

3,6-二取代-1,2,4-三嗪衍生物的微波辅助合成及生物活性
收稿日期: 2013-04-08
修回日期: 2013-05-14
网络出版日期: 2013-06-21
基金资助
辽宁省自然科学基金(No. 20102126)资助项目
Microwave-Assisted Synthesis and Biological Activities of 3,6-Disubstituted-1,2,4-triazine Derivatives
Received date: 2013-04-08
Revised date: 2013-05-14
Online published: 2013-06-21
Supported by
Project supported by the Natural Science Foundation of Liaoning Province (No. 20102126).
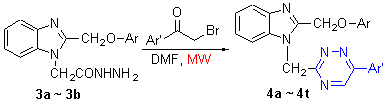
在微波辐射下, 利用2-芳氧甲基苯并咪唑-1-乙酰肼(3)与各种(未)取代的溴代苯乙酮进行缩合, 合成出了20个新的含苯并咪唑单元的3,6-二取代-1,2,4-三嗪衍生物4, 并利用1H NMR, IR和元素分析对所合成的目标化合物进行了结构表征. 探索出了最佳合成条件. 与经典方法相比较, 该合成方法具有反应时间短、操作简便、底物范围广、易纯化、产率较高等优点. 对所合成的目标化合物进行了Cdc25B和PTP1B抑制活性的筛选. 实验结果表明, 目标化合物4g和4j对Cdc25B表现出良好的抑制活性, 目标化合物4a, 4h, 4j, 4r和4s对PTP1B表现出良好的抑制活性. 值得注意的是, 目标化合物4j可作为用于治疗癌症和糖尿病的潜在的Cdc25B和PTP1B抑制剂.
关键词: 微波辐射; 1,2,4-三嗪; 苯并咪唑; 合成; Cdc25B和PTP1B抑制剂
李英俊 , 邵昕 , 高立信 , 靳焜 , 盛丽 , 罗潼川 , 于洋 , 李佳 . 3,6-二取代-1,2,4-三嗪衍生物的微波辅助合成及生物活性[J]. 有机化学, 2013 , 33(10) : 2178 -2185 . DOI: 10.6023/cjoc201304011
Twenty new 3,6-disubstituted-1,2,4-triazine derivatives containing benzimidazole moiety (4) were synthesized via microwave-assisted condensation reactions of 2-aryloxymethylbenzimidazole-1-acetylhydrazines (3) with various (un)substituted phenacyl bromides in DMF. The structures were characterized by IR, 1H NMR spectra and elemental analysis. The optimum experimental conditions were found. Compared with traditional methods, this synthetic method has such advantages as short reaction time, simple operation, broad substrate scope, easy purification and high yields. The synthesized target compounds were screened for Cdc25B and PTP1B inhibitory activities. The experimental results indicate that compounds 4g and 4j showed good inhibitory activities against Cdc25B, and 4a, 4h, 4j, 4r and 4s exhibited potent inhibitory activities against PTP1B. It is noteworthy that compound 4j can be used as potential Cdc25B and PTP1B inhibitors in the treatment of cancer and diabetes.

/
| 〈 |
|
〉 |