

卤素功能化靛红的合成及其烃基化、还原和酰基化反应研究
收稿日期: 2013-07-14
修回日期: 2013-09-14
网络出版日期: 2014-09-30
基金资助
辽宁省自然科学基金(No. 20120001)资助项目.
Synthesis, Alkylation, Reduction and Acylation of Halo-functionalized Isatins
Received date: 2013-07-14
Revised date: 2013-09-14
Online published: 2014-09-30
Supported by
Project supported by the Natural Science Foundation of Liaoning Province (No. 20120201).
高文涛 , 赵鹏波 , 赵宾宾 , 李阳 . 卤素功能化靛红的合成及其烃基化、还原和酰基化反应研究[J]. 有机化学, 2014 , 34(1) : 126 -136 . DOI: 10.6023/cjoc201307020
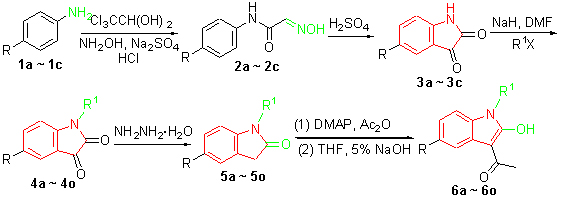
In this paper, 4-haloanilines were first underwent the Sandmeyer reaction to give the corresponding halo-functionalized isatins 3a~3c, which were further alkylated at the nitrogen atom to give 4a~4o followed by in situ reduction using hydrazine hydrate to obtain the oxindole products 5a~5o. 5a~5o were subjected to the acetylation reaction by the treatment with acetic anhydride in the presence of a catalytic amount of N,N-dimethylaminopyridine to afford the intermediates of 2-acetoxy-3-acetylindoles, which were used in next step without further purifucation. Subsequently, the hydrolysis reaction of the arising ester functions of the intermediates was carried out in tetrahydrofuran (THF) with the presence of 5% aq. NaOH at room temperature to give the targeted compounds 6a~6o. Some compounds synthesized are novel. Their structures were confirmed by 1H NMR, 13C NMR, IR, MS and elemental analysis.
Key words: halo-functionalized isatin; reduction; alkylation; acetylation
[1] Jun, Q. R.; Ning, H.; Hui, X.; Liu, M. Y.; Min, L.; Yong, T. Z. Bioorg. Med. Chem. Lett. 2010, 20, 3534.
[2] Pais, G. C. G.; Zhang, X.; Marchand, C.; Neamati, N.; Cowansage, K.; Svarovskaia, E. S.; Pathak, V. K.; Tang, Y.; Nicklaus, M.; Pommier, Y.; Burke, Jr. T. R. J. Med. Chem. 2002, 45, 3184.
[3] Ferro, S.; Barreca, M. L.; Luca, L. D.; Rao, A.; Monforte, A. M.; Debyser, Z.; Witrouw, M.; Chimirri, A. Arch. Pharm. Chem. Life Sci. 2007, 340, 292.
[4] Abdel-gawad, H.; Mohamed, H. A.; Dawood, K. M.; Badria, F. A.-R. Chem. Pharm. Bull. 2010, 58, 1529.
[5] Sriniva, P.; Raghavan, S. A. V.; Jagadeeh-bababu, R.; Gupta, C. N. V. H. B.; Sridhar, N.; Veeranjaneyulu, A.; Parimoo, P. Pharmacol. Commun. 1999, 5, 95.
[6] Giraud, F.; Alves, G.; Debiton, E.; Nauton, L.; Thery, V.; Durieu, E.; Ferandin, Y.; Lozach, O.; Meijer, L.; Anizon, F.; Pereira, E.; Moreau, P. J. Med. Chem. 2011, 54, 4474.
[7] Chennamaneni, S.; Zhong, B.; Lama, R.; Su, B. Eur. J. Med. Chem. 2012, 56, 17.
[8] Bruel, A.; Logé, C.; Tauzia, M. L.; Ravache, M.; Guevel, R.; Guillouzo, C.; Lohier, J. F.; Oliveira, Santos. J. S.; Lozach, O.; Meijer, L.; Ruchaud, S.; Bénédetti, H.; Robert, J. M. Eur. J. Med. Chem. 2012, 57, 225.
[9] Martel-Frachet, V.; Kadri, M.; Boumendjel, A.; Ronot, X. Bioorg. Med. Chem. 2011, 19, 6143.
[10] Kumar, D.; Kumar, N. M.; Sundaree, S.; Johnson, E. O.; Shah, K. Eur. J. Med. Chem. 2010, 45, 1244.
[11] Subba Reddy, B. V. S.; Rajeswari, N.; Sarangapani, M.; Reddy, G. R.; Msdan, C.; Kumar, K. P.; Rao, M. S. Bioorg. Med. Chem. Lett. 2011, 21, 6510.
