Acta Chimica Sinica ›› 2026, Vol. 84 ›› Issue (3): 299-304.DOI: 10.6023/A25120430 Previous Articles Next Articles
Article
王怡可†, 陈丽萍†, 舒敬利†, 朱雪华*( ), 汪洋*(
), 汪洋*( )
)
投稿日期:2025-12-30
发布日期:2026-02-10
基金资助:
Wang Yike, Chen Liping, Shu Jingli, Zhu Xuehua*( ), Wang Yang*(
), Wang Yang*( )
)
Received:2025-12-30
Published:2026-02-10
Contact:
*E-mail: zhuxuehua@usts.edu.cn,
ywang@usts.edu.cn
About author:† These authors contributed equally to this work.
Supported by:Share
Wang Yike, Chen Liping, Shu Jingli, Zhu Xuehua, Wang Yang. Efficient Anti-Markovnikov Hydrothiolation of Alkenes via Phase-Transfer Catalysis in Aqueous Phase[J]. Acta Chimica Sinica, 2026, 84(3): 299-304.
| Entry | Variation from the standard conditions | Yieldb/% |
|---|---|---|
| 1 | none | 85 |
| 2 | nBu4NBF4 instead of nBu4NOAc | 65 |
| 3 | nBu4NPF6 instead of nBu4NOAc | 25 |
| 4 | TEBAC instead of nBu4NOAc | 55 |
| 5 | nBu4NClO4 instead of nBu4NOAc | n.r. |
| 6 | nBu4NI instead of nBu4NOAc | trace |
| 7 | nBu4NBr instead of nBu4NOAc | trace |
| 8 | nBu4NCl instead of nBu4NOAc | trace |
| 9 | 18-Crown-6 instead of nBu4NOAc | n.r. |
| 10 | KOAc instead of nBu4NOAc | 31 |
| 11 | K2CO3 instead of nBu4NOAc | n.r. |
| 12 | Na2CO3 instead of nBu4NOAc | n.r. |
