

O,O'-二烷基-α-苯基-α-(取代苯甲酰氧基)-甲基膦酸酯的合成与抗肿瘤活性
收稿日期: 2012-11-20
修回日期: 2012-12-20
网络出版日期: 2013-01-07
基金资助
国家自然科学基金(No. 81160385)和贵州省科技厅基金(No. 黔科合J字LKZ[2010]47)资助项目.
Synthesis and Antitumor Activity of O,O'-Dialkyl-α-phenyl-α-(substituted benzoylanoxy)-methylphosphonate
Received date: 2012-11-20
Revised date: 2012-12-20
Online published: 2013-01-07
Supported by
Project supported by the National Natural Science Foundation of China (No. 81160385) and the Science and Technology Department of Guizhou Province (No. LKZ[2010]47).
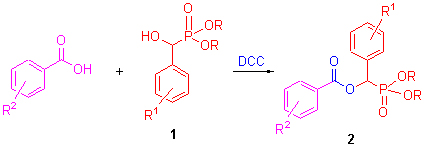
为了寻找新型抗肿瘤先导物, 采用缩合剂N,N'-二环己基碳二亚胺(DCC)合成酯的方法, 以O,O'-二烷基-1-羟 基-(取代苯基)甲基膦酸酯、取代苯甲酸为原料合成16个目标物, 其中12个化合物未见报道. 经IR, 1H NMR, 13C NMR及元素分析对所合成的化合物进行了结构确认和表征. 并首次研究了该系列化合物的抗肿瘤活性. 结果表明: 目标物具有一定的抑制肿瘤细胞增殖作用, 其中2i, 2k, 2m对SGC-7901的IC50值分别为18.8, 18.5, 24.5 μmol/L, 对A-549的IC50值分别为24.1, 29.0, 20.7 μmol/L, 显示出潜在的抗肿瘤活性, 值得进一步研究. 在此基础上, 初步研究其构效关系.
杨家强 , 谷晴 , 束波 , 宋宝安 , 刘静姿 . O,O'-二烷基-α-苯基-α-(取代苯甲酰氧基)-甲基膦酸酯的合成与抗肿瘤活性[J]. 有机化学, 2013 , 33(05) : 1113 -1118 . DOI: 10.6023/cjoc201211038
In search of more effective anticancer agents, the title compounds were prepared via the esterification reaction of α-hydroxyphosphonate and substituted bezoic acid with N,N'-dicyclohexylcarbodiimide (DCC) as condensing agent. Twelve of them were reported for the first time. Their structures were clearly established by elemental analysis, IR, 1H NMR and 13C NMR spectra. The antitumor activities of target compounds were tested for the first time. The result indicated that they exhibited certain antitumor activities, and compounds 2i, 2k, 2m showed more potent activities against SGC-7901 with IC50 values of 18.8, 18.5 and 24.5 μmol/L, against A-549 with IC50 values of 24.1, 29.0 and 20.7 μmol/L. Meanwhile, preliminary structure-activity relationships of the title compounds were studied.

Key words: α-hydroxyphosphonate; synthesis; esterification; antitumor activity
/
| 〈 |
|
〉 |