[12] Subba, R. B. V.; Rajeswari, N.; Sarangapani, M.; Prashanthi, Y.; Ganji, R. J.; Addlagatta, A. Bioorg. Med. Chem. Lett. 2012, 22, 2460.
[13] Jørgensen, M.; Jørgensen, P. N.; Christoffersen, C. T.; Jensen, K. G.; Balle, T.; Bang-Andersen, B. Bioorg. Med. Chem. 2013, 21, 196.
[14] Shen, X. Q.; Wu Y. L.; Qian, H. J. Chem. Ind. Times 2012, 26, 29 (in Chinese).
(沈学全, 吴金龙, 钱海均, 化工时刊, 2012, 26, 29.)
[15] Zhang, X. F.; Liu, H. Y.; Gao, W. T. J. Bohai Univ. (Nat. Sci.) 2009, 30, 212 (in Chinese).
(张晓飞, 刘华业, 高文涛, 渤海大学学报(自然科学版), 2009, 30, 212.)
[16] Singh, R. P.; Majumder, U.; Shreeve, J. M. J. Org. Chem. 2001, 19, 6263.
[17] Lei, J.; Fang, Q.; Yuan, M.-S.; Liu, Z.-Q.; Shen, Y.-X.; Chen, H.-F. Org. Lett. 2010, 22, 5192.
[18] Hennessy, E. J.; Buchwald, S. L. J. Am. Chem. Soc. 2003, 40, 12084.
[19] Prandi, C.; Occhiato, E. G.; Tabasso, S.; Bonfante, P.; Scarpi, D.; Bova, M. E.; Miletto, I. Eur. J. Org. Chem. 2011, 20, 3781.
[20] Hamaue, N.; Mimami, M.; Terado, M.; Hirafuji, M.; Endo, T.; Machida, M.; Hiroshige, T.; Ogata, A.; Tashiro, K.; Saito, H.; Parrez, S. Neurotoxicology 2004. 25, 205.
[21] Igosheva, N.; Lorz, C.; Conner, E. Neurochem. Int. 2005, 47, 216.
[22] Minami, M.; Hamaue, N.; Hirafuji, M.; Saito, H.; Hiroshige, T.; Ogata, A.; Tashiro, K.; Parvez, S. H. J. Neural Transm. Suppl. 2006, 71, 87.
[23] Aulabaugh, A.; Kapoor, B.; Huang, X. Y.; Dollings, P.; Hum, W. T.; Banker, A.; Wood, A.; Ellestad, G. Biochemistry 2007, 46, 9462.
[24] Ogata, A.; Hamaue, N.; Terado, M.; Mimami, M.; Nagashima, K.; Tashiro, K. J. Neurol. Sci. 2003, 206, 79.
[25] Kenner, C.; Rice, B. J.; Boone, A. B.; Rubin, T. J. J. Med. Chem. 1976, 19, 887.
[26] Welstead, Jr.; W. J.; Moran, H. W.; Stauffer, H. F.; Turnbull, L. B.; Sancilio, L. F. J. Med. Chem. 1979, 22, 1074.
[27] Rivalle, C.; Bisagni, E. J. Heterocycl. Chem. 1997, 34, 441.
[28] Gassman, P. G.; Cue Jr. B. W.; Luh, T. Y. J. Org. Chem. 1977, 42, 1344.
[29] Lackey, K.; Besterman, J. M.; Fletcher, W.; Leitner, P.; Morton, B.; Sternbach, D. D. J. Med. Chem. 1995, 38, 906
[30] Garden, S. J.; Torees, J.; Ferrira, A. A.; Silva, R. B.; Pinto, A. C. Tetrahedron Lett. 1997, 38, 1501.
[31] Kawaguchi, H.; Mizuta, Y.; Sugai, F.; Saito, S. EP 19950308606, 1996 [Chem. Abstr. 1996, 125, 181167].
[32] Friedman, S. J. US 3659011, 1972 [Chem. Abstr. 1972, 77, 125053].
[33] Marti, C.; Carreira, E. M. J. Am. Chem. Soc. 127, 11505.
[34] Cui, X. J.; Shi, F.; Zhang, Y. Tetrahedron Lett. 2010, 51, 2048.
[35] Matos, I.; Prez-Mayora, E.; Soriano, E.; Zukal, A.; Martín-Aranda R. M.; López-Peinado, A. J.; Fonseca, I.; ?ejka, J. Chem. Eng. J. 2010, 161, 377.
[36] Reddy, C. R.; Jithender, E. Tetrahedron Lett. 2009, 50, 5633.
[37] Shah, H. C.; Shah, V. H.; Desai, N. D. ChemInform 2010, 40, 540.
[38] Jha, M.; Chou, T. Y.; Blunt, B. Tetrahedron 2011, 67, 982.
/
| 〈 |
|
〉 |