化学学报 ›› 2023, Vol. 81 ›› Issue (6): 582-587.DOI: 10.6023/A23030075 上一篇 下一篇
研究通讯
坎比努尔•努尔买买提a, 王超a, 罗时玮a,b,*( ), 阿布都热西提•阿布力克木a,*(
), 阿布都热西提•阿布力克木a,*( )
)
投稿日期:2023-03-10
发布日期:2023-05-15
基金资助:
Kanbinuer Nuermaimaitia, Chao Wanga, Shiwei Luoa,b( ), Abudu Rexit Abulikemua(
), Abudu Rexit Abulikemua( )
)
Received:2023-03-10
Published:2023-05-15
Contact:
*E-mail: aarexit@xjnu.edu.cn; luosw@ustc.edu.cn
Supported by:文章分享

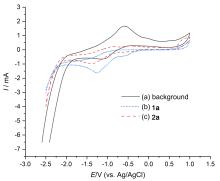
α,α-二卤(氯, 溴)甲基酮类化合物是关键的有机合成原料和最有价值的中间体之一. 本工作研究了电化学条件下α,α,α-三卤甲基酮类化合物的选择性脱卤反应. 室温下, 以铂片作为电极、1,2-二氯乙烷(或乙腈)为溶剂、n-Bu4NHSO4 (或四丁基氟化铵, TBAF, 0.5 equiv.)为电解质, 对α,α,α-三卤甲基酮类化合物进行电化学反应2~7.5 h, 得到相应α,α-二卤甲基酮, 分离产率高达92%. 此反应条件温和、底物普适性广、官能团兼容性好, 为制备α,α-二卤甲基酮提供了一种高效且经济的克级制备方法.
坎比努尔•努尔买买提, 王超, 罗时玮, 阿布都热西提•阿布力克木. 电化学条件下α,α,α-三卤(氯, 溴)甲基酮类化合物的选择性脱卤反应研究[J]. 化学学报, 2023, 81(6): 582-587.
Kanbinuer Nuermaimaiti, Chao Wang, Shiwei Luo, Abudu Rexit Abulikemu. Research on Selective Dehalogenation of α,α,α-Trihalogen (Chloro, Bromo) methyl Ketones Under Electrochemical Conditions[J]. Acta Chimica Sinica, 2023, 81(6): 582-587.
| Entry | Variations of standard conditions | Yieldb/% |
|---|---|---|
| 1 | None | 88 |
| 2 | CH3CN as solvent (14 h) | 80 |
| 3 | Dioxane/H2O (V/V, 9∶1) as solvent (8 h) | 74 |
| 4 | DMF as solvent (24 h) | 50 |
| 5 | EtOAc/H2O (V/V, 9∶1) as solvent (10 h) | 67 |
| 6 | THF as solvent (24 h) | 7 |
| 7 | CH3OH as solvent (24 h) | 0 |
| 8 | n-Bu4NClO4 instead of n-Bu4NHSO4 | 50 |
| 9 | n-Bu4NBF4 instead of n-Bu4NHSO4 | 39 |
| 10 | NH4Cl instead of n-Bu4NHSO4 | 30 |
| 11 | TBAI instead of n-Bu4NHSO4 | 25 |
| 12 | TBAB instead of n-Bu4NHSO4 | 17 |
| 13 | KI instead of n-Bu4NHSO4 | 16 |
| 14 | C(+)-Pt(−) | 37 |
| 15 | C(+)-C(−) | 36 |
| 16 | 5 mA (28 h) instead of 10 mA | 87 |
| 17 | 7 mA (4.5 h) instead of 10 mA | 74 |
| 18 | no n-Bu4NHSO4 | 0 |
| 19 | without electric current (24 h) | 0 |
| Entry | Variations of standard conditions | Yieldb/% |
|---|---|---|
| 1 | None | 88 |
| 2 | CH3CN as solvent (14 h) | 80 |
| 3 | Dioxane/H2O (V/V, 9∶1) as solvent (8 h) | 74 |
| 4 | DMF as solvent (24 h) | 50 |
| 5 | EtOAc/H2O (V/V, 9∶1) as solvent (10 h) | 67 |
| 6 | THF as solvent (24 h) | 7 |
| 7 | CH3OH as solvent (24 h) | 0 |
| 8 | n-Bu4NClO4 instead of n-Bu4NHSO4 | 50 |
| 9 | n-Bu4NBF4 instead of n-Bu4NHSO4 | 39 |
| 10 | NH4Cl instead of n-Bu4NHSO4 | 30 |
| 11 | TBAI instead of n-Bu4NHSO4 | 25 |
| 12 | TBAB instead of n-Bu4NHSO4 | 17 |
| 13 | KI instead of n-Bu4NHSO4 | 16 |
| 14 | C(+)-Pt(−) | 37 |
