

吲哚与硝基烯的Friedel-Crafts反应的研究进展
收稿日期: 2013-01-21
修回日期: 2013-03-18
网络出版日期: 2013-04-07
基金资助
国家自然科学基金(Nos. 21262034, 20962018, 20862015, 20562011)资助项目
Research Progress in Friedel-Crafts Reaction of Indoles and Nitroalkenes
Received date: 2013-01-21
Revised date: 2013-03-18
Online published: 2013-04-07
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21262034, 20962018, 20862015, 20562011)
吲哚作为核心结构广泛存在于生物碱和药物中, 吲哚衍生物在生物体的生理活动中起着重要的作用, 其中吲哚和硝基烯的Friedel-Crafts是生成吲哚衍生物的重要方法. 按照反应中催化剂种类的不同分为: (1)无催化剂, (2)离子液体催化剂, (3)金属盐催化剂, (4)配合物催化剂, (5) Brønsted酸催化剂, (6)有机小分子催化剂, (7)其他类型催化剂. 对吲哚与硝基烯的Friedel-Crafts反应的情况进行了综述.
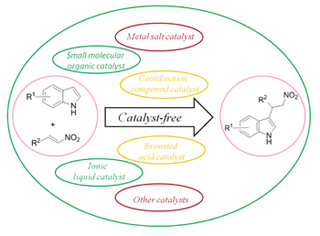
关键词: Friedel-Crafts反应; 吲哚; 硝基烯; 催化剂
王春超 , 谢绍雷 , 解正峰 . 吲哚与硝基烯的Friedel-Crafts反应的研究进展[J]. 有机化学, 2013 , 33(9) : 1919 -1931 . DOI: 10.6023/cjoc201301051
Indoles, as the core structure, exist widely in many alkaloid natural products and pharmaceuticals. The derivatives of indole play an important role in organism physiological activities. The Friedel-Crafts reaction of indoles and nitroalkenes is an important method to generate indole derivatives. According to the kinds of the catalyst in the reaction, the Friedel-Crafts reactions of indoles and nitroalkenes were summarized in this paper as follows: (1) catalyst-free, (2) ionic liquid catalyst, (3) metal salt catalyst, (4) coordination compound catalyst, (5) Brønsted acid catalyst, (6) small molecular organic catalyst, (7) other catalysts.

Key words: Friedel-Crafts reaction; indole; nitroolefin; catalyst
[1] Sartori, G.; Maggi, R. Chem. Rev. 2011, 111, 181.
[2] Zhuang, W.; Gathergood, N.; Hazell, R. G.; Jørgensen, K. A. J. Org. Chem. 2001, 66, 1009.
[3] (a) Choudhury, J.; Podder, S.; Roy, S. J. Am. Chem. Soc. 2005, 127, 6162.
(b) Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F. Tetrahedron Lett. 2003, 44, 5343.
(c) Cardoso, L. A. M.; Jr, W. A.; Gonzaga, A. R. E.; Aguiar, L. M. G.; Andrade, H. M. C. J. Mol. Catal. A: Chem. 2004, 209, 189.
(d) Huang, J. W.; Shi, M. Tetrahedron Lett. 2003, 44, 9343.
(e) Kuwabara, J.; Takeuchi, D.; Osakada, K. J. Mol. Catal. A: Chem. 2004, 208, 39.
(f) Zhou, Y.; Li, X.; Hou, S.; Xu, J. J. Mol. Catal. A: Chem. 2012, 365, 203.
[4] (a) Boon, J. A.; Levisky, J. A.; Pflug, J. L.; Wilkes, J. S. J. Org. Chem. 1986, 51, 480.
(b) Moore, R. E.; Cheuk, C.; Yang, X. Q. G.; Patterson, G. M. L. J. Org. Chem. 1987, 52, 1036.
(c) Dalpozzo, R.; Bartoli, G.; Bencivenni, G. Chem. Soc. Rev. 2012, 41, 7247.
(d) Miller, K. A.; Williams, R. M. Chem. Soc. Rev. 2009, 38, 3160.
[5] (a) Fukuyama, T.; Chen, X. J. Am. Chem. Soc. 1994, 116, 3125.
(b) Moore, R. E.; Cheuk, C.; Patterson, G. M. L. J. Am. Chem. Soc. 1984, 106, 6456.
(c) Chen, Y.-C.; Xie, Z.-F. Chin. J. Org. Chem. 2012, 32, 462 (in Chinese).
(陈永诚, 解正峰, 有机化学, 2012, 32, 462.)
[6] Noland, W. E. Chem. Rev. 1954, 137.
[7] Suter, W.; Hartmann, A.; Poetter, F.; Sagelsdorff, P.; Hoffmann, P.; Martus, H. J. Mater. Res. 2002, 518, 181.
[8] (a) Hudlicky, T.; Entwistle, D. A.; Pitzer, K. K.; Thorpe, A. J. Chem. Rev. 1996, 96, 1195.