| 13 | No PTC | n.r. |
| 14 | 40 ℃ | 83 |
| 15 | N2 | 85 |
| 16 | Neat | n.r. |
| Entry | Variation from the standard conditions | Yieldb/% |
|---|---|---|
| 1 | none | 85 |
| 2 | nBu4NBF4 instead of nBu4NOAc | 65 |
| 3 | nBu4NPF6 instead of nBu4NOAc | 25 |
| 4 | TEBAC instead of nBu4NOAc | 55 |
| 5 | nBu4NClO4 instead of nBu4NOAc | n.r. |
| 6 | nBu4NI instead of nBu4NOAc | trace |
| 7 | nBu4NBr instead of nBu4NOAc | trace |
| 8 | nBu4NCl instead of nBu4NOAc | trace |
| 9 | 18-Crown-6 instead of nBu4NOAc | n.r. |
| 10 | KOAc instead of nBu4NOAc | 31 |
| 11 | K2CO3 instead of nBu4NOAc | n.r. |
| 12 | Na2CO3 instead of nBu4NOAc | n.r. |
| 13 | No PTC | n.r. |
| 14 | 40 ℃ | 83 |
| 15 | N2 | 85 |
| 16 | Neat | n.r. |
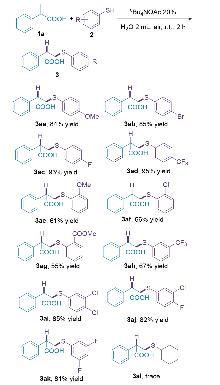
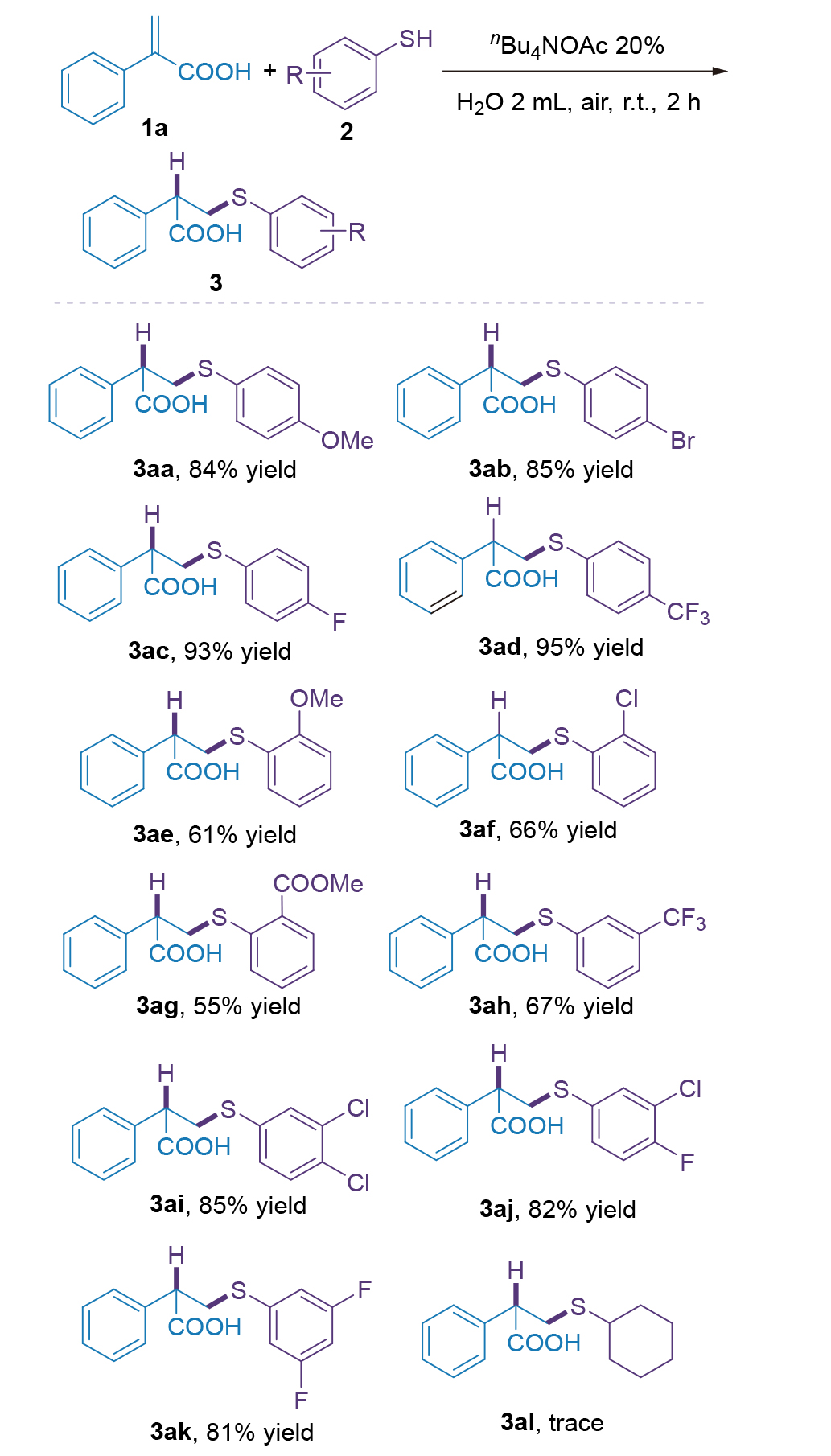
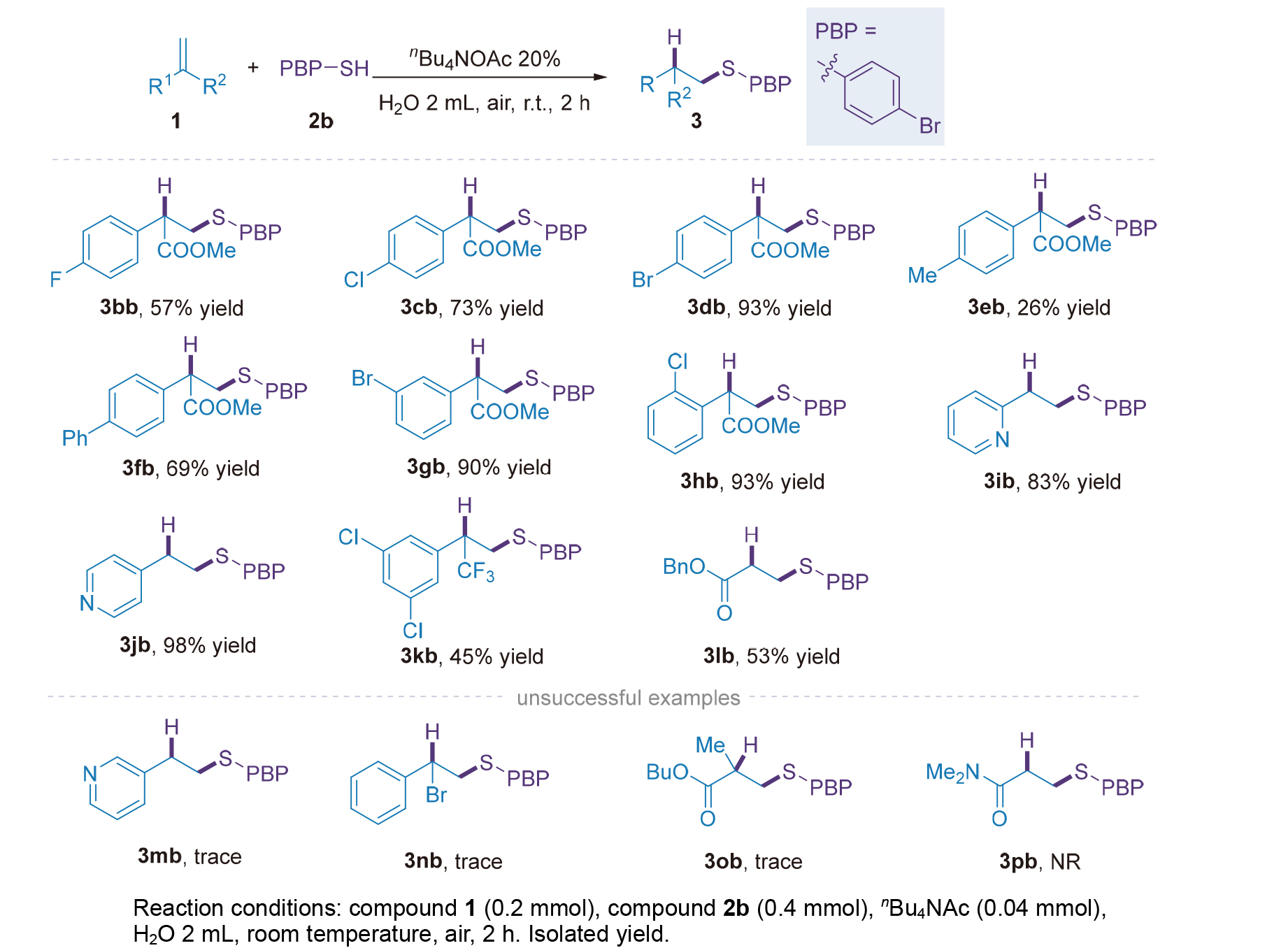
| [1] |
doi: 10.1039/C8NP00093J |
| [2] |
doi: 10.1021/acs.chemrev.6b00697 pmid: 28418240 |
| [3] |
doi: 10.1039/b609523m |
| [4] |
doi: 10.2174/1568026615666150915111741 |
| [5] |
|
| [6] |
doi: 10.1016/j.ejmech.2018.11.017 |
| [7] |
|
| [8] |
doi: 10.1002/advs.v9.4 |
| [9] |
doi: 10.1016/j.mattod.2023.02.027 |
| [40] |
doi: 10.1039/D1GC00098E |
| [41] |
doi: 10.1038/s41467-018-03698-8 |