| 15 | C(+)-C(−) | 36 |
| 16 | 5 mA (28 h) instead of 10 mA | 87 |
| 17 | 7 mA (4.5 h) instead of 10 mA | 74 |
| 18 | no n-Bu4NHSO4 | 0 |
| 19 | without electric current (24 h) | 0 |
| Entry | Variations of standard conditions | Yieldb/% |
|---|---|---|
| 1 | None | 68 |
| 2 | DCE as solvent | 51 |
| 3 | Dioxane/H2O (V/V, 9∶1) as solvent | 36 |
| 4 | CH3CN as solvent | 60 |
| 5 | DMF as solvent | 55 |
| 6 | THF as solvent | 35 |
| 7 | EtOAc/H2O (V/V, 9∶1) as solvent | 23 |
| 8 | n-Bu4NClO4 instead of TBAF | 27 |
| 9 | n-Bu4NBF4 instead of TBAF | 30 |
| 10 | TBAI instead of TBAF | 44 |
| 11 | C(+)-Pt(−) | 59 |
| 12 | C(+)-C(−) | 55 |
| 13 | 5 mA instead of 10 mA | 54 |
| Entry | Variations of standard conditions | Yieldb/% |
|---|---|---|
| 1 | None | 68 |
| 2 | DCE as solvent | 51 |
| 3 | Dioxane/H2O (V/V, 9∶1) as solvent | 36 |
| 4 | CH3CN as solvent | 60 |
| 5 | DMF as solvent | 55 |
| 6 | THF as solvent | 35 |
| 7 | EtOAc/H2O (V/V, 9∶1) as solvent | 23 |
| 8 | n-Bu4NClO4 instead of TBAF | 27 |
| 9 | n-Bu4NBF4 instead of TBAF | 30 |
| 10 | TBAI instead of TBAF | 44 |
| 11 | C(+)-Pt(−) | 59 |
| 12 | C(+)-C(−) | 55 |
| 13 | 5 mA instead of 10 mA | 54 |

| [1] |
Zheng, Z. B.; Han, B. B.; Cheng, P. Tetrahedron 2014, 70, 9814.
doi: 10.1016/j.tet.2014.11.004 |
| [2] |
Tu, D. W.; Luo, J.; Jiang, W. G.; Tang, Q. Tetrahedron Lett. 2021, 81, 153335.
|
| [3] |
Madabhushi, S.; Jillella, R.; Mallu, K. K. R.; Godala, K. R.; Vangipuram, V. S. Tetrahedron Lett. 2013, 54, 3993.
doi: 10.1016/j.tetlet.2013.05.072 |
| [4] |
Chelucci, G. Chem. Rev. 2012, 112, 1344.
doi: 10.1021/cr200165q pmid: 22085400 |
| [5] |
Jiang, J.; Zou, H.; Dong, Q.; Wang, R.; Lu, L.; Zhu, Y.; He, W. J. Org. Chem. 2015, 81, 51.
doi: 10.1021/acs.joc.5b02093 |
| [6] |
Li, W.; Li, J.; DeVincentis, D.; Mansour, T. S. Tetrahedron Lett. 2004, 45, 1071.
doi: 10.1016/j.tetlet.2003.11.072 |
| [7] |
Syam, K. U.; Shanmugapriya, D.; Shankar, R.; Satyanarayana, G.; Dahanukar, V.; Vembu, N. Synlett 2008, 19, 2945.
|
| [8] |
(a) Finck, L.; Brals, J.; Pavuluri, B.; Gallou, F.; Handa, S. J. Org. Chem. 2018, 83, 7366.
doi: 10.1021/acs.joc.7b03143 |
|
(b) Wang, Y. B.; Chen, F.; Li, M.; Bu, Q. Q.; Du, Z. H.; Liu, J. C.; Dai, B.; Liu, N. Green Chem. 2023, 25, 1191.
doi: 10.1039/D2GC03989C |
|
| [9] |
(a) Li, Y. M.; Mou, T.; Lu, L. L.; Jiang, X. F. Chem. Commun. 2019, 55, 14299.
doi: 10.1039/C9CC07655G |
|
(b) Xiang, J. C.; Wang, J. W.; Yuan, P.; Ma, J. T.; Wu, A. X.; Liao, Z. X. J. Org. Chem. 2022, 87, 15101.
