

过渡金属催化端炔的氧化交叉偶联反应研究进展
收稿日期: 2013-07-20
修回日期: 2013-09-03
网络出版日期: 2013-09-17
基金资助
山西省自然科学基金(Nos. 2012021007-2,2011011010-2)及山西省高等教育机构科技创新(No. 20120006)资助项目.
Progress in Transition-Metal-Catalyzed Oxidative Cross-Coupling of Terminal Alkynes
Received date: 2013-07-20
Revised date: 2013-09-03
Online published: 2013-09-17
Supported by
Project supported by the Natural Science Foundation of Shanxi Province (Nos. 2012021007-2, 2011011010-2), and the Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi Province (No. 20120006).
张聪霞 , 李娜娜 , 李兴 , 常宏宏 , 刘强 , 魏文珑 . 过渡金属催化端炔的氧化交叉偶联反应研究进展[J]. 有机化学, 2014 , 34(1) : 81 -91 . DOI: 10.6023/cjoc201307024
Alkynyl compounds are one kind of important functional compounds in organic synthesis. Various new alkynyl compounds can be obtained via oxidative cross-coupling reactions of terminal alkynes. Recent oxidative cross-coupling reactions involving terminal alkynes are reviewed. The advances of cross-coupling reactions catalyzed by Pd, Cu, Fe and so on are particularly described.
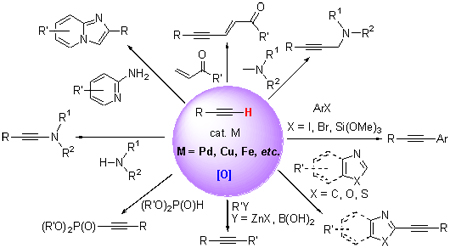
Key words: transition metal; C—H bond; terminal alkyne; oxidative cross-coupling; progress
[1] Ullmann, F. Ber. Dtsch. Chem. Ges. 1903, 36, 2382.
[2] Zhang, B. B.; Zhan, D.; Zhang, X. P.; Xiang, Q. J.; Zeng, Q. L. Acta Chim. Sinica 2012, 70, 1655 (in Chinese).
(张斌彬, 詹丹, 张小平, 向沁洁, 曾庆乐, 化学学报, 2012, 70, 1655.)
[3] Darses, S.; Genet, J. P. Chem. Rev. 2008, 108, 288.
[4] Miyaura, N.; Suzuki, A. Chem. Rev. 1995, 95, 2457.
[5] Alonso, F.; Beletskaya, I. P.; Yus, M. Tetrahedron 2005, 61, 11771.
[6] (a) Li, X.; Yan, X. Y.; Chang, H. H.; Wang, L. C.; Zhang, Y.; Chen, W. W.; Li, Y. W.; Wei, W. L. Org. Biomol. Chem. 2012, 10, 495.
(b) Li, X.; Wang, L. C.; Chang, H. H.; Zhang, C. X.; Wei, W. L. Appl. Catal. A: Gen. 2013, 462~463, 15.
[7] Wen, Y. M.; Jiang, H. F. Acta Chim. Sinica 2012, 70, 1716 (in Chinese).
(温燕梅, 江焕峰, 化学学报, 2012, 70, 1716.)
[8] Ritleng, V.; Sirlin, C.; Pfeffer, M. Chem. Rev. 2002, 102, 1731.
[9] Arockiam, P. B.; Bruneau, C.; Dixneuf, P. H. Chem. Rev. 2012, 112, 5879.
[10] Jia, C. G.; Kitamura, T.; Fujiwara, Y. Acc. Chem. Res. 2001, 34, 633.
[11] Li, H.; Li, B. J.; Shi, Z. J. Catal. Sci. Technol. 2011, 1, 191.
[12] (a) Pan, F.; Shi, Z. J. Acta Chim. Sinica 2012, 70, 1679 (in Chinese).
