

含氮芥基查尔酮类衍生物的合成、晶体结构及抗肿瘤活性
收稿日期: 2012-11-21
修回日期: 2012-12-17
网络出版日期: 2012-12-20
基金资助
国家自然科学基金(No. 21172178)资助项目.
Synthesis, Crystal Strutures and Antitumor Activity of Novel Nitrogen Mustard-Linked Chalcones
Received date: 2012-11-21
Revised date: 2012-12-17
Online published: 2012-12-20
Supported by
Project supported by the National Natural Science Foundation of China (No. 21172178).
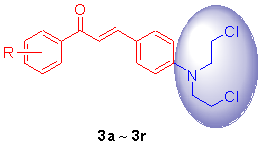
根据拼药原理, 将氮芥类抗肿瘤药物的药效基团N,N-二(2-氯乙基)氨基拼接到查尔酮结构中, 合成出了一系列新型含氮芥基的查尔酮衍生物3a~3r, 并用1H NMR, IR, HRMS-ESI及X射线对其结构进行了确证. 采用MTT法对所合成的化合物进行了体外抗肿瘤活性测试, 结果表明部分化合物对所选肿瘤细胞的增殖有较强的抑制作用, 其中, 化合物3j和3l对K562的IC50值分别为20.01和18.6 μmol/L; 对HepG2的IC50值分别为43.46和21.39 μmol/L. 实验培养并得到了目标产物3e和3l的晶体结构, X射线分析表明化合物3e和3l都属三斜晶系, P-1空间群.
方线文 , 周丽珍 , 成昭 , 杨美盼 , 杨秉勤 . 含氮芥基查尔酮类衍生物的合成、晶体结构及抗肿瘤活性[J]. 有机化学, 2013 , 33(03) : 523 -529 . DOI: 10.6023/cjoc201211041
A series of novel nitrogen mustard-linked chalcones 3a~3r were synthesized by linking the N,N-bis(2- chloroethyl)amine pharmacophore of nitrogen mustards to the chalcone’s framework. Their structures were characterized by 1H NMR, IR, ESI-HRMS and X-ray techniques. The antitumor activities of the nitrogen mustard-linked chalcones were evaluated against K562 and HepG2 cell lines by an MTT assay. The results reveal that some of the title compounds exhibit potent anti-proliferative activities against selected tumor cells. Among which, compounds 3j against K562 and HepG2 with IC50 values of 20.01 and 43.46 μmol/L, and 3l against K562 and HepG2 with IC50 values of 18.6 and 21.39 μmol/L, respectively. Suitable crystals of 3e and 3l were obtained, and the X-ray diffraction analysis indicates that compounds 3b and 3e are both belong to triclinic, space group P-1.

Key words: chalcones; nitrogen mustard; synthesis; crystal structure; anti-tumor activity
/
| 〈 |
|
〉 |