doi: 10.1021/acs.joc.2c01591 |
|
| [10] |
(a) Latham, J.; Brandenburger, E.; Shepherd, S. A.; Menon, B.; Micklefield, J. Chem. Rev. 2018, 118, 232.
doi: 10.1021/acs.chemrev.7b00032 |
|
(b) Touqeer, S.; Senatore, R.; Malik, M.; Urban, E.; Pace, V. Adv. Synth. Catal. 2020, 362, 5056.
doi: 10.1002/adsc.v362.22 |
|
| [11] |
(a) Saikia, I.; Borah, A. J.; Phukan, P. Chem. Rev. 2016, 116, 6837.
doi: 10.1021/acs.chemrev.5b00400 |
|
(b) Abulipizi, G.; Hasimujiang, B.; Rexit, A. A. Heterocycles 2021, 102, 318.
doi: 10.3987/COM-20-14360 |
|
| [12] |
(a) Arora, L.; Prakash, R.; Pundeer, R. Synth. Commun. 2019, 49, 1486.
doi: 10.1080/00397911.2019.1579915 |
|
(b) Zhang, Z. L.; Yang, J. S.; Wu, K. R.; Yu, R. J.; Bu, J. P.; Huang, Z. J.; Li, S. K.; Ma, X. T. Tetrahedron Lett. 2022, 88, 153575.
|
|
| [13] |
Diwu, Z.; Beachdel, C.; Klaubert, D. H. Tetrahedron Lett. 1998, 39, 4987.
doi: 10.1016/S0040-4039(98)00975-7 |
| [14] |
Liu, Y. Y.; Xiong, J.; Wei, L.; Wan, J. P. Adv. Synth. Catal. 2020, 362, 877.
doi: 10.1002/adsc.v362.4 |
| [15] |
Rather, S. A.; Kumar, A.; Ahmed, Q. N. Chem. Commun. 2019, 55, 4511.
doi: 10.1039/C9CC00346K |
| [16] |
Chen, Z.; Zhou, B.; Cai, H.; Zhu, W.; Zou, X. Green Chem. 2009, 11, 275.
doi: 10.1039/B815169E |
| [17] |
Conte, V.; Floris, B.; Galloni, P.; Silvagni, A. Adv. Synth. Catal. 2005, 347, 1341.
doi: 10.1002/(ISSN)1615-4169 |
| [18] |
Gallo, R. D. C.; Ahmad, A.; Metzker, G.; Burtoloso, A. C. B. Chem. Eur. J. 2017, 23, 16980.
doi: 10.1002/chem.201704609 |
| [19] |
Juneja, S. K.; Choudhary, D.; Paul, S.; Gupta, R. Synth. Commun. 2006, 36, 2877.
doi: 10.1080/00397910600770714 |
| [20] |
Paul, S.; Gupta, V.; Gupta, R.; Loupy, A. Tetrahedron Lett. 2003, 44, 439.
doi: 10.1016/S0040-4039(02)02601-1 |
| [21] |
Pandit, P.; Gayen, K. S.; Khamarui, S.; Chatterjee, N.; Maiti, D. K. Chem. Commun. 2011, 47, 6933.
doi: 10.1039/c1cc11685a |
| [22] |
Nobuta, T.; Hirashima, S.; Tada, N.; Miura, T.; Itoh, A. Tetrahedron Lett. 2010, 51, 4576.
doi: 10.1016/j.tetlet.2010.06.120 |
| [23] |
Wang, J. Y.; Jiang, Q.; Guo, C. C. Synth. Commun. 2014, 44, 3130.
doi: 10.1080/00397911.2014.928938 |
| [24] |
Yadav, L.; Chawla, R.; Singh, A. Synlett 2013, 24, 1558.
doi: 10.1055/s-00000083 |
| [25] |
Imanishi, T.; Fujiwara, Y.; Sawama, Y.; Monguchi, Y.; Sajiki, H. Adv. Synth. Catal. 2012, 354, 771.
doi: 10.1002/adsc.201100778 |
| [26] |
Rafael, D. H.; Ivann, Z. G.; Horacio, O. F.; Moise, R. O. ChemistrySelect 2017, 2, 10067.
doi: 10.1002/slct.201702003 |
| [27] |
Moises, A. R. R.; Ivann, Z. G.; Horacio, O. F.; Moises, R. O. J. Org. Chem. 2016, 81, 9515.
doi: 10.1021/acs.joc.6b02044 |
| [28] |
Wang, H.; Zheng, M. X.; Guo, H. M.; Huang, G. Z.; Cheng, Z.; Rexit, A. A. Eur. J. Org. Chem. 2020, 41, 6455.
