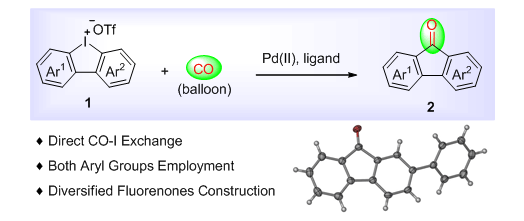
| [1] Selected examples, see:
(a) Krueger, R. F.; Mayer, G. D. Science 1970, 169, 1213;
(b) Greenlee, M. L.; Laub, J. B.; Rouen, G. P.; DiNinno, F.; Hammond, M. L.; Huber, J. L.; Sundelof, J. G.; Hammond, G. G. Bioorg. Med. Chem. Lett. 1999, 9, 3225;
(c) Perry, P. J.; Read, M. A.; Davies, R. T.; Gowan, S. M.; Reszka, A. P.; Wood, A. A.; Kelland, L. R.; Neidle, S. J. Med. Chem. 1999, 42, 2679.
[2] (a) Talapatra, S. K.; Bose, S.; Malik, A. K.; Talapatra, B. Tetrahedron 1985, 41, 2765;
(b) Jone, Jr., W. D.; Ciske, F. L. J. Org. Chem. 1996, 61, 3920.
[3] Selected examples, see:
(a) Uckert, F.; Tak, Y.-H.; Müllen, K.; Bässler, H. Adv. Mater. 2000, 12, 905;
(b) Gong, X.; Moses, D.; Heeger, A. J. J. Phys. Chem. B 2004, 108, 8601;
(c) Qin, C.; Islam, A.; Han, L. J. Mater. Chem. 2012, 22, 19236.
[4] Representative reviews, see:
(a) Wu, X.-F.; Neumann, H.; Beller, M. Chem. Soc. Rev. 2011, 40, 4986;
(b) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Chem. Rev. 2015, 115, 5301. Selected examples, see:
(c) Campo, M. A.; Larock, R. C. Org. Lett. 2000, 2, 3675;
(d) Campo, M. A.; Larock, R. C. J. Org. Chem. 2002, 67, 5616;
(e) Zhao, J.; Yue, D.; Campo, M. A.; Larock, R. C. J. Am. Chem. Soc. 2007, 129, 5288;
(f) Sun, C.-L.; Liu, N.; Li, B.-J.; Yu, D.-G.; Wang, Y.; Shi, Z.-J. Org. Lett. 2010, 12, 184;
(g) Li, H.; Zhu, R.-Y.; Shi, W.-J.; He, K.-H.; Shi, Z.-J. Org. Lett. 2012, 14, 4850;
(h) Gandeepan, P.; Hung, C.-H.; Cheng, C.-H. Chem. Commun. 2012, 48, 9379;
(i) Song, J.; Wei, F.; Sun, W.; Li, K.; Tian, Y.; Liu, C.; Li, Y.; Xie, L. Org. Lett. 2015, 17, 2106;
(j) Sun, D.; Li, B.; Lan, J.; Huang, Q.; You, J. Chem. Commun. 2016, 52, 3635;
(k) Wu, J.; Liu, Y.; Ma, X.; Liu, P.; Gu, C.; Dai, B. Chin. J. Chem. 2017, 35, 1391.
[5] Hartmann, C.; Meyer, V. Ber. Dtsch. Chem. Ges. 1894, 27, 426.
[6] Representative reviews, see:
(a) Stang, P. J.; Zhdankin, V. V. Chem. Rev. 1996, 96, 1123;
(b) Grushin, V. V. Chem. Soc. Rev. 2000, 29, 315;
(c) Deprez, N. R.; Sanford, M. S. Inorg. Chem. 2007, 46, 1924;
(d) Zhdankin, V. V.; Stang, P. J. Chem. Rev. 2008, 108, 5299;
(e) Merritt, E. A.; Olofsson, B. Angew. Chem., Int. Ed. 2009, 48, 9052;
(f) Yusubov, M. S.; Maskaev, A. V.; Zhdankin, V. V. ARKIVOC 2011, 370;
(g) Kita, Y.; Dohi, T. Chem. Rec. 2015, 15, 886;
(h) Yoshimura, A.; Zhdankin, V. V. Chem. Rev. 2016, 116, 3328;
(i) Wirth, T. Topics in Current Chemistry, 2016, 373, 1-316;
(j) Fañanás-Mastral, M. Synthesis 2017, 49, 1905.