(b) Corey, E. J.; Zhang, F. Y. Angew. Chem., Int. Ed. 1999, 38, 1931.
(c) Kieß, F. M.; Poggendorf, P.; Picasso, S.; Jäger, V. Chem. Commun. 1998, 119.
(d) Shiri, M. Chem. Rev. 2012, 112, 3508.
(e) Shiri, M.; Zolfigol, M. A.; Kruger, H. G.; Tanbakouchian, Z. Chem. Rev. 2010, 110, 2250.
[9] Jeon, S. J.; Li, H.; Walsh, P. J. J. Am. Chem. Soc. 2005, 127, 16416.
[10] Habib, P. M.; Kavala, V.; Kuo, C. W.; Yao, C. F. Tetrahedron Lett. 2008, 49, 7005.
[11] Gu, Y.; Barrault, J.; Jérôme F. Adv. Synth. Catal. 2008, 350, 2007.
[12] Habib, P. M.; Kavala, V.; Raju, B. R.; Kuo, C. W.; Huang, W. C.; Yao, C. F. Eur. J. Org. Chem. 2009, 4503.
[13] Rosa, M. D.; Soriente, A. Tetrahedron 2010, 66, 2981.
[14] Habib, P. M.; Kavala, V.; Kuo, C. W.; Raihan, M. J.; Yao, C. F. Tetrahedron 2010, 66, 7050.
[15] Fu, Z.; Shao, H. Ultrason. Sonochem. 2011, 18, 520.
[16] (a) Sheldon, R. Chem. Commun. 2001, 2399.
(b) Dupont, J.; Souza, R. F.; Suarez, P. A. Z. Chem. Rev. 2002, 102, 3667.
(c) Stolte, S.; Abdulkarim, S.; Arning, J.; Blomeyer-Nienstedt, A. K.; Bottin-Weber, U.; Matzke, M.; Ranke, J.; Jastorff, B.; Thöming, J. Green Chem. 2008, 10, 214.
(d) Hasegawa, E.; Hiroi, N.; Osawa, C.; Tayama, E.; Iwamoto, H. Tetrahedron Lett. 2010, 51, 6535.
[17] Yadav, J. S.; Reddy, B. V. S.; Baishya, G.; Reddy, K. V.; Narsaiah, A. V. Tetrahedron 2005, 61, 9541.
[18] Lin, J. H.; Zhang, C. P.; Zhu, Z. Q.; Chen, Q. Y.; Xiao, J. C. J. Fluorine Chem. 2009, 130, 394.
[19] Shen, W.; Wang, L.; Tang, J.; Qian, Z.; Tong, X. Chin. J. Chem. 2010, 28, 443.
[20] Poulsen, T. B.; Jørgensen, K. A. Chem. Rev. 2008, 108, 2903.
[21] Ballini, R.; Gabrielli, S.; Palmieri, A.; Petrini, M. Tetrahedron 2008, 64, 5435.
[22] Tu, Z.; Raju, B. R.; Liou, T. R.; Kavala, V.; Kuo, C. W.; Jang, Y.; Shih, Y. H.; Wang, C. C.; Yao,C. F. Tetrahedron 2009, 65, 2436.
[23] Zahouily, M.; Abrouki, Y.; Rayadh, A.; Sebti, S.; Dhimane, H.; David, M. Tetrahedron. Lett. 2003, 44, 2463.
[24] An, L.; Zhang, L.; Zou, L. J. Chin. J. Chem. 2009, 27, 2223.
[25] Meshram, H. M.; Kumar, D. A.; Reddy, B. C. HeIv. Chim. Acta 2009, 92, 1002.
[26] Schwalm, C. S.; Ceschi, M. A.; Russowsky, D. J. Braz. Chem. Soc. 2011, 22, 623.
[27] (a) Guo, F.; Lai, G.; Xiong, S.; Wang, S.; Wang, Z. Chem. Eur. J. 2010, 16, 6438.
(b) Nakamura, A.; Lectard, S.; Hashizume, D.; Hamashima, Y.; Sodeoka, M. J. Am. Chem. Soc. 2010, 132, 4036.
[28] Huang, G.; Sun, H.; Qiu, X.; Shen, Y.; Jiang, J.; Wang, L. J. Organomet. Chem. 2011, 696, 2949.
[29] Wan, N. N.; Yang, Y. L.; Wang, W. P.; Xie, Z. F.; Wang, J. D. Chin. Chem. Lett. 2011, 22, 1155.
[30] Wan, N.; Hui, Y.; Xie, Z.; Wang, J. Chin. J. Chem. 2012, 30, 311.
[31] Jia, Y. X.; Zhu, S. F.; Zhou, Q. L. J. Org. Chem. 2006, 71, 75.
[32] Singh, P. K.; Bisai, A.; Singh, V. K. Tetrahedron Lett. 2007, 48, 1127.
[33] Itoh, J.; Fuchibe, K.; Akiyama, T. Angew. Chem., Int. Ed. 2008, 47, 4016.