(潘菲, 施章杰, 化学学报, 2012, 70, 1679.;
b) Zhang, D.; Qin, Y. Acta Chim. Sinica 2013, 71, 147 (in Chinese).
(张丹, 秦勇, 化学学报, 2013, 71, 147.)
[13] Li, C. J. Acc. Chem. Res. 2009, 42, 335.
[14] Li, Z. P.; Li, C. J. J. Am. Chem. Soc. 2005, 127, 3672.
[15] Cai, G. X.; Fu, Y.; Li, Y. Z.; Wan, X. B.; Shi, Z. J. J. Am. Chem. Soc. 2007, 129, 7666.
[16] Huber, S. M.; Ertem, M. Z.; Aquilante, F.; Gagliardi, L.; Tolman, W. B.; Cramer, C. J. Chem. Eur. J. 2009, 15, 4886.
[17] Sonogashira, K. J. Organomet. Chem. 2002, 653, 46.
[18] Glaser, C. Ber. Dtsch. Chem. Ges. 1869, 2, 422.
[19] Glaser, C. Ann. Chem. Pharm. 1870, 154, 137.
[20] Kim, S. H.; Yoon, J.; Chang, S. Org. Lett. 2011, 13, 1474.
[21] Liang, B.; Dai, M. J.; Chen, J. H.; Yang, Z. J. Org. Chem. 2005, 70, 391.
[22] Li, J. H.; Liang, Y.; Xie, Y. X. J. Org. Chem. 2005, 70, 4393.
[23] Hadi, V.; Yoo, K. S.; Jeong, M.; Jung, K. W. Tetrahedron Lett. 2009, 50, 2370.
[24] Chen, M.; Zheng, X. L.; Li, W. Q.; He, J.; Lei, A. W. J. Am. Chem. Soc. 2010, 132, 4101.
[25] Luh, T. Y.; Leung, M. K.; Wong, K. T. Chem. Rev. 2000, 100, 3187.
[26] Facoetti, D.; Abbiati, G.; d'Avolio, L.; Ackermann, L.; Rossi, E. Synlett 2009, 2273.
[27] Yue, D. W.; Larock, R. C. Org. Lett. 2004, 6, 1037.
[28] Zhang, H. M.; Larock, R. C. J. Org. Chem. 2002, 67, 7048.
[29] Gu, Y. H.; Wang, X. M. Tetrahedron Lett. 2009, 50, 763.
[30] Besselièvre, F.; Piguel, S. Angew. Chem., Int. Ed. 2009, 48, 9553.
[31] Brand, J. P.; Charpentier, J.; Waser, J. Angew. Chem., Int. Ed. 2009, 48, 9346.
[32] Yang, L.; Zhao, L.; Li, C. J. Chem. Commun. 2010, 46, 4184.
[33] Zou, G.; Zhu, J. R.; Tang, J. Tetrahedron Lett. 2003, 44, 8709.
[34] Yang, F.; Wu, Y. J. Eur. J. Org. Chem. 2007, 3476.
[35] Ye, Z. S.; Liu, M. C.; Lin, B. D.; Wu, H. Y.; Ding, J. C.; Cheng, J. Tetrahedron Lett. 2009, 50, 530.
[36] Wu, C. R.; Li, P.; Fang, Y. S.; Zhao, J. J.; Xue, W. C.; Li, Y.; Larock, R. C.; Shi, F. Tetrahedron Lett. 2011, 52, 3797.
[37] Jie, X. M.; Shang, Y. P.; Hu, P.; Su, W. P. Angew. Chem., Int. Ed. 2013, 52, 3535.
[38] Zhou, L.; Ye, F.; Ma, J. C.; Zhang, Y.; Wang, J. B. Angew. Chem., Int. Ed. 2011, 50, 3510.
[39] Hamada, T.; Ye, X.; Stahl, S. S. J. Am. Chem. Soc. 2008, 130, 833.
