

柚皮苷半合成E-柚皮素肟类化合物及细胞活性
收稿日期: 2013-07-05
修回日期: 2013-07-30
网络出版日期: 2013-08-16
基金资助
国家自然科学基金(No. 81060261)、广西自然科学基金重点(No. 2011jjD20002)和广西师范学院博士点建设开放课题资助项目.
Semisynthesis and Cytotoxicity of E-Naringenin Oximes from Naringin
Received date: 2013-07-05
Revised date: 2013-07-30
Online published: 2013-08-16
刘志平 , 韦万兴 , 甘春芳 , 黄燕敏 , 刘盛 , 周敏 , 崔建国 . 柚皮苷半合成E-柚皮素肟类化合物及细胞活性[J]. 有机化学, 2013 , 33(12) : 2551 -2558 . DOI: 10.6023/cjoc201307008
Naringenin (N-1) and naringenin derivatives N-2~N-6 were synthesized by the hydrolysis, selective methylation or selective O-prenylation using natural naringin as starting material. Six E-naringenin oximes N-1a~N-6a and ten E-naringenin oxime ethers N-1b~N-6c were synthesiszed by reacting N-1~N-6 with hydroxylamine hydrochloride, methoxylamine hydrochloride and benzyloxygen amine hydrochloride, respectively. The structures of the compounds were identified by NMR and HR-ESI-MS. The cytotoxicities of the compounds were evaluated by MTT assay and the results showed that N-1a, N-1b, N-1c, N-2a, N-4a displayed moderate activity against human cancer cell line SGC-7901.
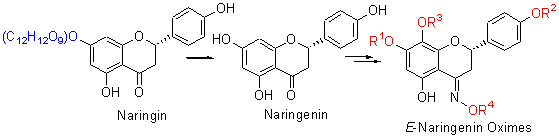
Key words: naringin; naringenin; E-naringenin oximes; semisynthesis; cytotoxicity
[1] Borska, S.; Chmielewska, M.; Wysocka, T.; Zalesinska, M. D.; Zabel, M.; Dziegiel, P. Food Chem. Toxicol. 2012, 50(9), 3375.
[2] Snyder, R. D.; Gillies, P. J. Environ. Mol. Mutagen. 2002, 40, 266.
[3] Chen, C.; Li, X.; Hu, X. F.; Yin, S. F. Chin. J. Org. Chem. 2011, 11, 1878 (in Chinese). (陈超, 李霞, 胡晓枫, 李颖, 尹述凡, 有机化学, 2011, 11, 1878.)
[4] Chavi, Y. J.; Suchana, W. Bioorg. Med. Chem Lett. 2010, 20, 2821.
[5] Chavi, Y. J.; Suchana, W.; Siripit, P.; Bungon, S. Arch. Pharm. Res. 2009, 32, 1179.
[6] Turkkan, B.; Ozyurek, M.; Bener, M.; Guclu, K.; Apak, R. Spectrochim. Acta A 2012, 85, 235
[7] Liu, Z. P.; Wei, W. X.; Gan, C. F.; Huang, Y. M.; Liu, S.; Cui, J. G. J. Org. Chem. 2013, 33, 1970 (in Chinese). (刘志平, 韦万兴, 甘春芳, 黄燕敏, 刘盛, 崔建国, 有机化学, 2013, 33, 1970.)
[8] Ramesh, E.; Alshatwi, A. A. Food. Chem. Toxicol. 2013, 51, 97
[9] Kim, J. H.; Kang, J. W.; Kim, M. S.; Bak, Y.; Park, Y. S.; Jung, K. Y.; Lim, ` Y. H.; Yoon, D. Y. Toxicol. In Vitro 2006, 26, 67.
[10] Liu, Y.; Wu, H.; Nie, Y. C.; Chen, J. L.; Su, W. W.; Li, P. B. Toxicol. Lett. 2013, 220(3), 219.
[11] Shan, Y.; Li, G. Y.; Wang, Q. A.; Li, Z. H. Chin. J. Org. Chem. 2008, 28, 1024 (in Chinese). (单杨, 李高阳, 汪秋安, 李忠海, 有机化学, 2008, 28, 1024.)
[12] Cai, S. L.; Wu, Z.; Wu, J.; Wang, Q. A.; Shan, Y. Chin. J. Org. Chem. 2012, 32, 560 (in Chinese). (蔡双莲, 吴峥, 吴进, 汪秋安, 单杨, 有机化学, 2012, 32, 560.)
[13] Cui, J. G.; Fan, Lei,; Huang, Y. M.; Xin, Y.; Zhou, A. M. Steroids 2009, 74, 989.
/
| 〈 |
|
〉 |