[34] Liu, H.; Lu, S. F.; Xu, J.; Du, D. M. Chem. Asian J. 2008, 3, 1111.
[35] McKeon, S. C.; Müller-Bunz, H.; Guiry, P. J. Eur. J. Org. Chem. 2009, 4833.
[36] Cattoën, X.; Pericàs, M. A. Tetrahedron 2009, 65, 8199.
[37] Wu, L. Y.; Hao, X. Q.; Xu, Y. X.; Jia, M. Q.; Wang, Y. N.; Gong, J. F.; Song, M. P. Organometallics 2009, 28, 3369.
[38] Kim, H Y.; Kim, S.; Oh, K. Angew. Chem. Int. Ed. 2010, 49, 4476.
[39] Liu, H.; Du, D. M. Adv. Synth. Catal. 2010, 352, 1113.
[40] Wu, J.; Li, X.; Wu, F.; Wan, B. Org. Lett. 2011, 13, 4834.
[41] McKeon, S. C.; Müller-Bunz, H.; Guiry, P. J. Eur. J. Org. Chem. 2011, 7107.
[42] Peng, J.; Du, D. M. Eur. J. Org. Chem. 2012, 4042.
[43] (a) Akiyama, T.; Morita, H.; Itoh, J.; Fuchibe, K. Org. Lett. 2005, 7, 2583.
(b) Wabnitz, T. C.; Spencer, J. B. Org. Lett. 2003, 5, 2141.
(c) Uraguchi, D.; Terada, M. J. Am. Chem. Soc. 2004, 126, 5356.
(d) Li, Z.; Zhang, J.; Brouwer, C.; Yang, C. G.; Reich, N. W.; He, C. Org. Lett. 2006, 8, 4275.
(e) Niu, R.; Xiao, J.; Liang, T.; Li, X.; Org. Lett. 2012, 14, 676.
[44] An, L. T.; Zou, J. P.; Zhang, L. L.; Zhang, Y. Tetrahedron Lett. 2007, 48, 4297.
[45] Sheng, Y. F.; Li, G. Q.; Kang, Q.; Zhang, A. J.; You, S. L. Chem. Eur. J. 2009, 15, 3351.
[46] Zheng, C.; Sheng,Y. F.; Li, Y. X. You, S. L. Tetrahedron 2010, 66, 2875.
[47] Li, N.; Chen, X. H.; Zhou, S. M.; Luo, S. W.; Song, J.; Ren, L.; Gong, L. Z. Angew. Chem., Int. Ed. 2010, 49, 6378.
[48] Marqués-López, E.; Alcaine, A.; Tejero, T.; Herrera, R. P. Eur. J. Org. Chem. 2011, 3700.
[49] (a) Kumar, V. P.; Sridhar, R.; Srinivas, B.; Narender, M.; Rao, K. R. Can. J. Chem. 2008, 86, 907.
(b) Ganesh, M.; Seidel, D. J. Am. Chem. Soc. 2008, 130, 16464.
[50] Kuo, C. W.; Wang, C. C.; Fang, H. L.; Raju, B. R.; Kavala, V.; Habib, P. M.; Yao, C. F. Molecules 2009, 14, 3954.
[51] Schafer, A. G.; Wieting, J. M.; Mattson, A. E. Org. Lett. 2011, 13, 5228.
[52] Tran, N. T.; Wilson, S. O.; Franz, A. K. Org. Lett. 2012, 4, 186.
[53] Herrera, R. P.; Sgarzani, V.; Bernardi, L.; Ricci, A. Angew. Chem., Int. Ed. 2005, 44, 6576.
[54] Zhuang, V.; Hazell, R. G.; Jørgensen, K. A. Org. Biomol. Chem. 2005, 3, 2566.
[55] Bui, T.; Syed, S.; Barbas, C. F. J. Am. Chem. Soc. 2009, 131, 8758.
[56] Schneider, J. F.; Falk, F. C.; Fröhlich, R.; Paradies, J. Eur. J. Org. Chem. 2010, 2265.
[57] So, S. S.; Burkett, J. A.; Mattson, A. E. Org. Lett. 2011, 13, 716.
[58] Curti, C.; Rassu, G.; Zambrano, V.; Pinna, L.; Pelosi, G.; Sartori, A.; Battistini, L.; Zanardi, F.; Casiraghi, G. Angew. Chem., Int. Ed. 2012, 51, 1.
[59] Lin, C.; Hsu, J.; Sastry, M. N. V.; Fang, H.; Tu, Z.; Liu, J.-T.; Yao, C.-F. Tetrahedron 2005, 61, 11751.
[60] Kusurkar, R. S.; Alkobati, N. A. H.; Gokule, A. S.; Puranik, V. G. Tetrahedron 2008, 64, 1654.
[61] Chen, Y.-C.; Xie, S.-L.; Xie, Z.-F. Chin. J. Org. Chem. 2012, 32, 1970 (in Chinese).
(陈永诚, 谢绍雷, 解正峰, 有机化学, 2012, 32, 1970.)
/
| 〈 |
|
〉 |