

无溶剂超声辐射下磷钨酸铈催化合成5-亚烃基-2,2-亚戊基-1,3-二噁烷-4,6-二酮
收稿日期: 2012-12-23
修回日期: 2013-03-06
网络出版日期: 2013-03-14
基金资助
国家科技攻关计划(No. 2001BA323C)和江西省研究生创新基金(No. YC10A051)资助项目.
Solvent-Free Synthesis of 5-Alkenyl-2,2-pentamethylene-1,3-dioxane-4,6-diones under Ultrasonic Irradiation with CePW12O40 as Catalyst
Received date: 2012-12-23
Revised date: 2013-03-06
Online published: 2013-03-14
Supported by
Project supported by the National Science and Technology Project (No. 2001BA323C) and the Graduate Innovation Fundation of Jiangxi Province (No. YC10A051).
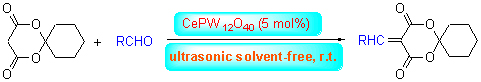
在CePW12O40催化下, 芳香醛和2,2-亚戊基-1,3-二噁烷-4,6-二酮在无溶剂条件下经超声辐射合成了8种5-亚烃基-2,2-亚戊基-1,3-二噁烷-4,6-二酮. 当催化剂的用量为5%(摩尔分数)时, 室温反应20~35 min, 收率为87.6%~94.1%.此外, 还探讨了CePW12O40的可能催化机理. 该方法具有条件温和, 反应时间短且收率高的优点. 催化剂CePW12O40对环境友好且可循环利用.
关键词: 2,2-亚戊基-1,3-二噁烷-4,6-二酮; 磷钨酸铈; 芳香醛; 缩合反应
许招会 , 林春花 . 无溶剂超声辐射下磷钨酸铈催化合成5-亚烃基-2,2-亚戊基-1,3-二噁烷-4,6-二酮[J]. 有机化学, 2013 , 33(07) : 1540 -1544 . DOI: 10.6023/cjoc201212039
Eight kinds of 5-alkenyl-2,2-pentamethylene-1,3-dioxane-4,6-diones were synthesized by the Knoevenagel condensation reaction of aromatic aldehydes with 2,2-pentamethylene-1,3-dioxane-4,6-dione using CePW12O40 as catalyst, without solvent under ultrasonic irradiation. The results indicate that the yields ranged from 87.6% to 94.1% when using 5% (molar fraction) CePW12O40 and reacting at room temperature for 20~35 min. Furthermore, a proposed reaction mechanism for the reaction catalyzed by CePW12O40 was speculated. Compared to the classical Knoevenagel condensation reaction, the main advantages of the present procedure were milder conditions, shorter reaction time and higher yields, which afforded an effective method to synthesize 5-alkenyl-isopropylidene malonate derivatines. Further study showed that CePW12O40 was environmentally friendly and reused for four times without any noticeable decrease in the catalytic activity.

/
| 〈 |
|
〉 |