Acta Chimica Sinica ›› 2023, Vol. 81 ›› Issue (7): 691-696.DOI: 10.6023/A23030074 Previous Articles Next Articles
Communication
刘洵a,b, 江辉波a, 荆凯强a, 徐忠宁a,*( ), 郭国聪a,*(
), 郭国聪a,*( )
)
投稿日期:2023-03-10
发布日期:2023-05-17
基金资助:
Xun Liua,b, Hui-Bo Jianga, Kai-Qiang Jinga, Zhong-Ning Xua( ), Guo-Cong Guoa(
), Guo-Cong Guoa( )
)
Received:2023-03-10
Published:2023-05-17
Contact:
*E-mail: Supported by:Share
Xun Liu, Hui-Bo Jiang, Kai-Qiang Jing, Zhong-Ning Xu, Guo-Cong Guo. Introduction of Zn2+ Promotes the Catalytic Performance of Pd-based Catalyst for CO Esterification Reaction via Electron Transfer[J]. Acta Chimica Sinica, 2023, 81(7): 691-696.
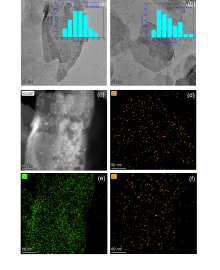

| [1] |
Zhao T. J.; Chen D.; Dai Y. C.; Yuan W. K.; Holmen A. Ind. Eng. Chem. Res. 2004, 43, 4595.
doi: 10.1021/ie030728z |
| [2] |
Hu Q.; Fan G.; Yang L.; Li F. ChemCatChem 2014, 6, 3501.
doi: 10.1002/cctc.201402674 |
| [3] |
He Z.; Lin H.; He P.; Yuan Y. J. Catal. 2011, 277, 54.
doi: 10.1016/j.jcat.2010.10.010 |
| [4] |
Yue H.; Zhao Y.; Ma X.; Gong J. Chem. Soc. Rev. 2012, 41, 4218.
doi: 10.1039/c2cs15359a |
| [5] |
Donald M.; Fenton P. J. S. J. Org. Chem. 1973, 39 701.
|
| [6] |
Peng S.-Y.; Xu Z.-N.; Chen Q.-S.; Wang Z.-Q.; Chen Y.; Lv D.-M.; Lu G.; Guo G.-C. Catal. Sci. Technol. 2014, 4, 1925.
doi: 10.1039/C4CY00245H |
| [7] |
Peng S.-Y.; Xu Z.-N.; Chen Q.-S.; Chen Y.-M.; Sun J.; Wang Z.-Q.; Wang M.-S.; Guo G.-C. Chem. Commun. 2013, 49, 5718.
doi: 10.1039/c3cc00219e |
| [8] |
Peng S.-Y.; Xu Z.-N.; Chen Q.-S.; Wang Z.-Q.; Lv D.-M.; Sun J.; Chen Y.; Guo G.-C. ACS Catal. 2015, 5, 4410.
doi: 10.1021/acscatal.5b00365 |
| [9] |
Hu C.; Jing K.-Q.; Lin X.-Q.; Sun J.; Xu Z.-N.; Guo G.-C. Catal. Lett. 2021, 22, 503.
|
| [10] |
Liu L.; Lin Z.; Lin S.; Chen Y.; Zhang L.; Chen S.; Zhang X.; Lin J.; Zhang Z.; Wan S.; Wang Y. J. Energy Chem. 2021, 58, 564.
doi: 10.1016/j.jechem.2020.10.003 |
| [11] |
Benavidez A. D.; Kovarik L.; Genc A.; Agrawal N.; Larsson E. M.; Hansen T. W.; Karim A. M.; Datye A. K. ACS Catal. 2012, 2, 2349.
doi: 10.1021/cs3005117 |
| [12] |
Thomas W.; Hansen A. T. D.; Sivakumar R.; Datye A. K. Acc. Chem. Res. 2013, 468, 1720.
|
| [13] |
Zhang K.; Zhang H.; Feng X.; Wang Y.; Wang G.; Zhu X.; Li C. Catal. Lett. 2022, 153, 921.
doi: 10.1007/s10562-022-04027-x |
| [14] |
Jin E.; Zhang Y.; He L.; Harris H. G.; Teng B.; Fan M. Applied Catalysis A: General. 2014, 476, 158.
