Acta Chimica Sinica ›› 2024, Vol. 82 ›› Issue (3): 314-322.DOI: 10.6023/A23110484 Previous Articles Next Articles
Article
黄广峥, 李坤玮, 罗艳楠, 张强, 潘远龙, 高洪林*( )
)
投稿日期:2023-11-01
发布日期:2024-01-26
基金资助:
Guangzheng Huang, Kunwei Li, Yannan Luo, Qiang Zhang, Yuanlong Pan, Honglin Gao( )
)
Received:2023-11-01
Published:2024-01-26
Contact:
*E-mail: Supported by:Share
Guangzheng Huang, Kunwei Li, Yannan Luo, Qiang Zhang, Yuanlong Pan, Honglin Gao. Hydrothermal Treatment for Constructing K Doping and Surface Defects in g-C3N4 Nanosheets Promote Photocatalytic Hydrogen Production[J]. Acta Chimica Sinica, 2024, 82(3): 314-322.
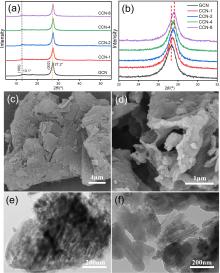







| [1] |
Dai, L.; Chang, D. W.; Baek, J.-B.; Lu, W. Small 2012, 8, 1122.
doi: 10.1002/smll.v8.8 |
| [2] |
An, P.; Zhang, Q.; Yang, Z.; Wu, J.; Zhang, J.; Wang, Y.; Li, Y.; Jiang, G. Acta Chim. Sinica 2022, 80, 1629 (in Chinese).
doi: 10.6023/A22080362 |
|
(安攀, 张庆慧, 杨状, 武佳星, 张佳颖, 王雅君, 李宇明, 姜桂元, 化学学报, 2022, 80, 1629.)
doi: 10.6023/A22080362 |
|
| [3] |
Fujishima, A.; Honda, K. J. n. Nature 1972, 238, 37.
doi: 10.1038/238037a0 |
| [4] |
Wolff, C. M.; Frischmann, P. D.; Schulze, M.; Bohn, B. J.; Wein, R.; Livadas, P.; Carlson, M. T.; Jäckel, F.; Feldmann, J.; Würthner, F.; Stolarczyk, J. K. Nat. Energy 2018, 3, 862.
doi: 10.1038/s41560-018-0229-6 |
| [5] |
Lee, B.-H.; Gong, E.; Kim, M.; Park, S.; Kim, H. R.; Lee, J.; Jung, E.; Lee, C. W.; Bok, J.; Jung, Y.; Kim, Y. S.; Lee, K.-S.; Cho, S.-P.; Jung, J.-W.; Cho, C.-H.; Lebègue, S.; Nam, K. T.; Kim, H.; In, S.-I.; Hyeon, T. Energ. Environ. Sci. 2022, 15, 601.
doi: 10.1039/D1EE01574E |
| [6] |
Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J. M.; Domen, K.; Antonietti, M. Nat. Mater. 2009, 8, 76.
doi: 10.1038/nmat2317 |
| [7] |
Cao, S.; Low, J.; Yu, J.; Jaroniec, M. Adv. Mater. 2015, 27, 2150.
doi: 10.1002/adma.v27.13 |
| [8] |
Xie, Z.; Xue, Z.; Xu, Z.; Li, Q.; Wang, H.; Li, W. Acta Chim. Sinica 2022, 80 1231 (in Chinese).
doi: 10.6023/A22040183 |
|
(解众舒, 薛中鑫, 许子文, 李倩, 王洪宇, 李维实, 化学学报, 2022, 80 1231.)
doi: 10.6023/A22040183 |
|
| [9] |
Li, K.; Bao, L.; Cao, S.; Xue, Y.; Yan, S.; Gao, H. ACS Appl. Energy Mater. 2021, 4, 12965.
doi: 10.1021/acsaem.1c02602 |
| [10] |
Wang, S.; Zhao, H.; Zhao, X.; Zhang, J.; Ao, Z.; Dong, P.; He, F.; Wu, H.; Xu, X.; Shi, L.; Zhao, C.; Wang, S.; Sun, H. Chem. Eng. J. 2020, 381, 122593.
doi: 10.1016/j.cej.2019.122593 |
| [11] |
Jang, D.; Lee, S.; Kwon, N. H.; Kim, T.; Park, S.; Jang, K. Y.; Yoon, E.; Choi, S.; Han, J.; Lee, T.-W.; Kim, J.; Hwang, S.-J.; Park, S. Carbon 2023, 208, 290.
