

萘酰亚胺-多胺缀合物的合成、生物活性和荧光光谱
收稿日期: 2013-01-04
修回日期: 2013-01-31
网络出版日期: 2013-02-05
基金资助
中国博士后科学基金(No. 20110490991);河南省自然科学(No. 112300410181);河南省科技攻关(No. 132102310026)和河南省教育厅自然科学基金(No. 2011B3500001)资助项目.
Synthesis, Biological Activitity and Fluorescence Spectroscopy of Naphthalimide-polyamine Conjugates
Received date: 2013-01-04
Revised date: 2013-01-31
Online published: 2013-02-05
Supported by
Project supported by the China Postdoctoral Science Foundation Funded Project (No. 20110490991), the Natural Science Foundation of Henan Province (No. 112300410181), the Henan Provincial Programs for Science and Technology Development (No. 132102310026) and the Natural Science Foundation of Education Department of Henan Province (No. 2011B3500001).
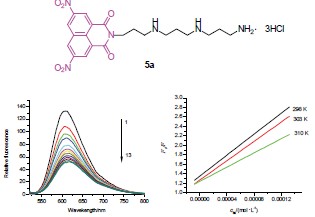
合成了9个新的萘酰亚胺-多胺缀合物, 化合物的结构经元素分析, 1H NMR, 13C NMR和MS确证. 经MTT[3-(4,5-二甲基噻唑-2)-2,5-二苯基四氮唑溴盐]法对白血病细胞(K562)、人乳腺癌细胞(MB-231)和肝癌细胞(7721)体外活性测试. 结果显示多数化合物对肿瘤细胞具有抑作用, 尤其是化合物5a的抗肿瘤活性优于处于III期临床试验阶段药物氨萘非特(Amonafide). 化合物5a与鲱鱼精DNA-EB作用的荧光光谱研究提示: DNA-EB与萘酰亚胺-多胺作用引起的荧光淬灭机制属于静态淬灭, DNA与萘酰亚胺-多胺物结合模式是扦插结合.
田智勇 , 苏雷朋 , 谢松强 , 赵瑾 , 王超杰 . 萘酰亚胺-多胺缀合物的合成、生物活性和荧光光谱[J]. 有机化学, 2013 , 33(07) : 1514 -1521 . DOI: 10.6023/cjoc201301008
Nine novel naphthalimide-polyamine conjugates were synthesized and their structures were confirmed by elemental analysis, 1H NMR, 13C NMR and MS techniques. Their antitumor activities in vitro using MTT[3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay were assessed on leukemia cells (K562), human breast cancer cells (MB-231) and human hepatoma cells (7721). The results showed that most of them exhibited antitumor activities. Especially, compound 5a was better than amonafide which has been in phase III clinical trials. Fluorescence data revealed that the fluorescence quenching of herring sperm DNA-EB complex by compound 5a was a static quenching type and naphthalimide-polyamine conjugates intercalate into the DNA base pairs.

/
| 〈 |
|
〉 |