doi: 10.1016/j.apcata.2014.02.035 |
| [15] |
Liu L.; Corma A. Chem. Rev. 2018, 118, 4981.
doi: 10.1021/acs.chemrev.7b00776 |
| [16] |
Jing K. Q.; Fu Y. Q.; Wang Z. Q.; Chen Z. N.; Tan H. Z.; Sun J.; Xu Z. N.; Guo G. C. Nanoscale 2020, 12, 14825.
doi: 10.1039/D0NR02987D |
| [17] |
Zheng J.; Lin H.; Wang Y.-N.; Zheng X.; Duan X.; Yuan Y. J. Catal. 2013, 297, 110.
doi: 10.1016/j.jcat.2012.09.023 |
| [18] |
Jiang H.-B.; Lin S.-S.; Xu Y.-P.; Sun J.; Xu Z.-N.; Guo G.-C. Acta Chim. Sinica 2022, 80, 438. (in Chinese)
doi: 10.6023/A21120619 |
|
(江辉波, 林珊珊, 徐玉平, 孙径, 徐忠宁, 郭国聪, 化学学报, 2022, 80, 438.)
|
|
| [19] |
Yu J.; Yang Y. S.; Wei M. Acta Chim. Sinica 2019, 77, 1129. (in Chinese)
doi: 10.6023/A19070260 |
|
(余俊, 杨宇森, 卫敏, 化学学报, 2019, 77, 1129)
|
|
| [20] |
Cui G.; Meng X.; Zhang X.; Wang W.; Xu S.; Ye Y.; Tang K.; Wang W.; Zhu J.; Wei M.; Evans D. G.; Duan X. Appl. Catal. B: Environ. 2019, 248, 394.
doi: 10.1016/j.apcatb.2019.02.042 |
| [21] |
Xu S.; Gu Q. Y.; Luo M. S. Appl. Chem. Ind. 2019, 48, 1. (in Chinese)
|
|
(徐舜, 谷庆阳, 罗明生, 应用化工, 2019, 48, 1.)
|
|
| [22] |
Lv Z.; Duan X. Chinese J. Catal. 2008, 29, 839. (in Chinese)
|
|
(吕志, 段雪, 催化学报, 2008, 29, 839.)
|
|
| [23] |
Li J. X.; Li B.; Wang J. K.; He L.; Zhao Y. F. Acta Chim. Sinica 2021, 79, 238. (in Chinese)
doi: 10.6023/A20090441 |
|
(李佳欣, 李蓓, 王纪康, 何蕾, 赵宇飞, 化学学报, 2021, 79, 238.)
|
|
| [24] |
Huang L.; Zou Y.; Chen D.; Wang S. Chinese J. Catal. 2019, 40, 1822.
doi: 10.1016/S1872-2067(19)63284-5 |
| [25] |
Xiong S.; Qian X.; Zhong Z.; Wang Y. J. Membrane Sci. 2022, 658, 120740.
doi: 10.1016/j.memsci.2022.120740 |
| [26] |
Huang Y.; Liu L. Sci. China Mater. 2019, 627, 913.
|
| [27] |
Amin M.; Shah N. A.; Bhatti A. S.; Malik M. A. CrystEngComm 2014, 16, 6080.
doi: 10.1039/C4CE00153B |
| [28] |
Wang F.; Zhao C.; Liu B.; Yuan S. J. Phys. D: Appl. Phys. 2009, 42, 115411.