doi: 10.1016/j.carbon.2023.03.038 |
| [12] |
Chang, B.; Guo, Y.; Liu, H.; Li, L.; Yang, B. J. Mater. Chem. A 2022, 10, 3134.
doi: 10.1039/D1TA09941H |
| [13] |
Huang, Y.; Mei, F.; Zhang, J.; Dai, K.; Dawson, G. Acta Phys.-Chim. Sin. 2022, 38, 13 (in Chinese).
|
|
(黄悦, 梅飞飞, 张金锋, 代凯, Dawson, G., 物理化学学报, 2022, 38, 13.)
|
|
| [14] |
Wang, X.; Meng, J.; Zhang, X.; Liu, Y.; Ren, M.; Yang, Y.; Guo, Y. Adv. Funct. Mater. 2021, 31, 2010763.
doi: 10.1002/adfm.v31.20 |
| [15] |
Zhao, Z.; Long, Y.; Chen, Y.; Zhang, F.; Ma, J. Chem. Eng. J. 2022, 430, 132682.
doi: 10.1016/j.cej.2021.132682 |
| [16] |
Adekoya, D.; Zhang, S.; Hankel, M. Carbon 2021, 176, 480.
doi: 10.1016/j.carbon.2021.02.050 |
| [17] |
Xie, H.; Li, Z.; Zhu, J.; Li, H.; Yang, Q.; Yang, Y.; Li, C. J. Phys. Chem. Lett. 2022, 13, 11982.
doi: 10.1021/acs.jpclett.2c03250 |
| [18] |
Li, J.; Banis, M. N.; Ren, Z.; Adair, K. R.; Doyle-Davis, K.; Meira, D. M.; Finfrock, Y. Z.; Zhang, L.; Kong, F.; Sham, T.-K.; Li, R.; Luo, J.; Sun, X. Small 2021, 17, 2007245.
doi: 10.1002/smll.v17.11 |
| [19] |
Zhai, B.; Li, H.; Gao, G.; Wang, Y.; Niu, P.; Wang, S.; Li, L. Adv. Funct. Mater. 2022, 32, 2207375.
doi: 10.1002/adfm.v32.47 |
| [20] |
Long, H.; Mo, C.; Mo, Y.; Qiu, C.; Jiang, M. Jiangsu University (Nat. Sci. Ed.) 2020, 41, 712.
|
| [21] |
Lin, B.; Yang, G.; Wang, L. Angew. Chem. Int. Ed. 2019, 58, 4587.
doi: 10.1002/anie.201814360 pmid: 30734453 |
| [22] |
Han, Q.; Wang, B.; Gao, J.; Cheng, Z.; Zhao, Y.; Zhang, Z.; Qu, L. ACS Nano 2016, 10, 2745.
doi: 10.1021/acsnano.5b07831 |
| [23] |
Lu, Y.; Shang, G.; Zhang, H.; Wang, Y.; Tan, Y.; Sun, J.; Liu, G. Chinese J. Inorg. Chem. 2021, 37, 668 (in Chinese).
|
|
(陆杨, 上官莉, 张慧, 王岩, 谭宇烨, 孙建华, 刘光祥, 无机化学学报, 2021, 37, 668.)
|
|
| [24] |
Niu, P.; Zhang, L.; Liu, G.; Cheng, H.-M. Adv. Funct. Mater. 2012, 22, 4763.
doi: 10.1002/adfm.v22.22 |
| [25] |
Wu, P.; Wang, J.; Zhao, J.; Guo, L.; Osterloh, F. E. J. Mater. Chem. A 2014, 2, 20338.
doi: 10.1039/C4TA04100C |
| [26] |
Zhang, X.; Xie, X.; Wang, H.; Zhang, J.; Pan, B.; Xie, Y. J. Am. Chem. Soc. 2013, 135, 18.
doi: 10.1021/ja308249k |
| [27] |
Dong, C.; Ma, Z.; Qie, R.; Guo, X.; Li, C.; Wang, R.; Shi, Y.; Dai, B.; Jia, X. Appl. Catal. B: Environ. 2017, 217, 629.
doi: 10.1016/j.apcatb.2017.06.028 |
| [28] |
Ruan, X.; Cui, X.; Jia, G.; Wu, J.; Zhao, J.; Singh, D. J.; Liu, Y.; Zhang, H.; Zhang, L.; Zheng, W. Chem. Eng. J. 2022, 428, 132579.
doi: 10.1016/j.cej.2021.132579 |
| [29] |
Zhao, D.; Wang, Y.; Dong, C.-L.; Huang, Y.-C.; Chen, J.; Xue, F.; Shen, S.; Guo, L. Nat. Energy 2021, 6, 388.