| [42] |
doi: 10.1021/jacs.0c11815 |
| [10] |
doi: 10.1039/D1DT04371D |
| [11] |
doi: 10.1038/s41570-023-00505-x |
| [12] |
doi: 10.1039/C8GC01337C |
| [13] |
doi: 10.1021/acs.orglett.1c02738 |
| [14] |
|
| [15] |
doi: 10.1021/acscatal.8b04633 |
| [16] |
doi: 10.1016/j.tet.2022.132711 |
| [17] |
doi: 10.1039/d1ob01091c pmid: 34333579 |
| [18] |
doi: 10.1021/acssuschemeng.1c07636 |
| [19] |
doi: 10.1002/ajoc.201800639 |
| [20] |
|
| [21] |
doi: 10.1021/jo3020825 |
| [22] |
|
| [23] |
doi: 10.1038/s41467-024-52249-x |
| [24] |
doi: 10.1002/anie.v54.3 |
| [25] |
doi: 10.1021/acs.joc.3c02722 pmid: 38564378 |
| [26] |
doi: 10.1016/S0040-4020(03)00289-8 |
| [27] |
doi: 10.1002/ajoc.v7.9 |
| [28] |
doi: 10.1021/acsomega.9b01968 pmid: 31552354 |
| [29] |
doi: 10.1002/adsc.v365.24 |
| [30] |
doi: 10.1021/ja202292q pmid: 21728292 |
| [31] |
doi: 10.1002/asia.v15.6 |
| [32] |
doi: 10.1002/cptc.v2.10 |
| [33] |
|
| [34] |
doi: 10.6023/cjoc202207032 |
|
(王维, 张哲宇, 张雪, 于海丰, 罗辉, 霍东月, 徐玉澎, 赵晓波, 有机化学, 2023, 43, 742.)
|
|
| [35] |
doi: 10.6023/cjoc201904002 |
|
(胡小宇, 于海丰, 王文举, 姜思傲, 刘奇, 何洁, 有机化学, 2019, 39, 3183.)
doi: 10.6023/cjoc201904002 |
|
| [36] |
doi: 10.6023/cjoc201709021 |
|
(王伟, 吕梦娇, 赵利霞, 张娅玲, 李本浩, 李宝林, 有机化学, 2018, 38, 883.)
