

酰胺为氮源的氨化反应及其机理
Amide as Nitrogen Source: Amination and Mechanism
Received date: 2013-06-04
Revised date: 2013-06-21
Online published: 2013-07-10
通过实验证明酰胺作为氮源在分子中引入胺基时,首先发生酰胺碳氮键的断裂生成胺的Lewis酸复合物,该复合物再与底物发生反应生成新的碳氮键. 以酰胺为氮源,在Lewis酸的配合下,将卤代芳烃、芳香三氟甲磺酸酯、硼酸、芳香硅化物和苯并唑类化合物胺化,证实这种新胺化方法具有很好的普适性.
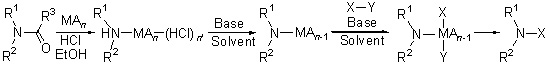
关键词: 酰胺; 氮源; 有机胺的Lewis酸复合物; 机理; 碳氮键断裂和构建
徐娟 , 李加荣 , 魏真 , 张奇 , 史大昕 . 酰胺为氮源的氨化反应及其机理[J]. 有机化学, 2013 , 33(11) : 2435 -2439 . DOI: 10.6023/cjoc201305053
The mechanism of C—N bond formation with amide as nitrogen source was disclosed. The process included the cleavage of amide C—N bond to form amine and then to construct new C—N bond. With the cooperation of Lewis acid, aryl halides, aryl triflates, boronic acids, aryl siloxanes and azoles could be aminated by amide as nitrogen source.
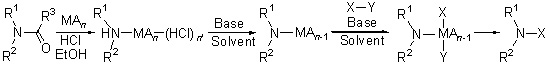
[1] (a) He,F.; Foxman,B.M.; Snider,B.B.J.Am.Chem.Soc.1998,120,6417.
(b) Wang,M.; Liang,Y.; Song,Z.G.; Jiang,H.Chin.J.Org.Chem.2010,30,295 (in Chinese).(王敏,梁燕,宋志国,姜恒,有机化学,2010,30,295.)
(c) Zhang,G.P.; Xia,Y.Chin.J.Org.Chem.2010,30,449 (in Chinese).(张国平,夏燕,有机化学,2010,30,449.)
(d) Boonen,J.; Bronselaer,A.; Nielandt,.J.; Veryser,L.; De Tre,G.; De Spiegeleer,B.J.Ethnopharmacol.2012,142,563.
(e) Wei,Y.; Shi,D.; Wei,Z.; Xu,J.; Li,J.Chin.J.Org.Chem.2012,32,1126 (in Chinese).(魏莹菲,史大昕,魏真,徐娟,李加荣,有机化学,2012,32,1126.)
[2] (a) Kim,Y.M.; Yu,S.J.Am.Chem.Soc.2003,125,1696.
(b) Zim,D.; Buchwald,S.L.Org.Lett.2003,5,2413.
(c) Ma,D.; Cai,Q.; Zhang,H.Org.Lett.2003,5,2453.
(d) Urgaonkar,S.; Nagarajan,M.; Verkade,J.G.Org.Lett.2003,5,815.
(e) Leadbeater,N.E.; Marco,M.Angew.Chem.,Int.Ed.2003,42,1407.
(f) Fang,S.; Lv,M.X.; Long,Y.H.; Yang,D.Q.Chin.J.Org.Chem.2011,31,1573 (in Chinese).(方晒,吕梅香,龙玉华,杨定乔,有机化学,2011,31,1573.)
[3] (a) Coalter,J.N.; Huffman,J.C.; Caulton,K.G.Organometallics 2000,19,3569.
(b) Zhang,M.Synthesis 2011,3408.
(c) Weibel,J.M.; Blanc,A.; Pale,P.Chem.Rev.2008,108,3149.
(d) Furuya,T.; Storm,A.E.; Ritter,T.J.Am.Chem.Soc.2009,131,1662.
[4] Bogdal,D.Molecules 1999,4,333.
[5] Demko,Z.P.; Bartsch,M.; Sharpless,K.B.Org.Lett.2000,2,2221.
[6] Xiong,T.; Li,Y.; Lv,Y.; Zhang,Q.Chem.Commun.2010,46,6831.
[7] Barros,O.S.; Nogueira,C.W.; Stangherlin,E.C.; Menezes,P.H.; Zeni,G.J.Org.Chem.2006,71,1552.
[8] (a) Zhao,J.K.; Wang,Y.G.Chin.Chem.Lett.2002,13,1149.
(b) Agarwal,A.; Chauhan,P.M.S.Synth.Commun.2004,34,2925.
(c) Cho,S.H.; Kim,J.Y.; Lee,S.Y.; Chang,S.Angew.Chem.,Int.Ed.2009,48,9127.
(d) Samadi,A.; Silva,D.; Chioua,M.; Carreiras,M.; Mar-co-Contelles,J.Synth.Commun.2011,41,2859.
(e) Chen,W.X.; Shao,L.X.J.Org.Chem.2012,77,9236.
[9] Li,J.R.; Chen,X.; Shi,D.X.; Ma,S.L.; Li,Q.; Zhang,Q.; Tang,J.H.Org.Lett.2009,11,1193.
[10] (a) Xu,J.; Shi,D.X.; Wei,Z.; Wei,Y.F.; Zhang,Q.; Li,J.R.Chin.J.Org.Chem.2012,32,776(in Chinese).(徐娟,史大昕,魏真,魏莹菲,张奇,李加荣,有机化学,2012,32,776.)
(b) Xu,J.; Li,J.R.; Wei,Z.; Zhang,Q.; Shi,D.X.RSC Adv.2013,3,9622.
[11] Xu,J.; Li,J.R.J.Chem.Res.2012,36,381.
[12] Senear,A.E.; Rapport,M.M.; Mead,J.F.J.Org.Chem.1946,11,378.
[13] Medici,A.J.A.J.Chem.Soc.,Perkin Trans.1 1977,22,2517.
[14] Kost,A.N.Zh.Obshch.Khim.1964,34,4046.
[15] Brown,B.R.J.Chem.Soc.1957,3755.
[16] Pedersen,E.B.Synthesis 1978,844.
[17] Komaromi,A.Adv.Synth.Catal.2010,352,1523.
[18] Gribble,G.W.Synthesis 1987,709.
[19] Kohler,P.C.; Ritschel,T.; Schweizer,W.B.Chem.Eur.J.2009,15,10809.
[20] Prouillac,C.; Vicendo,P.; Garrigues,J.C.Free Radicals Biol.Med.2009,46,1139.
[21] Mavrova,A.T.; Denkova,P.; Tsenov,Y.A.Biol.Med.Chem.2007,15,6291.
[22] (a) Visnjevac,A.; Tusek-Bozic,L.; Majeric-Elenkov,M.; Hamersak,Z.; Kooijman,H.; De Clercq,E.; Kojic-Prodic,B.Polyhedron 2002,21,2567.
(b) Usama,E.A.; Alaa,A.M.A.A; Shar,A.S.Eur.J.Med.Chem.2007,42,1325.
(c) Do,H.-Q.; Daugulis,O.J.Am.Chem.Soc.2009,131,17052.
(d) Bresser,T.; Mosrin,M.; Monzon,G.; Knochel,P.J.Org.Chem.2010,75,4686.
(e) Zimdars,S.; Jourdin,X.M.D.; Crestey,F.; Carell,T.; Knochel,P.Org.Lett.2011,13,792.
(f) Duez,S.; Bernhardt,S.; Heppekausen,J.; Fleming,F.F.; Knochel,P.Org.Lett.2011,13,1690.
/
| 〈 |
|
〉 |