doi: 10.1038/s41560-021-00795-9 |
| [30] |
Xu, Y.; He, X.; Zhong, H.; Singh, D. J.; Zhang, L.; Wang, R. Appl. Catal. B: Environ. 2019, 246, 349.
doi: 10.1016/j.apcatb.2019.01.069 |
| [31] |
Murugesan, P.; Moses, J. A.; Anandharamakrishnan, C. J. Mater. Sci. 2019, 54, 12206.
doi: 10.1007/s10853-019-03695-2 |
| [32] |
Wang, C.; Fan, H.; Ren, X.; Ma, J.; Fang, J.; Wang, W. ChemSusChem 2018, 11, 700.
doi: 10.1002/cssc.v11.4 |
| [33] |
Ismael, M. J. Alloy. Compd. 2020, 846, 156446.
doi: 10.1016/j.jallcom.2020.156446 |
| [34] |
Vinu, A.; Ariga, K.; Mori, T.; Nakanishi, T.; Hishita, S.; Golberg, D.; Bando, Y. Adv. Mater. 2005, 17, 1648.
doi: 10.1002/adma.v17:13 |
| [35] |
Lau, V. W.-h.; Yu, V. W.-z.; Ehrat, F.; Botari, T.; Moudrakovski, I.; Simon, T.; Duppel, V.; Medina, E.; Stolarczyk, J. K.; Feldmann, J.; Blum, V.; Lotsch, B. V. Adv. Energy Mater. 2017, 7, 1602251.
doi: 10.1002/aenm.v7.12 |
| [36] |
Wu, S.; Yu, H.; Chen, S.; Quan, X. ACS Catal. 2020, 10, 14380.
doi: 10.1021/acscatal.0c03359 |
| [37] |
Zhang, J.; Chen, X.; Takanabe, K.; Maeda, K.; Domen, K.; Epping, J. D.; Fu, X.; Antonietti, M.; Wang, X. Angew. Chem. Int. Ed. 2010, 49, 441.
doi: 10.1002/anie.v49:2 |
| [38] |
Zhou, Z.; Shen, Y.; Li, Y.; Liu, A.; Liu, S.; Zhang, Y. ACS Nano 2015, 9, 12480.
doi: 10.1021/acsnano.5b05924 |
| [39] |
Yu, Y.; Yan, W.; Wang, X.; Li, P.; Gao, W.; Zou, H.; Wu, S.; Ding, K. Adv. Mater. 2018, 30, 1705060.
doi: 10.1002/adma.v30.9 |
| [40] |
Zhang, M.; Bai, X.; Liu, D.; Wang, J.; Zhu, Y. Appl. Catal. B: Environ. 2015, 164, 77.
doi: 10.1016/j.apcatb.2014.09.020 |
| [41] |
Wang, K.; Fu, J.; Zheng, Y. Appl. Catal. B: Environ. 2019, 254, 270.
doi: 10.1016/j.apcatb.2019.05.002 |
| [42] |
Zhang, G.; Li, G.; Lan, Z.-A.; Lin, L.; Savateev, A.; Heil, T.; Zafeiratos, S.; Wang, X.; Antonietti, M. Angew. Chem. Int. Ed. 2017, 56, 13445.
doi: 10.1002/anie.v56.43 |
| [43] |
Zeng, Z.; Yu, H.; Quan, X.; Chen, S.; Zhang, S. Appl. Catal. B: Environ. 2018, 227, 153.
doi: 10.1016/j.apcatb.2018.01.023 |
| [44] |
Jin, Z.; Jiang, X.; Zhang, Q.; Huang, S.; Zhang, L.; Huang, L.; He, T.; Zhang, H.; Ohno, T.; Ruan, S.; Zeng, Y.-J. Commun. Mater. 2020, 1, 90.
doi: 10.1038/s43246-020-00093-z |
| [45] |
Li, Y.; Xue, Y.; Gao, X.; Wang, L.; Liu, X.; Wang, Z.; Shen, S. Adv. Funct. Mater. 2023, n/a, 2312634.
|
| [46] |
Liu, C.; Zhang, Y.; Dong, F.; Reshak, A. H.; Ye, L.; Pinna, N.; Zeng, C.; Zhang, T.; Huang, H. Appl. Catal. B: Environ. 2017, 203, 465.
doi: 10.1016/j.apcatb.2016.10.002 |
| [47] |
Guo, Y.; Bae, J.; Fang, Z.; Li, P.; Zhao, F.; Yu, G. Chem. Rev. 2020, 120, 7642.
