

12-(2-氯苯基)-7,11,13,16-四氧杂二螺[5.2.5.2]十六烷的合成和结构研究
收稿日期: 2013-03-02
修回日期: 2013-04-19
网络出版日期: 2013-04-24
基金资助
湖南省重点建设学科(No. 2011-76);湖南省科技计划(No.2013FJ3004);湖南省教育厅(Nos. 13A030, 13B029)和湖南省创新团队(No. 2012-318)资助项目.
Synthesis and Crystal Structure of 12-(2-Chlorophenyl)-7,11,13,16-tetraoxaspiro[5.2.5.2]cetane
Received date: 2013-03-02
Revised date: 2013-04-19
Online published: 2013-04-24
Supported by
Project supported by the Construct Program of the Key Discipline in Hunan Province (No. 2011-76), the Science and Technology Planning Project of Hunan Province (No. 2013FJ3004), the Scientific Research Fund of Hunan Provincial Education Department (Nos. 13A030, 013B029), and the Science and Technology Innovative Research Team in Higher Educational Institutions of Hunan Province (No. 2012-318)
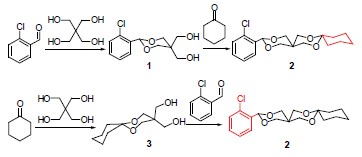
邻氯苯甲醛与季戊四醇反应得到中间体5,5-二(羟甲基)-2-(2-氯苯基)-1,3-二氧六环(1), 1再与环己酮反应生成12-(2-氯苯基)-7,11,13,16-四氧杂二螺[5.2.5.2]十六烷(2). 反之, 先用环己酮与季戊四醇反应生成9,9-二(羟甲基)-7,11-二氧杂螺[5.5]十一烷(3), 3再与邻氯苯甲醛反应也可生成标题化合物2. 化合物1和2的结构用X射线单晶衍射、元素分析和核磁共振氢谱进行了确定. X射线单晶衍射晶体结构表明化合物1属单斜晶系, 空间群为P2(1)/n, 晶体学参数: a=0.56768(15) nm, b=0.9807(3) nm, c=2.1785(6) nm, a=90°, b=90.204(4)°, g=90°, V=1.2128(5) nm3, Z=4, Dc=1.417 Mg/m3, μ=0.315 mm-1, F(000)=544. 化合物2也属于单斜晶系, 空间群为C2/c, 晶体学参数: a=1.5894(5) nm, b=1.5704(4) nm, c=2.8369(9) nm, a=90°, β=101.103(5)°, γ=90°, V=6.948(4) nm3, Z=16, Dc=1.296 Mg/m3, μ=0.237 mm-1, F(000)=2880.
袁霖 , 欧光川 , 时江华 , 张敏 , 袁先友 . 12-(2-氯苯基)-7,11,13,16-四氧杂二螺[5.2.5.2]十六烷的合成和结构研究[J]. 有机化学, 2013 , 33(08) : 1817 -1821 . DOI: 10.6023/cjoc201303003
12-(2-Chlorophenyl)-7,11,13,16-tetraoxaspiro[5.2.5.2]cetane (2) was prepared by cyclohexanone and 5,5-bis(hydroxymethyl)-2-(2-chlorophenyl)-1,3-dioxane (1) which synthesized from the reaction of the o-chlorobenzaldehyde and pentaerythriol, or prepared by o-chlorobenzaldehyde and 9,9-bis(hydroxymethyl)-7,11-dioxaspiro[5,5]undecane (3) which synthesized from the reaction of the cyclohexanone and pentaerythriol. Compounds 1 and 2 were characterized by X-ray diffraction, elemental analysis, 1H NMR. Single crystal X-ray diffraction analyses reveal that compound 1 belongs to monoclinic system with space group P2(1)/n with a=0.56768(15) nm, b=0.9807(3) nm, c=2.1785(6) nm, a=90°, b=90.204(4)°, g=90°, V= 1.2128(5) nm3, Z=4, Dc=1.417 Mg/m3, μ=0.315 mm-1, F(000)=544. Compound 2 also belongs to monoclinic system with space group C2/c with a=1.5894(5) nm, b=1.5704(4) nm, c=2.8369(9) nm, a=90°, b=101.103(5)°, g=90°, V=6.948(4) nm3, Z=16, Dc=1.296 Mg/m3, μ=0.237 mm-1, F(000)=2880.

Key words: o-chlorobenzaldehyde; pentaerythriol; crystal structure; 1H NMR
/
| 〈 |
|
〉 |