[40] Wang, L.; Huang, H.; Priebbenow, D. L.; Pan, F. F.; Bolm, C. Angew. Chem., Int. Ed. 2013, 52, 3478.
[41] Wei, Y.; Zhao, H. Q.; Kan, J.; Su, W. P.; Hong, M. C. J. Am. Chem. Soc. 2010, 132, 2522.
[42] Matsuyama, N.; Kitahara, M.; Hirano, K.; Satoh, T.; Miura, M. Org. Lett. 2010, 12, 2358.
[43] Iorga, B.; Eymery, F.; Carmichael, D.; Savignac, P. Eur. J. Org. Chem. 2000, 3103.
[44] Lera, M.; Hayes, C. J. Org. Lett. 2000, 2, 3873.
[45] Gao, Y. X.; Wang, G.; Chen, L.; Xu, P. X.; Zhao, Y. F.; Zhou,Y. B.; Han, L. B. J. Am. Chem. Soc. 2009, 131, 7956.
[46] Kitahara, M.; Hirano, K.; Tsurugi, H.; Satoh, T.; Miura, M. Chem. Eur. J. 2010, 16, 1772.
[47] Chu, L. L.; Qing, F. L. J. Am. Chem. Soc. 2010, 132, 7262.
[48] Li, Z. P.; Li, C. J. J. Am. Chem. Soc. 2004, 126, 11810.
[49] Zhao, L.; Li, C. J. Angew. Chem., Int. Ed. 2008, 47, 7075.
[50] Bi, H. P.; Zhao, L.; Liang, Y. M.; Li, C. J. Angew. Chem., Int. Ed. 2009, 48, 792.
[51] Niu, M. Y.; Yin, Z. M.; Fu, H.; Jiang, Y. Y.; Zhao, Y. F. J. Org. Chem. 2008, 73, 3961.
[52] Pan, C. D.; Luo, F.; Wang, W. H.; Ye, Z. S.; Cheng, J. Tetrahedron Lett. 2009, 50, 5044.
[53] Jiang, X. L.; Chu, L. L.; Qing, F. L. Org. Lett. 2012, 14, 2870.
[54] Xu, X. L.; Li, X. N. Org. Lett. 2009, 11, 1027.
[55] Xu, Z. W.; Yu, X. Q.; Feng, X. J.; Bao, M. J. Org. Chem. 2011, 76, 6901.
[56] Stavropoulos, P.; Çelenligil-Çetin, R.; Tapper, A. E. Acc. Chem. Res. 2001, 34, 745.
[57] Walling, C. Acc. Chem. Res. 1998, 31, 155.
[58] Volla, C. M. R.; Vogel, P. Org. Lett. 2009, 11, 1701.
[59] (a) Horner, L.; Junkermann, H. Ann. Chem. Justus Liebig 1955, 591, 53.
(b) Horner, L.; Kirmse, W. Ann. Chem. Justus Liebig 1955, 597, 48.
[60] Meng, X.; Li, C. B.; Han, B. C.; Wang, T. S.; Chen, B. H. Tetrahedron 2010, 66, 4029.
[61] You, X. L.; Xu, L.; Hu, T. Lett. Org. Chem. 2012, 9, 300.
[62] Yin, W. Y.; He, C.; Chen, M.; Zhang, H.; Lei, A. W. Org. Lett. 2009, 11, 709.
[63] He, C.; Hao, J.; Xu, H.; Mo, Y. P.; Liu, H. Y.; Han, J. J.; Lei, A. W. Chem. Commun. 2012, 48, 11073.
[64] He, C.; Guo, S.; Ke, J.; Hao, J.; Xu, H.; Chen, H. Y.; Lei, A. W. J. Am. Chem. Soc. 2012, 134, 5766.
[65] de Haro, T.; Nevado, C. J. Am. Chem. Soc. 2010, 132, 1512.
/
| 〈 |
|
〉 |