doi: 10.1021/acs.chemrev.0c00345 |
| [1] | Jianqiang Chen, Gangguo Zhu, Jie Wu. Recent Advances in Nickel-Catalyzed Ring Opening Cross-Coupling of Aziridines [J]. Acta Chimica Sinica, 2024, 82(2): 190-212. |
| [2] | Yuhan Wu, Dongdong Zhang, Hongyu Yin, Zhengnan Chen, Wen Zhao, Yuhua Chi. Density Functional Theory Study of Janus In2S2X Photocatalytic Reduction of CO2 under “Double Carbon” Target [J]. Acta Chimica Sinica, 2023, 81(9): 1148-1156. |
| [3] | Jiawen Liu, Weihuang Lin, Weijia Wang, Xueyi Guo, Ying Yang. Synthesis and Photocatalytic Degradation of Cu1.94S-SnS Nano-heterojunction [J]. Acta Chimica Sinica, 2023, 81(7): 725-734. |
| [4] | Minghui He, Ziqiu Ye, Guiqing Lin, Sheng Yin, Xinyi Huang, Xu Zhou, Ying Yin, Bo Gui, Cheng Wang. Research Progress of Porphyrin-Based Covalent Organic Frameworks in Photocatalysis★ [J]. Acta Chimica Sinica, 2023, 81(7): 784-792. |
| [5] | Li Liu, Gang Zheng, Guoqiang Fan, Hongguang Du, Jiajing Tan. Research Progress in Organic Reactions Involving 4-Acyl/Carbamoyl/Alkoxycarbonyl Substituted Hantzsch Esters [J]. Acta Chimica Sinica, 2023, 81(6): 657-668. |
| [6] | Fei Li, Huili Ding, Chaozhong Li. Hydrotrifluoromethylation of Alkenes with a Fluoroform-Derived Trifluoromethylboron Complex [J]. Acta Chimica Sinica, 2023, 81(6): 577-581. |
| [7] | Qi Xueping, Wang Fei, Zhang Jian. A Post-Synthetic Method for the Construction of Titanium-Based Metal Organic Frameworks and Their Applications [J]. Acta Chimica Sinica, 2023, 81(5): 548-558. |
| [8] | Jianqiang Chen, Gangguo Zhu, Jie Wu. Recent Advances in Radical-Based Dehydroxylation of Hydroxyl Groups via Oxalates [J]. Acta Chimica Sinica, 2023, 81(11): 1609-1623. |
| [9] | Chunhui Yang, Jingchao Chen, Xinhan Li, Li Meng, Kaimin Wang, Weiqing Sun, Baomin Fan. Difluoroallylation of Silanes under Photoirradiation [J]. Acta Chimica Sinica, 2023, 81(1): 1-5. |
| [10] | Zhongshu Xie, Zhongxin Xue, Ziwen Xu, Qian Li, Hongyu Wang, Wei-Shi Li. Conjugated Crosslinking Modification of Graphitic Carbon Nitrides and Its Effect on Visible Light-Driven Photocatalytic Hydrogen Production [J]. Acta Chimica Sinica, 2022, 80(9): 1231-1237. |
| [11] | Yu Qi, Fuxiang Zhang. Photocatalytic Water Splitting for Hydrogen Production※ [J]. Acta Chimica Sinica, 2022, 80(6): 827-838. |
| [12] | Heng Shu, Yide-Rigen Bao, Yong Na. Photocatalytic Oxidation of 5-Hydroxymethylfurfural Selectively into 2,5-Diformylfuran with CdS Nanotube [J]. Acta Chimica Sinica, 2022, 80(5): 607-613. |
| [13] | Xue Gong, Xinguo Ma, Fengda Wan, Wangyang Duan, Xiaoling Yang, Jinrong Zhu. Study on the Electronic Structure and Optical Properties of Two-dimensional Monolayer MoSi2X4 (X=N, P, As) [J]. Acta Chimica Sinica, 2022, 80(4): 510-516. |
| [14] | Pan An, Qinghui Zhang, Zhuang Yang, Jiaxing Wu, Jiaying Zhang, Yajun Wang, Yuming Li, Guiyuan Jiang. Research Progress of Solar Hydrogen Production Technology under Double Carbon Target [J]. Acta Chimica Sinica, 2022, 80(12): 1629-1642. |
| [15] | Xiaohan Yu, Wei Huang, Yanguang Li. Controllable Synthesis and Photocatalytic Applications of Two-dimensional Covalent Organic Frameworks [J]. Acta Chimica Sinica, 2022, 80(11): 1494-1506. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||