

4-甲基-2-杂环-2,3-二氢苯并[f]-1,5-硫氮杂(艹卓)-3-羧酸乙酯及其还原产物的合成、晶体结构及抑菌活性的研究
收稿日期: 2013-05-24
修回日期: 2013-06-10
网络出版日期: 2013-06-21
基金资助
国家自然科学基金(No. 20972040)资助项目
Studies on the Synthesis, Crystal Structure and Antimicrobial Activities of 4-Methyl-2-heterocylic-2,3-dihydro-benzo[f]-1,5-thiazepine-3-carboxylic Acid Ethyl Ester and Its Reduction Products
Received date: 2013-05-24
Revised date: 2013-06-10
Online published: 2013-06-21
Supported by
Project supported by the National Natural Science Foundation of China (No. 20972040).
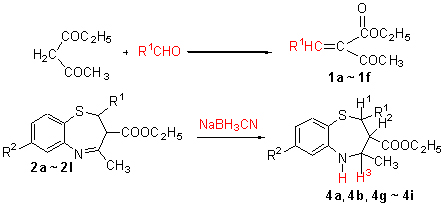
以乙酰乙酸乙酯和含不同取代基的杂环醛为原料, 经两步反应合成了一系列新颖的4-甲基-2-杂环-2,3-二氢苯并[f]-1,5-硫氮杂(艹卓)-3-羧酸乙酯(2a~2l), 并对其中的5个化合物进行了还原, 2a~2l及还原产物的结构经1H NMR IR, MS, 元素分析确证. 同时采用X射线衍射法测定了2a的晶体结构, 2a属三斜晶系, P-1空间群. 采用滤纸片法测试所合成目标化合物对枯草芽孢杆菌、白色念珠菌、标准新生隐球菌和临床新生隐球菌的抑菌活性, 结果表明大部分化合物都有一定的抑菌效果, 其中2a的抑菌效果尤其突出.
关键词: 1,5-苯并硫氮杂(艹卓); 杂环; 还原; 晶体结构; 抑菌活性
田克情 , 刘晓欣 , 薛子桥 , 杨田 , 杜星琼 , 张萍 . 4-甲基-2-杂环-2,3-二氢苯并[f]-1,5-硫氮杂(艹卓)-3-羧酸乙酯及其还原产物的合成、晶体结构及抑菌活性的研究[J]. 有机化学, 2013 , 33(10) : 2237 -2243 . DOI: 10.6023/cjoc201305039
Starting from ethyl acetoacetate and substituted heterocyclic aldehyde, a series of new 4-methyl-2-heterocyclic-2,3-dihydro-benzo[f]-1,5-thiazepine-3-carboxylic acid ethyl esters (2a~2l) were synthesized through 2-step reactions, and of which five compounds were reduced. The structures of the compounds were characterized by 1H NMR, IR, MS, elemental analysis and X-ray diffraction study. The crystal of compound 2a belongs to triclinic system, space group P-1. The antibacterical activities of the new compounds against B. subtilis, C. albicans and C. neoformans were evaluated by filter paper approach, and the results indicated that most of them showed antibacterical activities, especially the compound 2a prominent.

/
| 〈 |
|
〉 |