|
| [29] |
Yang, Y.; Hasimujiang, B.; Rexit, A. A. Chin. J. Org. Chem. 2019, 39, 727. (in Chinese)
doi: 10.6023/cjoc201810003 |
|
(杨莹, 哈斯木江•巴拉提, 阿布都热西提•阿布力克木, 有机化学, 2019, 39, 727.)
|
|
| [30] |
Essa, A. H.; Lerrick, R. I.; Tuna, F.; Harrington, R. W.; Clegga, W.; Hall, M. J. Chem. Commun. 2013, 49, 2756.
doi: 10.1039/c3cc39147g |
| [31] |
(a) Liu, Y. J.; Wang, Z. C.; Meng, J. P.; Li, C.; Sun, K. Chin. J. Org. Chem. 2022, 42, 100. (in Chinese)
doi: 10.6023/cjoc202106051 |
|
(刘颖杰, 王智传, 孟建萍, 李晨, 孙凯, 有机化学, 2022, 42, 100.)
doi: 10.6023/cjoc202106051 |
|
|
(b) Yang, G.; Wang, Y. W.; Qiu, Y. A. Chin. J. Org. Chem. 2021, 41, 3935. (in Chinese)
doi: 10.6023/cjoc202105054 |
|
|
(杨光, 王衍伟, 仇友爱, 有机化学, 2021, 41, 3935.)
|
|
| [32] |
(a) Yuan, Y.; Lei, A. W. Acc. Chem. Res. 2019, 52, 3309.
doi: 10.1021/acs.accounts.9b00512 |
|
(b) Wang, Z. H.; Ma, C.; Fang, P.; Xu, H. C.; Mei, T. S. Acta Chim. Sinica 2022, 80, 1115. (in Chinese)
doi: 10.6023/A22060260 |
|
|
(王振华, 马聪, 方萍, 徐海超, 梅天胜, 化学学报, 2022, 80, 1115.)
doi: 10.6023/A22060260 |
|
| [33] |
Sun, K.; Lei, J.; Liu, Y.; Liu, B.; Chen, N. Adv. Synth. Catal. 2020, 362, 3709.
doi: 10.1002/adsc.v362.18 |
| [34] |
(a) Yang, Q. L.; Wang, X. Y.; Weng, X. J.; Yang, X.; Xu, X. T.; Tong, X. F.; Mei, T. S. Acta Chim. Sinica 2019, 77, 866. (in Chinese)
doi: 10.6023/A19040135 |
|
(杨启亮, 王向阳, 翁信军, 杨祥, 徐学涛, 童晓峰, 梅天胜, 化学学报, 2019, 77, 866 )
doi: 10.6023/A19040135 |
|
|
(b) Li, X. Y.; Tao, P. F.; Cheng, Y. Y.; Hu, Q.; Huang, W. J.; Li, Y.; Luo, Z. H.; Huang, G. B. Chin. J. Org. Chem. 2022, 42, 4169. (in Chinese)
doi: 10.6023/cjoc202204066 |
|
|
(李秀英, 陶萍芳, 程泳渝, 胡琼, 黄伟娟, 李芸, 罗志辉, 黄国保, 有机化学, 2022, 42, 4169.)
doi: 10.6023/cjoc202204066 |
|
| [35] |
Meng, X. T.; Zhang, Y.; Luo, J. Y.; Wang, F.; Cao, X. J.; Huang, S. L. Org. Lett. 2020, 22, 1169.
doi: 10.1021/acs.orglett.0c00052 |
| [36] |
Wang, D.; Wan, Z. H.; Zhang, H.; Lei, A. W. Adv. Synth. Catal. 2020, 363, 1022.
doi: 10.1002/adsc.v363.4 |
| [37] |
Box, J. R.; Atkins, A. P.; Lennox, A. J. J. Chem. Sci. 2021, 12, 10252.
doi: 10.1039/D1SC01574E |
| [38] |
Li, Z. B.; Sun, Q.; Qian, P.; Hu, K. F.; Zha, Z. G.; Wang, Z. Y. Chin Chem. Lett. 2020, 31, 1855.
doi: 10.1016/j.cclet.2020.02.030 |
| [39] |
Zhou, B.; He, Y. J.; Tao, Y. F.; Liu, L. X.; Hu, M.; Chang, Z. H.; Du, G. B. Green Chem. 2022, 24, 2859.
doi: 10.1039/D1GC04584A |
| [1] | 王振华, 马聪, 方萍, 徐海超, 梅天胜. 有机电化学合成的研究进展[J]. 化学学报, 2022, 80(8): 1115-1134. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||