

4-甲基-3-乙酰基-2-杂环-2H,3H-苯并[f]-1,5-硫氮杂(卄卓)衍生物的合成、抑菌活性及副反应的研究
收稿日期: 2012-12-23
修回日期: 2013-02-08
网络出版日期: 2013-03-14
基金资助
国家自然科学基金(No. 20972040)资助项目.
Synthesis Antimicrobial Activities and Side Reactions of 4-Methyl-3-acetyl-2-heterocyclic-2H,3H-benzo[f]-1,5-thiazepines
Received date: 2012-12-23
Revised date: 2013-02-08
Online published: 2013-03-14
Supported by
Project supported by the National Natural Science Foundation of China (No. 20972040).
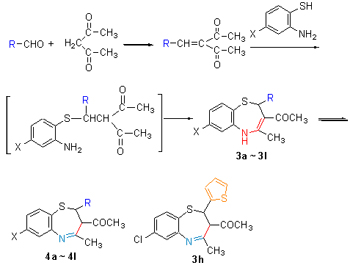
以含不同取代基的杂环醛和乙酰丙酮为原料, 经三步反应合成了一系列新型的4-甲基-3-乙酰基-2-杂环-2H,3H-苯并[f]-1,5-硫氮杂(卄卓)衍生物3a~3l, 产物的结构经1H NMR, IR, MS和元素分析确证, 且通过1H NMR测试, 发现目标化合物存在着烯胺型和亚胺型结构的互变. 测定了目标化合物的抑菌活性, 抑菌结果为杂(卄卓)类化合物的结构与抑菌的关系提供了启示. 同时, 确定了目标化合物3h发生分解反应时主要副产物的结构, 提出了其可能的生成机理.
关键词: 1,5-苯并硫氮杂(卄卓); 杂环; 副反应; 抑菌活性
武云云 , 田克情 , 杨田 , 杜星琼 , 张萍 . 4-甲基-3-乙酰基-2-杂环-2H,3H-苯并[f]-1,5-硫氮杂(卄卓)衍生物的合成、抑菌活性及副反应的研究[J]. 有机化学, 2013 , 33(07) : 1465 -1471 . DOI: 10.6023/cjoc201212027
Starting from substituted heterocyclic aldehyde and acetylacetone, a series of new 4-methyl-3-acetyl-2-heterocyc-lic-2H,3H-benzo[f]-1,5-thiazepines 3a~3l were synthesized through 3-step reactions. The structures of the new products were confirmed by 1H NMR, IR, MS and elemental analysis, and the imine-enamine tautomerism of the target compouds was investigated by means of 1H NMR spectroscopy. Their antifungal activities were evaluated, and the results provides the inspiration for structure-activity reationship of the benzothiazepines. Meanwhile, the structure of the main by-products of 3h was identified, and the possible mechanism of the by-products was proposed.

Key words: 1,5-benzothiazepine; heterocyclic; side reaction; antifungal activity
/
| 〈 |
|
〉 |