [33] |
Jiang, Y.; Wang, Y.; Hua, J.; Tang, J.; Li, B.; Qian, S.; Tian, H. Chem. Commun. 2010, 46, 4689.
doi: 10.1039/c0cc00803f |
| [34] |
Jiang, Y.; Wang, Y.; Wang, B.; Yang, J.; He, N.; Qian, S.; Hua, J. Chem. Asian J. 2011, 6, 157.
doi: 10.1002/asia.201000536 |
| [35] |
Lin, Z.; Emge, T. J.; Warmuth, R. Chemistry 2011, 17, 9395.
|
| [36] |
Pradhan, B.; Pathak, S. K.; Gupta, R. K.; Gupta, M.; Pal, S. K.; Achalkumar, A. S. J. Mater. Chem. C 2016, 4, 6117.
doi: 10.1039/C6TC01260D |
| [37] |
Huang, T.; Lin, X.; Liu, Y.; Zhao, J.; Lin, H.; Xu, Z.; Zhong, S.; Zhang, C.; Wang, X.; Fu, X.; Long, J. ChemSusChem 2020, 13, 672.
doi: 10.1002/cssc.201903334 pmid: 31883308 |
| [1] | Jianqiang Chen, Gangguo Zhu, Jie Wu. Recent Advances in Nickel-Catalyzed Ring Opening Cross-Coupling of Aziridines [J]. Acta Chimica Sinica, 2024, 82(2): 190-212. |
| [2] | Yuhan Wu, Dongdong Zhang, Hongyu Yin, Zhengnan Chen, Wen Zhao, Yuhua Chi. Density Functional Theory Study of Janus In2S2X Photocatalytic Reduction of CO2 under “Double Carbon” Target [J]. Acta Chimica Sinica, 2023, 81(9): 1148-1156. |
| [3] | Minghui He, Ziqiu Ye, Guiqing Lin, Sheng Yin, Xinyi Huang, Xu Zhou, Ying Yin, Bo Gui, Cheng Wang. Research Progress of Porphyrin-Based Covalent Organic Frameworks in Photocatalysis★ [J]. Acta Chimica Sinica, 2023, 81(7): 784-792. |
| [4] | Jiawen Liu, Weihuang Lin, Weijia Wang, Xueyi Guo, Ying Yang. Synthesis and Photocatalytic Degradation of Cu1.94S-SnS Nano-heterojunction [J]. Acta Chimica Sinica, 2023, 81(7): 725-734. |
| [5] | Li Liu, Gang Zheng, Guoqiang Fan, Hongguang Du, Jiajing Tan. Research Progress in Organic Reactions Involving 4-Acyl/Carbamoyl/Alkoxycarbonyl Substituted Hantzsch Esters [J]. Acta Chimica Sinica, 2023, 81(6): 657-668. |
| [6] | Fei Li, Huili Ding, Chaozhong Li. Hydrotrifluoromethylation of Alkenes with a Fluoroform-Derived Trifluoromethylboron Complex [J]. Acta Chimica Sinica, 2023, 81(6): 577-581. |
| [7] | Qi Xueping, Wang Fei, Zhang Jian. A Post-Synthetic Method for the Construction of Titanium-Based Metal Organic Frameworks and Their Applications [J]. Acta Chimica Sinica, 2023, 81(5): 548-558. |
| [8] | Jianqiang Chen, Gangguo Zhu, Jie Wu. Recent Advances in Radical-Based Dehydroxylation of Hydroxyl Groups via Oxalates [J]. Acta Chimica Sinica, 2023, 81(11): 1609-1623. |
| [9] | Chunhui Yang, Jingchao Chen, Xinhan Li, Li Meng, Kaimin Wang, Weiqing Sun, Baomin Fan. Difluoroallylation of Silanes under Photoirradiation [J]. Acta Chimica Sinica, 2023, 81(1): 1-5. |
| [10] | Zhongshu Xie, Zhongxin Xue, Ziwen Xu, Qian Li, Hongyu Wang, Wei-Shi Li. Conjugated Crosslinking Modification of Graphitic Carbon Nitrides and Its Effect on Visible Light-Driven Photocatalytic Hydrogen Production [J]. Acta Chimica Sinica, 2022, 80(9): 1231-1237. |
| [11] | Yu Qi, Fuxiang Zhang. Photocatalytic Water Splitting for Hydrogen Production※ [J]. Acta Chimica Sinica, 2022, 80(6): 827-838. |
| [12] | Heng Shu, Yide-Rigen Bao, Yong Na. Photocatalytic Oxidation of 5-Hydroxymethylfurfural Selectively into 2,5-Diformylfuran with CdS Nanotube [J]. Acta Chimica Sinica, 2022, 80(5): 607-613. |
| [13] | Xue Gong, Xinguo Ma, Fengda Wan, Wangyang Duan, Xiaoling Yang, Jinrong Zhu. Study on the Electronic Structure and Optical Properties of Two-dimensional Monolayer MoSi2X4 (X=N, P, As) [J]. Acta Chimica Sinica, 2022, 80(4): 510-516. |
| [14] | Pan An, Qinghui Zhang, Zhuang Yang, Jiaxing Wu, Jiaying Zhang, Yajun Wang, Yuming Li, Guiyuan Jiang. Research Progress of Solar Hydrogen Production Technology under Double Carbon Target [J]. Acta Chimica Sinica, 2022, 80(12): 1629-1642. |
| [15] | Xiaohan Yu, Wei Huang, Yanguang Li. Controllable Synthesis and Photocatalytic Applications of Two-dimensional Covalent Organic Frameworks [J]. Acta Chimica Sinica, 2022, 80(11): 1494-1506. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||