[7] Selected examples, see:
(a) Daugulis, O.; Zaitsev, V. G. Angew. Chem. Int. Ed. 2005, 44, 4046;
(b) Kalyani, D.; Deprez, N. R.; Desai, L. V.; Sanford, M. S. J. Am. Chem. Soc. 2005, 127, 7330;
(c) Deprez, N. R.; Kalyani, D.; Krause, A.; Sanford, M. S. J. Am. Chem. Soc. 2006, 128, 4972;
(d) Phipps, R. J.; Grimster, N. P.; Gaunt, M. J. J. Am. Chem. Soc. 2008, 130, 8172;
(e) Phipps, R. J.; Gaunt, M. J. Science 2009, 323, 1593;
(f) Deprez, N. R.; Sanford, M. S. J. Am. Chem. Soc. 2009, 131, 11234;
(g) Dohi, T.; Ito, M.; Yamaoka, N.; Morimoto, K.; Fujioka, H.; Kita, Y. Angew. Chem., Int. Ed. 2010, 49, 3334;
(h) Xiao, B.; Fu, Y.; Xu, J.; Gong, T.-J.; Dai, J.-J.; Yi, J.; Liu, L. J. Am. Chem. Soc. 2010, 132, 468;
(i) Ciana, C.; Phipps, R. J. Brandt, J. R.; Meyer, F.; Gaunt, M. J. Angew. Chem., Int. Ed. 2011, 50, 458;
(j) Storr, T. E.; Greaney, M. F. Org. Lett. 2013, 15, 1410.
[8] Selected examples, see:
(a) Kang, S.-K.; Yoon, S.-K.; Kim, Y.-M. Org. Lett. 2001, 3, 2697;
(b) Radhakrishnan, U.; Stang, P. J. Org. Lett. 2001, 3, 859;
(c) Xue, Z.; Yang, D.; Wang, C. J. Organomet. Chem. 2006, 691, 247;
(d) Collins, B. S. L.; Suero, M. G.; Gaunt, M. J. Angew. Chem., Int. Ed. 2013, 52, 5799;
(e) Suero, M. G.; Bayle, E. D.; Collins, B. S. L.; Gaunt, M. J. J. Am. Chem. Soc. 2013, 135, 5332;
(f) Chen, J.; Qu, H.; Peng, J.; Chen, C. Chin. J. Org. Chem. 2015, 35, 937. (陈静, 曲红梅, 彭静, 陈超, 有机化学, 2015, 35, 937.)
[9] Selected examples, see:
(a) Kina, A.; Miki, H.; Cho, Y.-H.; Hayashi, T. Adv. Synth. Catal. 2004, 346, 1728;
(b) Aydin, J.; Larsson, J. M.; Selander, N.; Szabo, K. J. Org. Lett. 2009, 11, 2852;
(c) Cahard, E.; Bremeyer, N.; Gaunt, M. J. Angew. Chem., Int. Ed. 2013, 52, 9284;
(d) Zhang, F.; Das, S.; Walkinshaw, A. J.; Casitas, A.; Taylor, M.; Suero, M. G.; Gaunt, M. J. J. Am. Chem. Soc. 2014, 136, 8851;
(e) Holt, D.; Gaunt, M. J. Angew. Chem., Int. Ed. 2015, 54, 7857;
(f) Yang, M.-N.; Yan, D.-M.; Zhao, Q.-Q.; Chen, J.-R.; Xiao, W.-J. Org. Lett. 2017, 19, 5208.
[10] Selected examples, see:
(a) Ryan, J. H.; Stang, P. J. Tetrahedron Lett. 1997, 38, 5061;
(b) Ochiai, M.; Kitagawa, Y.; Takayama, N.; Takaoka, Y.; Shiro, M. J. Am. Chem. Soc. 1999, 121, 9233;
(c) Aggarwal, V. K.; Olofsson, B. Angew. Chem., Int. Ed. 2005, 44, 5516;
(d) Allen, A. E.; MacMillan, D. W. C. J. Am. Chem. Soc. 2011, 133, 4260;
(e) Bigot, A.; Williamson, A. E.; Gaunt, M. J. J. Am. Chem. Soc. 2011, 133, 13778;
(f) Harvey, J. S.; Simonovich, S. P.; Jamison, C. R.; MacMillan, D. W. C. J. Am. Chem. Soc. 2011, 133, 13782.