doi: 10.1088/0022-3727/42/11/115411 |
| [1] | Junmin Chen, Chengqian Cui, Hanlin Liu, Guodong Li. Study on the Selective Hydrogenation of Quinoline Catalyzed by Composites of Metal-Organic Framework and Pt Nanoparticles※ [J]. Acta Chimica Sinica, 2022, 80(4): 467-475. |
| [2] | Yu Mohan, Cheng Yuanyuan, Liu Yajun. Mechanistic Study of Oxygenation Reaction in Firefly Bioluminescence [J]. Acta Chimica Sinica, 2020, 78(9): 989-993. |
| [3] | Qi Qige, Yang Chunfan, Xia Ye, Liu Kunhui, Su Hongmei. Photo-induced Electron Transfer between 4-Thiouracil and Tryptophan [J]. Acta Chimica Sinica, 2019, 77(6): 515-519. |
| [4] | Qiu Xuan, Shi Liang. Electrical Interplay between Microorganisms and Iron-bearing Minerals [J]. Acta Chim. Sinica, 2017, 75(6): 583-593. |
| [5] | Song Hao, Liu Xiaoyu, Qin Yong. Advances on Nitrogen-centered Radical Chemistry:A Photocatalytic N-H Bond Activation Approach [J]. Acta Chim. Sinica, 2017, 75(12): 1137-1149. |
| [6] | Yu Haibo, Li Hongling, Zhang Xinfu, Xiao Yi, Fang Peiju, Lv Chunjiao, Hou Wei. A Mitochondrial-Targetable and Turn-On Fluorescent Probe based on Nile Red and Monitoring for H2S in Living Cells [J]. Acta Chim. Sinica, 2015, 73(5): 450-456. |
| [7] | Kang Hongliang, Liu Ruigang, Huang Yong. Synthesis of Ethyl Cellulose Grafted Poly(N-isopropylacrylamide) Copolymer and Its Micellization [J]. Acta Chimica Sinica, 2013, 71(01): 114-120. |
| [8] | Wang Feng, Liang Wen-Jing, Wang Wen-Guang, Chen Bin, Feng Ke, Zhang Li-Ping, Tung Chen-Ho, Wu Li-Zhu. Bis-terpyridine Os(Ⅱ) Complex Sensitized [FeFe] Hydrogenase Mimic Systems: Synthesis and Photophysical Study [J]. Acta Chimica Sinica, 2012, 70(22): 2306-2310. |
| [9] | Chai Yunfeng, Gan Shifeng, Pan Yuanjiang. A Mechanistic Study of Formation of Radical Anion from Fragmentation of Deprotonated N,2-Diphenylacetamide Derivatives in Electrospray Ionization Tandem Mass Spectrometry [J]. Acta Chimica Sinica, 2012, 70(17): 1805-1811. |
| [10] | DONG Jiang-Zhou, ZHAO Jun-Yan, CHAO Hui, CAO Ya-An. Band Structure and Photo-induced Charge Transfer in Surface-sensitized TiO2-based Composite Films [J]. Acta Chimica Sinica, 2011, 69(23): 2781-2786. |
| [11] | DONG Yu-Pei, MOU Zhi-Gang, DU Yu-Kou, YANG Ping. Preparation, Electron Transfer and Photocatalysis of 4-(N,N-Diphenylamine) Benzyl Alcohol Functionalized Graphene [J]. Acta Chimica Sinica, 2011, 69(20): 2379-2384. |
| [12] | JIN Ying-Xue, YUAN Wang, QU Feng-Yu, YIN Xiong-Can, WEI Shu-Quan, YUE Qun-Feng, TAN Guang-Hui. Photoinduced Single Electron Transfer Cyclization Reactions of N-(ω-Tributylstannylpolyethoxyether)phthalimides [J]. Acta Chimica Sinica, 2011, 69(20): 2407-2412. |
| [13] | YIN Wei-Feng, OU Zhi-Ze , GAO Yun-Yan-*, HAO Ping, GUO Chuang-Long, WANG Zhong-Li. Interaction of 1,4-Dihydroxyanthraquinone and Its Yttrium Coordination Polymer with DNA [J]. Acta Chimica Sinica, 2010, 68(14): 1343-1348. |
| [14] | YIN An-Yuan, GUO Xiao-Yang, DAI Wei-Lin-*, FAN Kang-Nian. Textural Structure Effect of SiO2 on the Catalytic Performance for Hydrogenation of Dimethyl Oxalate to Ethylene Glycol over Different Cu-based Catalysts [J]. Acta Chimica Sinica, 2010, 68(13): 1285-1290. |
| [15] | CHEN Liang-Feng, SHU Yuan, LIU Xiao-Yu, FEI Yan, JIAO Meng-Hua, CHEN Wei, XU Hua-Long, FAN Kang-Nian. Cu/SiO2 Catalysts Prepared by Different Methods for Hydrogenolysis of Dimethyl Oxalate to Ethylene Glycol [J]. Acta Chimica Sinica, 2009, 67(23): 2739-2744. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||