|
|
| [37] |
doi: 10.6023/A24110332 |
|
(杨雪, 刘妍伶, 陈霞, 周晓玉, 王爱玲, 刘海龙, 化学学报, 2025, 83, 354.)
doi: 10.6023/A24110332 |
|
| [38] |
doi: 10.1039/C8SC04271C |
| [39] |
doi: 10.1039/D4SC02681K |
| [1] | Zhao Xinyu, Han Yannan, Xu Jilei, An Qingda, Xiao Zuoyi, Su Xin, Huang Jiahui. Preparation of Bio-based Polyacid Esters Catalyzed by Supported Ionic Liquids [J]. Acta Chimica Sinica, 2026, 84(3): 341-352. |
| [2] | Yuhan Liu, Pan Gao. Direct Borylation of Organohalides Using Mechanochemically Generated Calcium-Based Heavy Grignard Reagents (R-CaX) [J]. Acta Chimica Sinica, 2024, 82(11): 1114-1119. |
| [3] | Li Liu, Gang Zheng, Guoqiang Fan, Hongguang Du, Jiajing Tan. Research Progress in Organic Reactions Involving 4-Acyl/Carbamoyl/Alkoxycarbonyl Substituted Hantzsch Esters [J]. Acta Chimica Sinica, 2023, 81(6): 657-668. |
| [4] | Qiaowei Tang, Xiaoqing Cai, Dapeng Yin, Huating Kong, Xiangzhi Zhang, Jichao Zhang, Qinglong Yan, Ying Zhu, Chunhai Fan. Click Chemistry-based Synchrotron X-ray Imaging Tags★ [J]. Acta Chimica Sinica, 2023, 81(5): 441-444. |
| [5] | Chang Li, Zhendong Zheng, Jiangnan Zheng, Ruijun Tian. Glycoprotein Identification using Cleavable Bifunctional Probes★ [J]. Acta Chimica Sinica, 2023, 81(12): 1673-1680. |
| [6] | Jingpeng Li, Qi Yang, Zhou Zhang, Guiyun Zeng, Teng Liu, Chao Huang. Highly Selective Synthesis of (Z)-N-vinyl Ring N,O-Acetal Derivatives by Multi-component Continuous Flow [J]. Acta Chimica Sinica, 2022, 80(11): 1463-1468. |
| [7] | Wei Zheyu, Chang Yalin, Yu Han, Han Sheng, Wei Yongge. Application of Anderson Type Heteropoly Acids as Catalysts in Organic Synthesis [J]. Acta Chimica Sinica, 2020, 78(8): 725-732. |
| [8] | Dong Kui, Liu Qiang, Wu Li-Zhu. Cross-Coupling Hydrogen Evolution Reactions [J]. Acta Chimica Sinica, 2020, 78(4): 299-310. |
| [9] | Ye Wenbo, Yan Zicong, Wan Changfeng, Hou Haoqing, Wang Zhiyong. A New Decarboxylation/Methylation Process of Cinnamic Acids [J]. Acta Chim. Sinica, 2018, 76(2): 99-102. |
| [10] | Pei Pengkun, Zhang Fan, Yi Hong, Lei Aiwen. Visible Light Promoted Benzylic Csp3-H Bond Activation and Functionalization [J]. Acta Chim. Sinica, 2017, 75(1): 15-21. |
| [11] | Lu Qingquan, Yi Hong, Lei Aiwen. Autoxidative Coupling and Its Applications to C-H Functionalization [J]. Acta Chim. Sinica, 2015, 73(12): 1245-1249. |
| [12] | Zhang Lifang, Zhao Jie, Wang Yong. Construction and Chromatographic Performance of a Novel Triazole Bridged Hybrid Bilayer Cyclodextrin Chiral Stationary Phase [J]. Acta Chim. Sinica, 2015, 73(11): 1182-1188. |
| [13] | Lai Changwei, Sun Ying, Yang Hong, Zhang Xueqin, Lin Baoping . The Functionalization of Graphene and Graphene Oxide via Click Chemistry [J]. Acta Chimica Sinica, 2013, 71(9): 1201-1224. |
| [14] | Wang Huaisong, Peng Jiangtao, Wei Jiping, Jiang An. Synthesis of Novel Chiral Stationary Phase Based on Atom Transfer Radical Polymerization and Click Chemistry [J]. Acta Chimica Sinica, 2012, 70(12): 1355-1361. |
| [15] | LI Ying-Jun*,1; ZHANG Zhi-Guang; JIN Kun2; PENG Qin-Ji2; DING Wan-Gang; LIU Jun. Synthesis, Characterization and Biological Activities of Acylthiourea Derivatives [J]. Acta Chimica Sinica, 2007, 65(9): 834-840. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||