[11] (a) Jalalian, N.; Ishikawa, E. E.; Silva Jr., L. F.; Olofsson, B. Org. Lett. 2011, 13, 1552;
(b) Petersen, T. B.; Khan, R.; Olofsson, B. Org. Lett. 2011, 13, 3462;
(c) Lindstedt, E.; Ghosh, R.; Olofsson, B. Org. Lett. 2013, 15, 6070;
(d) Chan, L.; McNally, A.; Toh, Q. Y.; Mendoza, A.; Gaunt, M. J. Chem. Sci. 2015, 6, 1277;
(e) Tolnai, G. L.; Nilsson, U. J.; Olofsson, B. Angew. Chem., Int. Ed. 2016, 55, 11226;
(f) Lindstedt, E.; Stridfeldt, E.; Olofsson, B. Org. Lett. 2016, 18, 4234;
(g) Tinnis, F.; Stridfeldt, E.; Lundberg, H.; Adolfsson, H.; Olofsson, B. Org. Lett. 2015, 17, 2688;
(h) Krief, A.; Dumont, W.; Robert, M. Chem. Commun. 2005, 2167;
(i) Wagner, A. M.; Sanford, M. S. J. Org. Chem. 2014, 79, 2263;
(j) Martin-Santamaria, S.; Carroll, M. A.; Carroll, C. M.; Carter, C. D.; Rzepa, H. S.; Widdowson, D. A.; Pike, V. W. Chem. Commun. 2000, 649;
(k) Song, A.; Zhang, C. Acta Chim. Sinica 2015, 73, 1002. (宋爱茹, 张弛, 化学学报, 2015, 73, 1002.);
(l) Duan, Y.; Jiang, S.; Han, Y.; Sun, B.; Zhang, C. Chin. J. Org. Chem. 2016, 36, 1973. (段亚南, 姜山, 韩永超, 孙博, 张弛, 有机化学, 2016, 36, 1973.)
[12] (a) Zhu, D.; Liu, Q.; Luo, B.; Chen, M.; Pi, R.; Huang, P.; Wen, S. Adv. Synth. Catal. 2013, 355, 2172;
(b) Zhu, D.; Wu, Y.; Wu, B.; Luo, B.; Ganesan, A.; Wu, F.-H.; Pi, R.; Huang, P.; Wen, S. Org. Lett. 2014, 16, 2350;
(c) Liu, Z.; Zhu, D.; Luo, B.; Zhang, N.; Liu, Q.; Hu, Y.; Pi, R.; Huang, P.; Wen, S. Org. Lett. 2014, 16, 5600;
(d) Modha, S. G.; Greaney, M. F. J. Am. Chem. Soc. 2015, 137, 1416;
(e) Wu, B.; Yoshikai, N. Angew. Chem., Int. Ed. 2015, 54, 8736;
(f) Luo, B.; Cui, Q.; Luo, H.; Hu, Y.; Huang, P.; Wen, S. Adv. Synth. Catal. 2016, 358, 2733;
(g) Shimizu, M.; Ogawa, M.; Tamagawa, T.; Shigitani, R.; Nakatani, M.; Nakano, Y. Eur. J. Org. Chem. 2016, 2785;
(h) Teskey, C. J.; Sohel, S. M. A.; Bunting, D. L.; Modha, S. G.; Greaney, M. F. Angew. Chem., Int. Ed. 2017, 56, 5263.
[13] (a) Wang, M.; Fan, Q.; Jiang, X. Org. Lett. 2016, 18, 5756;
(b) Wang, M.; Wei, J.; Fan, Q.; Jiang, X. Chem. Commun. 2017, 53, 2918;
(c) Wang, M.; Chen, S.; Jiang, X. Org. Lett. 2017, 19, 4916;
(d) Wang, M.; Fan, Q.; Jiang, X. Org. Lett. 2018, 20, 216.
[14] CCDC 1525038(2j) can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac. uk/data_request/cif.
[15] (a) Feng, M.; Tang, B.; Wang, N.; Xu, H.-X.; Jiang, X. Angew. Chem., Int. Ed. 2015, 54, 14960;
(b) Willcox, D.; Chappell, B. G. N.; Hogg, K. F.; Calleja, J.; Smalley, A. P.; Gaunt, M. J. Science 2016, 354, 851. |